Advanced Manufacturing of Non-Peptide Antithrombin Inhibitors for Global Pharmaceutical Supply Chains
The global demand for effective anticoagulant therapies continues to drive innovation in the synthesis of direct thrombin inhibitors, particularly non-peptide variants that offer superior oral bioavailability. Patent CN100509799C discloses a groundbreaking method for preparing non-chiral, non-peptide antithrombin inhibitors such as BIBR-953 and its prodrug BIBR-1048, which feature a novel 1,2,5-trisubstituted benzimidazole central skeleton. This technological advancement addresses critical limitations in earlier synthetic routes by utilizing 3-nitro-4-chlorobenzoic acid as a cost-effective starting material, thereby establishing a highly efficient pathway for producing these potent pharmaceutical intermediates. The structural elegance of these compounds, characterized by their non-polar lipophilic interaction with thrombin, necessitates a manufacturing process that ensures high purity and consistent stereochemical integrity without the burden of chiral resolution. 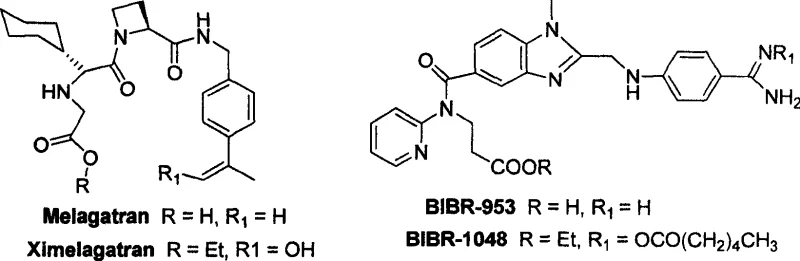
For procurement specialists and supply chain directors, understanding the molecular architecture is essential, as the presence of the fluorine atom or specific benzimidazole substitutions directly influences the pharmacokinetic profile and half-life of the final drug product. The transition from legacy molecules like Ximelagatran to next-generation inhibitors like BIBR-953 represents a significant shift in therapeutic potential, offering extended half-lives of approximately 12 hours compared to the 3-5 hours of previous generations. Consequently, the ability to manufacture these complex heterocyclic systems reliably is paramount for maintaining a steady supply of reliable pharmaceutical intermediate supplier materials to the global market. This report analyzes the technical merits of this patented process, highlighting its capacity for cost reduction in pharmaceutical intermediate manufacturing while ensuring the stringent quality standards required by regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art synthetic routes for generating benzimidazole-based thrombin inhibitors were plagued by significant economic and operational inefficiencies that hindered their viability for large-scale commercial production. The legacy methodology typically commenced with expensive, specialized raw materials that drove up the initial cost basis, creating a substantial financial barrier for generic manufacturers seeking to enter the market. Furthermore, the reliance on N,N-dimethylformamide (DMF) as a primary reaction solvent introduced severe complications in downstream processing, as the high boiling point of DMF necessitates energy-intensive distillation or complex aqueous workups to ensure complete removal from the final active pharmaceutical ingredient. The cumulative effect of these inefficiencies was manifested in a disappointingly low overall yield of approximately 11.1%, meaning that nearly 90% of the input mass was lost as waste or byproducts during the multi-step sequence. Additionally, specific steps involving carbonyldiimidazole (CDI) activation followed by ammoniation with ammonium carbonate resulted in inconsistent conversion rates, further exacerbating the material loss and complicating inventory planning for production managers. These factors combined to create a fragile supply chain vulnerable to raw material price fluctuations and regulatory scrutiny regarding solvent residues.
