Advanced Asymmetric Oxidation Technology for High-Value Pharmaceutical Intermediates
Advanced Asymmetric Oxidation Technology for High-Value Pharmaceutical Intermediates
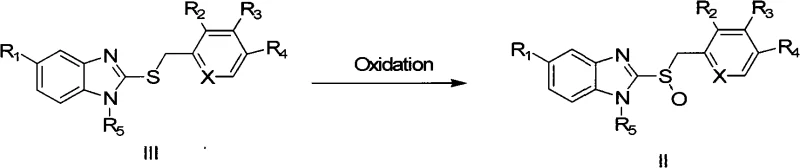
The global demand for proton pump inhibitors (PPIs) such as Omeprazole, Lansoprazole, and Pantoprazole continues to drive innovation in the synthesis of their chiral sulfoxide intermediates. Patent CN101323609A introduces a groundbreaking methodology for the asymmetric oxidation of thioethers to sulfoxides, utilizing a series of novel chiral camphor-derived peroxides. This technology addresses the critical industry need for high enantiomeric purity while simultaneously tackling the economic challenges associated with traditional chiral synthesis routes. By shifting away from expensive transition metal catalysts and inefficient resolution processes, this method offers a robust pathway for the commercial scale-up of complex pharmaceutical intermediates. For R&D directors and procurement specialists, understanding the mechanistic advantages and supply chain implications of this patent is essential for optimizing production strategies and securing a reliable pharmaceutical intermediate supplier partnership.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optically active benzimidazole sulfoxides has relied heavily on the resolution of racemic mixtures or the use of costly chiral metal complexes. Traditional resolution methods, while chemically straightforward, suffer from an inherent theoretical yield limit of 50%, meaning half of the synthesized material is discarded or requires energy-intensive recycling. Furthermore, methods employing chiral metal catalysts, such as the modified Kagan method using titanium tetraisopropoxide and diethyl tartrate, often involve expensive reagents that are difficult to recover and remove from the final product. The presence of heavy metal residues poses significant regulatory hurdles for API manufacturing, necessitating additional purification steps that increase both lead time and operational costs. These conventional approaches often struggle to balance high stereoselectivity with economic viability, creating a bottleneck for the cost reduction in API manufacturing.
The Novel Approach
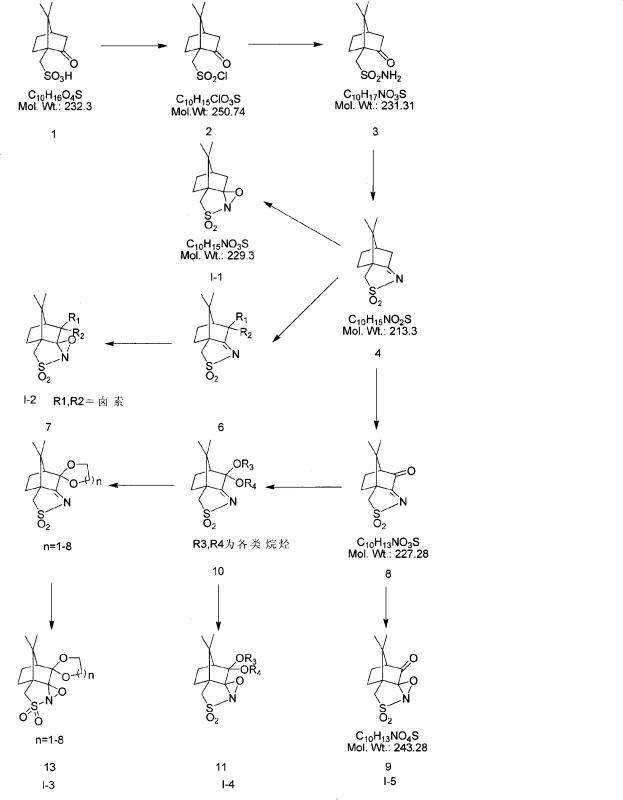
The methodology disclosed in CN101323609A represents a paradigm shift by employing a series of five distinct chiral camphor-based peroxides (labeled I-1 to I-5) as stoichiometric oxidants. Unlike metal-catalyzed systems, these organic peroxides are derived from abundant and inexpensive natural camphor, fundamentally altering the cost structure of the synthesis. The process operates under mild conditions, typically at room temperature in common halogenated solvents, eliminating the need for cryogenic cooling or high-pressure equipment. Crucially, the oxidants can be recovered and reused, and the resulting sulfoxides can be purified to high optical purity through simple recrystallization rather than complex chromatography. This approach not only simplifies the workflow but also enhances the environmental profile of the synthesis by reducing heavy metal waste.
