Advanced Continuous Manufacturing of Ibrutinib Intermediates for Global Pharma Supply Chains
Advanced Continuous Manufacturing of Ibrutinib Intermediates for Global Pharma Supply Chains
The pharmaceutical industry is constantly seeking robust methodologies to enhance the production efficiency of critical kinase inhibitors like Ibrutinib. Patent CN111116593A introduces a transformative continuous and low-cost preparation method that addresses significant bottlenecks in traditional synthesis routes. This innovation specifically targets the Mitsunobu reaction step, which is pivotal for constructing the core scaffold of the molecule. By optimizing reagent ratios and implementing a sophisticated pH-controlled extraction protocol, the process achieves a comprehensive yield improvement while drastically reducing the environmental footprint associated with triphenylphosphine oxide waste. For R&D directors and supply chain managers, this represents a shift towards more sustainable and economically viable manufacturing paradigms.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for Ibrutinib often rely heavily on the Mitsunobu reaction using substantial excesses of diisopropyl azodicarboxylate (DIAD) and triphenylphosphine to drive conversion. While effective in small-scale laboratory settings, these methods generate massive quantities of triphenylphosphine oxide, a solid waste byproduct that is notoriously difficult and expensive to remove on an industrial scale. Furthermore, conventional processes frequently necessitate silica gel column chromatography for purification, a technique that is labor-intensive, solvent-heavy, and impractical for multi-kilogram or ton-scale production. The inability to effectively recover unreacted starting materials, particularly the valuable pyrazolo-pyrimidine amine, leads to significant raw material loss and inflated production costs.
The Novel Approach
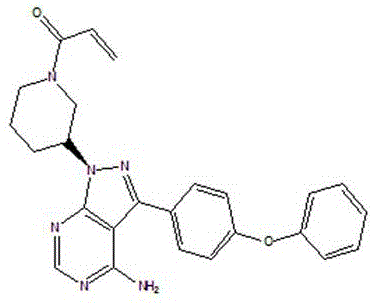
The methodology disclosed in CN111116593A revolutionizes this landscape by introducing a continuous workflow that eliminates the need for column chromatography entirely. Instead of relying on excessive reagents, the process utilizes a tightly controlled molar ratio of DIAD and triphenylphosphine, typically ranging from 0.7 to 1.5 equivalents relative to the substrate. This precision not only reduces the formation of phosphine oxide waste but also simplifies the post-reaction workup. Crucially, the invention incorporates a strategic recycling loop where unreacted 3-(4-phenoxyphenyl)-1H-pyrazolo[3,4-d]pyrimidin-4-amine is recovered through pH-adjusted extraction and reused in subsequent batches. This closed-loop system ensures that raw material utilization is maximized, directly translating to lower cost of goods sold (COGS).
Mechanistic Insights into pH-Controlled Separation and Acylation
The core technical breakthrough lies in the exploitation of differential solubility properties governed by pH levels during the workup phase. Following the initial Mitsunobu coupling and Boc-deprotection using hydrogen chloride gas, the reaction mixture contains the desired intermediate alongside unreacted amine starting material and phosphine byproducts. By introducing concentrated hydrochloric acid and subsequently adjusting the aqueous phase to a specific acidic range of pH 2.8, the process selectively partitions the unreacted amine into an ethyl acetate layer. This allows for the isolation and recrystallization of the starting material with high purity, ready for immediate reuse. The remaining aqueous phase is then basified to pH 11, causing the free base intermediate to precipitate or partition into the organic dichloromethane layer, effectively separating it from water-soluble impurities.
Following the separation, the intermediate undergoes acylation with acryloyl chloride in the presence of triethylamine. Unlike traditional methods that might require extensive purification after this step, the novel process achieves high purity through a simple adsorption treatment. The addition of silica gel and activated carbon directly into the reaction solution acts as a scavenger for residual impurities and colored byproducts. This heterogeneous treatment, followed by filtration and solvent evaporation, yields Ibrutinib with an HPLC purity exceeding 99%. This mechanism bypasses the need for complex crystallization sequences or chromatographic columns, streamlining the path from intermediate to final active pharmaceutical ingredient while maintaining rigorous quality standards required for oncology therapeutics.
How to Synthesize Ibrutinib Efficiently
The synthesis of Ibrutinib via this continuous method involves a sequence of highly optimized unit operations designed for scalability. It begins with the Mitsunobu coupling of the pyrazolo-pyrimidine amine and the protected piperidine alcohol, followed by in-situ deprotection. The critical step involves the pH-swing extraction to recover starting materials, ensuring minimal waste. Finally, the acylation is performed under controlled low temperatures to prevent polymerization of the acryloyl group. For a detailed breakdown of the specific reagent quantities, temperature profiles, and operational parameters described in the patent examples, please refer to the standardized synthesis guide below.
