Scalable Manufacturing of Ibrutinib Intermediates via Novel Condensation Routes
Scalable Manufacturing of Ibrutinib Intermediates via Novel Condensation Routes
The pharmaceutical industry continuously seeks robust synthetic pathways for kinase inhibitors like Ibrutinib, a critical treatment for mantle cell lymphoma and chronic lymphocytic leukemia. Patent CN113200987A introduces a transformative preparation method that fundamentally restructures the synthesis of Ibrutinib and its key intermediates. Unlike conventional routes that rely on cumbersome protection-deprotection sequences and low-yielding coupling reactions, this innovation leverages a direct condensation strategy starting from low-cost logistics. The core breakthrough lies in the construction of the chiral pyrazolo[3,4-d]pyrimidine scaffold through a highly efficient cyclization of Compound A and Compound B. This approach not only simplifies the operational complexity but also significantly enhances the overall atom economy of the process. By addressing the longstanding challenges of optical purity and waste generation, this technology offers a compelling value proposition for manufacturers aiming to optimize their API supply chains.
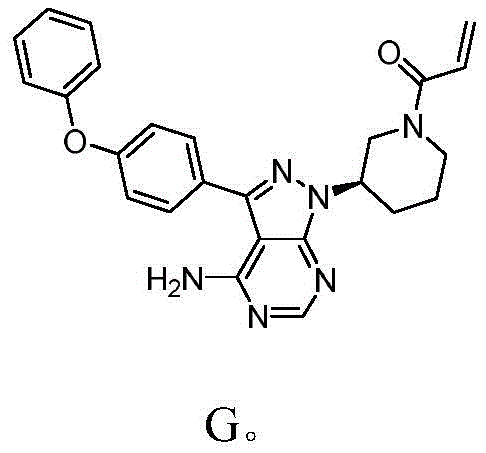
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Ibrutinib has been plagued by inefficient steps that hinder industrial scalability. Prior art, such as the route described in WO2014022390, relies heavily on the Mitsunobu reaction to introduce the chiral center. This specific transformation is notoriously problematic for large-scale operations due to its requirement for stoichiometric amounts of triphenylphosphine and azodicarboxylates, which generate substantial phosphine oxide waste that is difficult to remove. Furthermore, the conventional Suzuki coupling steps often necessitate high catalyst loadings, typically around 0.2 equivalents of tetrakis(triphenylphosphine)palladium, leading to prolonged reaction times of up to 24 hours and increased heavy metal contamination risks. The cumulative effect of these inefficiencies results in a dismal total yield of merely 9.3%, rendering the process economically unviable for cost-sensitive generic production. Additionally, the need for chromatographic purification in earlier stages further exacerbates production costs and limits throughput capacity.
The Novel Approach
In stark contrast, the methodology disclosed in CN113200987A presents a streamlined alternative that bypasses these bottlenecks entirely. The new route initiates with a condensation reaction between Compound A and the chiral hydrazine Compound B, directly establishing the stereocenter without the need for resolution or risky inversion steps. This is followed by a cyclization with formamide to construct the heterocyclic core, a reaction that proceeds under mild conditions with exceptional selectivity. The elimination of the Mitsunobu reaction is a pivotal improvement, as it removes the associated toxic by-products and simplifies the downstream workup to basic filtration and drying. The overall synthetic sequence is depicted in the comprehensive reaction scheme below, illustrating the logical progression from simple precursors to the complex final architecture.
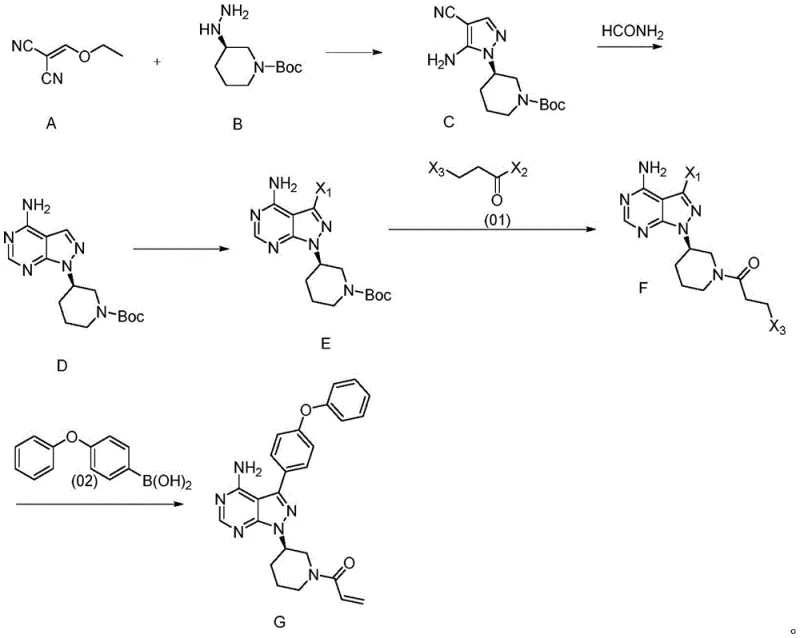
Mechanistic Insights into Condensation and Cyclization
The heart of this synthetic innovation lies in the precise control of the condensation and subsequent cyclization mechanisms. The reaction between Compound A (an ethoxymethylene malononitrile derivative) and Compound B (a chiral hydrazine) proceeds via a nucleophilic attack followed by intramolecular cyclization. Conducting this reaction in ethanol at temperatures between 78°C and 85°C ensures optimal kinetics while maintaining the integrity of the chiral center. The use of ethanol as a solvent is particularly advantageous from a green chemistry perspective, replacing hazardous polar aprotic solvents often used in similar transformations. The resulting Compound C is obtained with a yield exceeding 95% and a purity of 99.5%, demonstrating the high fidelity of this step. This high purity is critical as it minimizes the carryover of impurities into subsequent stages, thereby reducing the burden on purification units later in the pipeline.