The Novel Approach
In stark contrast, the innovative process detailed in the patent data revolutionizes the production landscape by introducing a streamlined sequence that prioritizes atom economy and operational simplicity. By shifting the starting point to 3-nitro-4-chlorobenzoic acid, a commodity chemical that is both cheap and readily available in bulk quantities, the process immediately decouples production costs from the volatility of specialized fine chemical markets. The strategic substitution of dichloromethane (CH2Cl2) for DMF in the acylation steps not only facilitates easier solvent recovery through simple evaporation but also significantly improves the safety profile of the manufacturing environment by reducing exposure to reproductive toxins. Crucially, the optimization of reaction conditions, such as the use of saturated ammonia in absolute ethanol for ammoniation instead of ammonium carbonate, has propelled the total yield of the target compound to nearly 50%, representing a four-fold increase in efficiency over conventional methods. This dramatic improvement in yield directly translates to a reduction in the environmental footprint per kilogram of product, aligning with modern green chemistry principles and reducing the burden on waste treatment facilities. 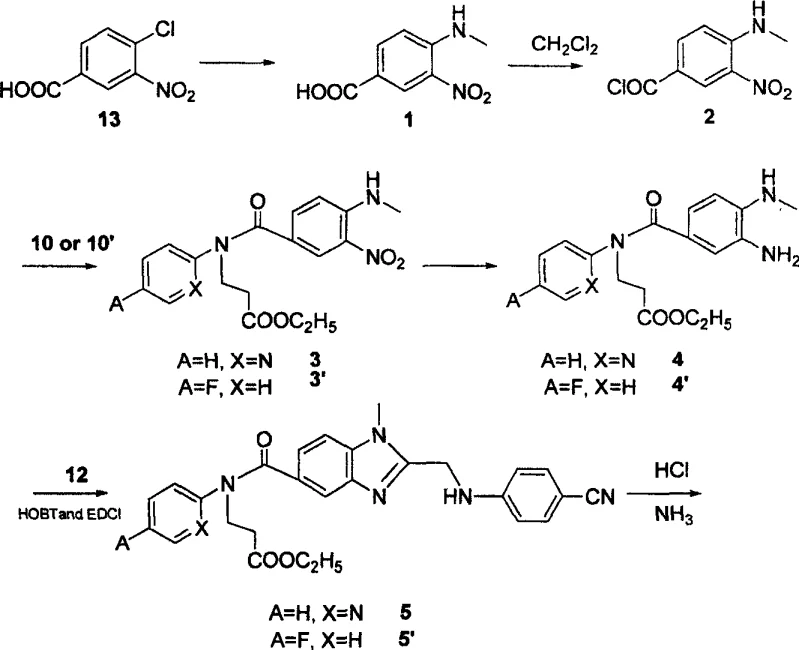
Mechanistic Insights into Benzimidazole Cyclization and Amidation
The core of this synthetic breakthrough lies in the meticulous construction of the 1,2,5-trisubstituted benzimidazole ring system, which serves as the pharmacophore responsible for high-affinity binding to the thrombin active site. The mechanism involves a condensation reaction between an ortho-phenylenediamine derivative and a carboxylic acid component, facilitated by powerful coupling agents such as 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide hydrochloride (EDCI) and 1-hydroxybenzotriazole (HOBT). This coupling strategy is superior to traditional acid chloride methods because it proceeds under milder conditions, minimizing the risk of racemization or degradation of sensitive functional groups elsewhere in the molecule. The subsequent cyclization to form the benzimidazole core is driven by refluxing in acetic acid, a step that effectively promotes dehydration and aromatization to lock the molecular conformation into its bioactive state. For R&D directors, understanding this mechanistic pathway is vital for troubleshooting potential impurities, as incomplete cyclization can lead to open-chain urea byproducts that are difficult to separate and may pose toxicity risks. The robustness of this EDCI-mediated coupling ensures that even on a multi-ton scale, the reaction kinetics remain predictable and controllable, providing a solid foundation for technology transfer.
Impurity control is further enhanced by the specific selection of reagents for the side-chain modifications, particularly the introduction of the pyridine or fluorophenyl moieties which modulate the lipophilicity of the final drug. The process employs a catalytic hydrogenation step using 10% Pd/C at 30 atm to reduce the nitro group to an amine, a transformation that must be carefully monitored to prevent over-reduction or dehalogenation of the aromatic ring. Following this, the amidation with 1-(4-cyano-phenylimine)-acetic acid derivatives is executed with high precision, leveraging the nucleophilicity of the newly formed aniline to attack the activated carboxyl group. The final conversion to the active free acid or the lipophilic prodrug ester is achieved through controlled hydrolysis or esterification, respectively, allowing manufacturers to produce either the immediate API or a stable precursor suitable for formulation. This level of mechanistic control ensures that the impurity profile remains well within the strict limits required for high-purity pharmaceutical intermediate batches, thereby reducing the risk of costly batch rejections during quality control testing.
How to Synthesize BIBR-953 Efficiently
The practical implementation of this synthesis requires adherence to a standardized protocol that maximizes yield while maintaining safety across all unit operations. The process begins with the preparation of key building blocks, such as ethyl 3-(pyridine-2-imine)-propionate, via a Michael addition reaction that sets the stage for subsequent coupling events. Detailed standard operating procedures for each reaction step, including precise temperature controls and stoichiometric ratios, are essential for reproducing the high yields reported in the patent literature. Operators must be trained to handle reagents like thionyl chloride and EDCI with appropriate personal protective equipment, as these materials require careful management to prevent exothermic runaways.