- Perform Mitsunobu reaction using optimized molar ratios of DIAD and triphenylphosphine to minimize waste generation.
- Execute pH-controlled extraction to separate and recover unreacted 3-(4-phenoxyphenyl)-1H-pyrazolo[3,4-d]pyrimidin-4-amine for reuse.
- Conduct direct acylation followed by silica gel and activated carbon treatment to achieve high purity without complex chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this continuous manufacturing process offers substantial strategic benefits beyond mere technical feasibility. The primary advantage is the drastic reduction in auxiliary material costs. By minimizing the usage of expensive reagents like DIAD and triphenylphosphine, and eliminating the need for large volumes of chromatography-grade solvents, the overall variable cost per kilogram of product is significantly lowered. Furthermore, the ability to recycle unreacted high-value starting materials creates a buffer against raw material price volatility, ensuring more stable long-term pricing for downstream partners. This efficiency makes the supply chain more resilient and less susceptible to fluctuations in the global market for fine chemical intermediates.
- Cost Reduction in Manufacturing: The elimination of silica gel column chromatography represents a major operational cost saving. Column chromatography is not only slow but also consumes vast amounts of solvents and silica, which incur high disposal costs. By replacing this with a simple filtration step using silica gel and activated carbon, the process reduces labor hours, solvent purchase costs, and waste treatment fees. Additionally, the reduced stoichiometric requirement for triphenylphosphine means less triphenylphosphine oxide waste is generated, further lowering the environmental compliance costs associated with hazardous waste disposal in pharmaceutical manufacturing facilities.
- Enhanced Supply Chain Reliability: The continuous nature of this process, coupled with the internal recycling of key intermediates, enhances the reliability of supply. Traditional batch processes often suffer from yield losses due to incomplete reactions and difficult purifications, leading to unpredictable output volumes. This optimized method ensures a consistent throughput by recovering and refeeding unreacted materials, thereby maximizing the output from each batch of raw materials. This predictability is crucial for maintaining inventory levels and meeting the stringent delivery schedules demanded by global pharmaceutical clients who require just-in-time delivery of critical oncology ingredients.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this process is inherently more scalable. The reduction in triphenylphosphine oxide waste aligns with green chemistry principles, making it easier to obtain environmental permits for large-scale production facilities. The simplified workup procedure, which avoids complex solvent swaps and multiple crystallization steps, reduces the energy consumption associated with heating and cooling cycles. This energy efficiency, combined with the reduced solvent load, facilitates a smoother scale-up from pilot plant to commercial production, ensuring that the supply chain can rapidly respond to increased market demand without compromising on environmental safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this continuous Ibrutinib synthesis method. These insights are derived directly from the experimental data and process descriptions provided in the patent literature, offering clarity on how this technology translates to practical manufacturing advantages. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing production capabilities.
Q: How does this process reduce triphenylphosphine oxide waste?
A: By optimizing the molar ratio of triphenylphosphine and DIAD to near stoichiometric levels (1:1 to 1:1.5) rather than large excesses, the generation of triphenylphosphine oxide byproducts is significantly minimized, simplifying downstream waste treatment.
Q: What is the mechanism for recovering unreacted starting materials?
A: The process utilizes precise pH control during extraction. By adjusting the aqueous phase to pH 2.8, unreacted 3-(4-phenoxyphenyl)-1H-pyrazolo[3,4-d]pyrimidin-4-amine is selectively extracted into ethyl acetate, allowing it to be recrystallized and reused, thereby improving overall yield.
Q: Is column chromatography required for final purification?
A: No, the novel method eliminates the need for time-consuming silica gel column chromatography. Instead, it employs a simple stirring treatment with silica gel and activated carbon followed by filtration, which drastically reduces processing time and solvent consumption.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ibrutinib Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for high-value kinase inhibitors like Ibrutinib. Our team of expert chemists has extensively analyzed the technological advancements presented in recent patents, including the continuous low-cost preparation methods that optimize Mitsunobu reactions and streamline purification. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of high-quality intermediates. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch meets the exacting standards required for pharmaceutical applications.
We invite potential partners to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific project needs. By leveraging our expertise in process optimization and cost-effective synthesis, we can provide a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance your supply chain efficiency and reduce time-to-market for your therapeutic programs.
