Scalable Continuous Manufacturing of Ibrutinib: Advanced Process Technology for Global Supply Chains
Scalable Continuous Manufacturing of Ibrutinib: Advanced Process Technology for Global Supply Chains
The global demand for Bruton's Tyrosine Kinase (BTK) inhibitors has surged due to their critical role in treating hematological malignancies such as Chronic Lymphocytic Leukemia. Patent CN111116593A introduces a groundbreaking continuous and low-cost preparation method for Ibrutinib, addressing significant bottlenecks in traditional synthetic routes. This technology focuses on optimizing the Mitsunobu reaction conditions and implementing a novel workup procedure that allows for the efficient recycling of unreacted starting materials. By shifting away from batch-wise crystallization and column chromatography, this process offers a robust pathway for high-purity API production. For R&D directors and supply chain managers, understanding this continuous flow methodology is essential for securing a reliable ibrutinib supplier capable of meeting stringent quality and volume requirements.
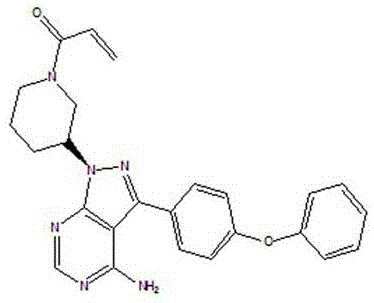
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for Ibrutinib, such as those described in earlier patents like US200680056438.5, rely heavily on the Mitsunobu reaction using excessive amounts of diisopropyl azodicarboxylate (DIAD) and triphenylphosphine. A major drawback of these conventional methods is the generation of substantial quantities of triphenylphosphine oxide, a difficult-to-remove byproduct that complicates downstream purification. Furthermore, existing processes often require silica gel column chromatography to achieve acceptable purity levels, a technique that is notoriously labor-intensive, solvent-heavy, and impractical for multi-ton commercial manufacturing. The inability to effectively recover unreacted 3-(4-phenoxyphenyl)-1H-pyrazolo[3,4-d]pyrimidin-4-amine leads to significant raw material wastage and inflated production costs.
The Novel Approach
The innovative process disclosed in CN111116593A overcomes these inefficiencies by employing a continuous workflow that integrates reaction and separation steps seamlessly. Instead of using a large excess of reagents, this method optimizes the molar ratios of DIAD and triphenylphosphine to near-stoichiometric levels, drastically reducing chemical waste. The most significant advancement lies in the post-reaction treatment, where the reaction mixture undergoes a specific pH-controlled extraction. This allows for the selective separation and recovery of unreacted amine starting material, which can be directly recycled into the next batch. By eliminating the need for intermediate crystallization and final column chromatography, the process achieves high purity through simple washing and filtration steps.
Mechanistic Insights into pH-Controlled Extraction and Acylation
The core chemical innovation of this patent revolves around the precise manipulation of solubility properties during the workup phase. Following the Mitsunobu coupling and subsequent Boc-deprotection with hydrogen chloride gas, the reaction mixture contains the desired intermediate alongside unreacted starting amine and phosphine oxide byproducts. The process utilizes the differential solubility of these components in organic solvents like ethyl acetate and dichloromethane under varying pH conditions. By adjusting the aqueous phase to a specific acidic range, typically between pH 1.8 and 3.5, the unreacted amine is protonated and extracted into the aqueous layer or selectively retained, while the intermediate remains in the organic phase. This mechanistic precision ensures that the valuable heterocyclic amine is not lost but rather recovered with high purity for reuse.
Following the separation, the organic phase containing the deprotected piperidine intermediate is subjected to direct acylation without isolation. The addition of triethylamine and acryloyl chloride proceeds efficiently in the same solvent system, minimizing solvent exchange operations. The final purification is achieved not through chromatography but by stirring the crude reaction mixture with silica gel and activated carbon. This adsorption technique effectively removes colored impurities and residual phosphine oxides, yielding Ibrutinib with an HPLC purity exceeding 99%. This approach demonstrates a sophisticated understanding of impurity profiles, allowing for the production of high-quality active pharmaceutical ingredients suitable for clinical and commercial use.