- Preparation of key intermediates including ethyl 3-(pyridine-2-imine)-propionate via Michael addition.
- Coupling of nitro-benzoyl chloride with amine intermediates using triethylamine in dichloromethane.
- Cyclization to form the benzimidazole core followed by amidation and final hydrolysis or esterification.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this optimized synthetic route offers profound benefits for procurement managers tasked with reducing the cost of goods sold (COGS) for anticoagulant therapies. The elimination of expensive starting materials in favor of commodity chemicals creates a structural cost advantage that is resilient to market fluctuations, ensuring long-term price stability for downstream drug manufacturers. Furthermore, the simplified workup procedures resulting from the use of volatile solvents like dichloromethane significantly reduce the energy consumption associated with solvent recovery and drying, contributing to substantial cost savings in utility expenses. The drastic simplification of the purification process, moving away from complex chromatographic separations towards crystallization and filtration, also lowers the capital expenditure required for manufacturing equipment and reduces the cycle time per batch. These efficiencies collectively enhance the margin profile of the product, making it a more attractive candidate for inclusion in generic drug portfolios or cost-sensitive healthcare systems globally.
- Cost Reduction in Manufacturing: The transition to a high-yield process inherently reduces the amount of raw material required to produce a single kilogram of active ingredient, directly lowering the variable cost of production. By avoiding the use of precious metal catalysts that require complex removal steps, the process eliminates the need for expensive scavenger resins or additional purification stages, further driving down operational expenditures. The use of common reagents such as triethylamine and sodium hydroxide ensures that supply chains are not dependent on single-source vendors for exotic chemicals, fostering a competitive purchasing environment. Additionally, the improved throughput resulting from shorter reaction times and easier isolation allows for better utilization of existing reactor capacity, effectively increasing production volume without the need for new capital investment.
- Enhanced Supply Chain Reliability: Sourcing 3-nitro-4-chlorobenzoic acid is significantly more reliable than procuring specialized intermediates, as it is produced by multiple chemical manufacturers worldwide, mitigating the risk of supply disruptions. The robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality, reducing the likelihood of batch failures that can delay shipments to customers. Moreover, the stability of the intermediates generated during the synthesis allows for strategic stockpiling of key precursors, enabling manufacturers to respond rapidly to surges in demand without compromising on lead times. This resilience is critical for maintaining continuity of supply for life-saving medications, ensuring that patients have uninterrupted access to their anticoagulant therapy regardless of external logistical challenges.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing unit operations such as filtration and distillation that are easily transferred from pilot plants to multi-purpose commercial reactors. The reduction in hazardous waste generation, particularly through the avoidance of DMF and the minimization of heavy metal usage, simplifies compliance with increasingly stringent environmental regulations in major manufacturing hubs. This eco-friendly profile not only reduces disposal costs but also enhances the corporate social responsibility standing of the manufacturing organization, appealing to partners who prioritize sustainable supply chains. The ability to scale this process from 100 kgs to 100 MT annual commercial production demonstrates its maturity and readiness for immediate industrial deployment without the need for extensive re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the manufacturing of BIBR-953 and related antithrombin inhibitors. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry stakeholders. Understanding these details is crucial for making informed decisions about sourcing and partnership opportunities in the cardiovascular therapeutic sector.
Q: What is the primary advantage of the new synthesis route for BIBR-953?
A: The new route utilizes cheap and readily available 3-nitro-4-chlorobenzoic acid as a starting material, significantly improving the overall yield to nearly 50% compared to the prior art's 11.1%.
Q: How does the solvent system change impact production safety?
A: Replacing N,N-dimethylformamide (DMF) with dichloromethane (CH2Cl2) simplifies post-treatment procedures and reduces the environmental burden associated with high-boiling polar aprotic solvents.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process uses common reagents like thionyl chloride and EDCI, avoids complex chromatographic separations in later steps, and demonstrates high step-wise yields conducive to scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable BIBR-953 Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure necessary to bring this advanced synthetic route to life on a global scale. Our team of process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering high-purity pharmaceutical intermediate batches that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our dedication to quality assurance means that every shipment is accompanied by comprehensive documentation, providing our partners with the confidence they need to integrate our materials into their own drug development pipelines.
We invite you to collaborate with us to leverage these manufacturing efficiencies for your next project. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your supply chain goals and drive value for your organization.