Following the formation of Compound C, the construction of the pyrimidine ring is achieved through condensation with formamide. This step effectively closes the bicyclic system to form Compound D. The mechanism involves the nucleophilic attack of the amino group on the nitrile functionality, facilitated by the thermal energy provided during reflux. The patent data indicates that this cyclization can be performed with a molar ratio of formamide to Compound C ranging from 2:1 to 3:1, ensuring complete conversion without excessive reagent waste. The subsequent halogenation of Compound D to form Compound E is carefully controlled to occur at the 3-position of the pyrazole ring, setting the stage for the final Suzuki coupling. This regioselectivity is paramount, as incorrect halogenation would lead to inactive isomers that are difficult to separate. The entire sequence is designed to maximize yield at every junction, ensuring that the final coupling step receives a high-quality substrate.
How to Synthesize Ibrutinib Intermediate Efficiently
The synthesis of the key intermediate Compound C serves as the foundation for the entire Ibrutinib production line. This process is characterized by its operational simplicity and robustness, making it highly suitable for transfer to manufacturing sites. The procedure involves mixing the reactants in a standard reactor, heating to reflux, and isolating the product via filtration, which significantly reduces equipment downtime compared to extraction-heavy protocols. For detailed operational parameters, safety guidelines, and specific workup instructions, please refer to the standardized synthesis protocol provided below.
- React compound A and compound B in ethanol at 78-85°C to form Compound C with high optical purity.
- Cyclize Compound C with formamide under reflux conditions to generate the pyrazolo[3,4-d]pyrimidine core (Compound D).
- Perform halogenation and subsequent acylation to prepare the final coupling precursor Compound F.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route translates into tangible strategic advantages. The primary benefit is the drastic reduction in raw material costs driven by the use of commercially available, low-cost starting materials like Compound A and formamide. By eliminating the need for expensive reagents associated with the Mitsunobu reaction, such as DEAD or DIAD, the direct material cost per kilogram of the intermediate is significantly lowered. Furthermore, the simplified purification process, which relies on crystallization rather than column chromatography, reduces the consumption of silica gel and organic solvents, leading to substantial savings in waste disposal fees. These factors combined create a much leaner cost structure that allows for more competitive pricing in the global API market.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the elimination of high-cost catalytic systems and stoichiometric reagents. In the final Suzuki coupling step, the catalyst loading is reduced to just 0.01 equivalents, a twenty-fold decrease compared to traditional methods that require 0.2 equivalents. This reduction not only lowers the direct cost of palladium catalysts but also minimizes the expense associated with heavy metal scavenging and removal to meet regulatory limits. Additionally, the high yields observed at each step, particularly the over 95% yield in the initial condensation, mean that less raw material is required to produce the same amount of final product, effectively amplifying the purchasing power of the procurement budget.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by reliance on specialized or hazardous reagents that have volatile availability. This new route mitigates such risks by utilizing common solvents like ethanol and water, which are universally available and less subject to supply chain disruptions. The avoidance of light-sensitive reactions and unstable intermediates further stabilizes the production schedule, allowing for consistent batch-to-batch output. Since the process does not require cryogenic conditions or specialized high-pressure equipment, it can be executed in a wider range of manufacturing facilities, thereby diversifying the potential supplier base and reducing dependency on single-source vendors.
- Scalability and Environmental Compliance: As regulatory pressure regarding environmental, social, and governance (ESG) criteria intensifies, the green credentials of this synthesis become a major asset. The process generates significantly less three-waste (wastewater, waste gas, and solid waste) due to the high atom economy and the use of benign solvents. The ability to scale this process from laboratory to commercial tonnage is supported by the straightforward exothermic profile of the reactions and the ease of heat management in ethanol. This scalability ensures that as demand for Ibrutinib grows, the supply chain can expand capacity without encountering the technical bottlenecks typical of more complex synthetic routes, ensuring long-term supply security for downstream partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on performance metrics and operational feasibility. Understanding these details is crucial for technical teams evaluating the integration of this route into existing production frameworks.
Q: How does this new route improve optical purity compared to traditional methods?
A: By utilizing a chiral starting material (Compound B) and avoiding the racemization-prone Mitsunobu reaction, the new process maintains high optical selectivity throughout the synthesis.
Q: What are the environmental benefits of this synthesis method?
A: The process utilizes ethanol as a primary solvent, reduces heavy metal catalyst loading in the Suzuki step to 0.01 equivalents, and eliminates toxic reagents like trimethylsilyldiazomethane.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the route features mild reaction conditions, simple post-treatment procedures like filtration and crystallization, and avoids chromatographic purification, making it ideal for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ibrutinib Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and compliant supply chains in the pharmaceutical sector. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in handling complex heterocyclic chemistry allows us to consistently achieve the high yields and optical purity described in the patent, providing our clients with a reliable source of quality materials.
We invite you to collaborate with us to leverage these advanced synthetic capabilities for your Ibrutinib projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our optimized manufacturing processes can enhance your competitive position in the market.