How to Synthesize Ibrutinib Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for transitioning from laboratory scale to industrial production. The process begins with the careful addition of reagents to maintain thermal control during the exothermic Mitsunobu reaction. Subsequent steps involve gas introduction for deprotection and liquid-liquid extractions that are easily automated in continuous flow reactors. The detailed标准化 synthesis steps见下方的指南 ensure that operators can replicate the high yields and purity consistently. This structured approach minimizes operator error and enhances batch-to-batch reproducibility, which is critical for regulatory compliance in pharmaceutical manufacturing.
- Perform Mitsunobu reaction between 3-(4-phenoxyphenyl)-1H-pyrazolo[3,4-d]pyrimidin-4-amine and Boc-protected hydroxypiperidine using controlled stoichiometry of DIAD and triphenylphosphine.
- Execute Boc deprotection with HCl gas followed by precise pH-adjusted extraction to recover unreacted amine starting material for reuse.
- Conduct direct acylation of the intermediate with acryloyl chloride and purify the final crude product using silica gel and activated carbon stirring instead of column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic implications of this patented process are profound. The primary driver for cost reduction is the drastic decrease in the consumption of expensive reagents like DIAD and triphenylphosphine. By optimizing the stoichiometry and enabling the recycling of the costly heterocyclic amine starting material, the overall material cost per kilogram of Ibrutinib is significantly lowered. Additionally, the elimination of silica gel column chromatography removes a major bottleneck in production throughput, reducing solvent consumption and labor hours associated with purification. These efficiencies translate directly into a more competitive pricing structure for the final API without compromising on quality standards.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings by minimizing the usage of auxiliary chemicals and maximizing raw material utilization. The ability to recover and reuse unreacted starting materials means that less fresh material needs to be purchased for each production cycle. Furthermore, avoiding column chromatography reduces the expenditure on large volumes of chromatographic solvents and silica gel, which are significant cost centers in traditional fine chemical synthesis. The simplified workflow also lowers energy consumption by reducing the number of heating and cooling cycles required for multiple crystallization steps.
- Enhanced Supply Chain Reliability: Adopting a continuous manufacturing process inherently improves supply chain resilience by shortening the overall production lead time. The streamlined nature of the synthesis, which avoids complex isolation steps, allows for faster turnover of batches and more responsive fulfillment of market demand. The robustness of the pH-controlled extraction method ensures consistent output quality, reducing the risk of batch failures that can disrupt supply schedules. This reliability is crucial for maintaining uninterrupted production of downstream drug products in a highly regulated environment.
- Scalability and Environmental Compliance: The reduction in triphenylphosphine oxide waste addresses a critical environmental challenge in pharmaceutical manufacturing. Lower waste generation simplifies effluent treatment and reduces the environmental footprint of the production facility. The process is designed for scalability, meaning it can be easily adapted from pilot plant scales to multi-ton commercial production without losing efficiency. This scalability ensures that suppliers can meet growing global demand for Ibrutinib while adhering to increasingly stringent environmental regulations regarding chemical waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this continuous synthesis technology. Understanding these details helps stakeholders evaluate the feasibility of adopting this method for their specific supply chain needs. The answers are derived directly from the experimental data and process descriptions provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: How does this patent address the high cost of triphenylphosphine oxide waste?
A: The process utilizes a significantly reduced molar ratio of triphenylphosphine and DIAD (approx. 1:1 to 1:1.5) compared to traditional methods that often use excess reagents. This minimizes the generation of triphenylphosphine oxide waste, thereby reducing disposal costs and environmental impact.
Q: What is the key innovation in purifying the intermediate without column chromatography?
A: The method employs a unique pH-controlled extraction strategy where unreacted starting materials are separated based on solubility differences in ethyl acetate versus dichloromethane at specific pH levels (around pH 2.8). This allows the intermediate to proceed to the next step without crystallization or column purification.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the patent explicitly describes a continuous process design that eliminates time-consuming steps like repeated recrystallization and silica gel column chromatography. The use of simple washing and filtration steps makes it highly adaptable for industrial scale-up and continuous manufacturing lines.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ibrutinib Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of continuous manufacturing technologies like the one described in CN111116593A. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex synthetic routes are translated into efficient industrial processes. Our facilities are equipped with rigorous QC labs and advanced processing equipment capable of maintaining stringent purity specifications required for oncology APIs. We are committed to delivering high-purity Ibrutinib intermediates and APIs that meet global regulatory standards.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to leverage these advanced synthesis capabilities. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team to request specific COA data and route feasibility assessments for your upcoming projects. Together, we can optimize your supply chain and secure a sustainable source of high-quality Ibrutinib for the global market.