Mechanistic Insights into Camphor-Peroxide Mediated Asymmetric Oxidation
The core of this technology lies in the efficient transfer of an oxygen atom from the chiral peroxide to the sulfur atom of the prochiral thioether substrate. The bulky camphor skeleton creates a rigid chiral environment around the reactive peroxide bond, effectively shielding one face of the sulfur atom during the oxidation event. This steric hindrance forces the oxygen transfer to occur preferentially from one direction, thereby inducing asymmetry and generating the sulfoxide with a specific configuration (R or S). The patent details how modifications to the camphor structure, such as the introduction of bromine, chlorine, or ketal groups at the 6,6-position, fine-tune the electronic and steric properties of the oxidant. These structural variations allow chemists to optimize the enantioselectivity for different thioether substrates, providing a versatile toolkit for synthesizing various PPI analogues.
Impurity control is another critical aspect where this mechanism excels. In traditional oxidation processes, over-oxidation to the sulfone is a common side reaction that degrades yield and complicates purification. The controlled reactivity of the camphor peroxides, combined with the use of specific additives like weak bases or molecular sieves, suppresses this over-oxidation pathway. The additives likely function by modulating the acidity of the reaction medium or by coordinating with transient species to stabilize the transition state leading to the sulfoxide. This precise control ensures that the crude product contains minimal sulfone impurities, reducing the burden on downstream purification units. For quality assurance teams, this means a more consistent impurity profile and a higher probability of meeting stringent pharmacopeial standards without extensive reprocessing.
How to Synthesize Chiral Benzimidazole Sulfoxides Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production. The process begins with the dissolution of the thioether precursor in a solvent such as dichloromethane, followed by the addition of the chiral oxidant and necessary additives. Reaction monitoring is typically conducted via HPLC to ensure complete conversion while minimizing side reactions. Upon completion, the reaction mixture undergoes a straightforward workup involving aqueous washes and acidification to isolate the product. The detailed standardized synthesis steps see the guide below for specific molar ratios and temperature controls optimized for different substrates.
- Dissolve the prochiral thioether substrate in a suitable halogenated hydrocarbon solvent such as dichloromethane or chloroform under controlled temperature conditions.
- Add the chiral camphor-derived peroxide oxidant (Series I-1 to I-5) in a stoichiometric ratio, typically ranging from 1: 0.5 to 1:5 relative to the thioether.
- Introduce specific additives such as weak bases or molecular sieves to enhance stereoselectivity, stir at room temperature until completion, and purify via recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this asymmetric oxidation technology translates into tangible strategic benefits beyond mere chemical elegance. The shift from precious metal catalysts to bio-sourced camphor derivatives decouples the production cost from the volatile pricing of rare earth metals and specialized ligands. This stability in raw material sourcing is crucial for long-term supply contracts and budget forecasting. Furthermore, the ability to recycle the chiral auxiliary significantly lowers the effective cost per kilogram of the active oxidant, contributing to substantial cost savings in the overall manufacturing budget. The simplified purification process also reduces solvent consumption and waste disposal costs, aligning with modern green chemistry initiatives and reducing the environmental compliance burden.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts like titanium and chiral ligands like tartrates removes a major cost driver from the bill of materials. Additionally, the recovery and reuse of the camphor-based oxidants mean that the effective consumption of chiral reagents is drastically minimized. This economic efficiency allows for competitive pricing strategies in the generic pharmaceutical market while maintaining healthy margins. The reduced need for complex chromatographic purification further lowers operational expenditures related to silica gel, solvents, and labor.
- Enhanced Supply Chain Reliability: Camphor is a widely available natural product with a stable global supply chain, unlike specialized chiral ligands which may have limited suppliers. This abundance ensures that production schedules are not disrupted by raw material shortages. The robustness of the reaction conditions, which tolerate room temperature and standard solvents, means that the process can be easily transferred between different manufacturing sites without requiring specialized cryogenic infrastructure. This flexibility enhances supply continuity and reduces the risk of production bottlenecks.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory experiments to multi-ton commercial production. The absence of heavy metals simplifies the regulatory filing process and reduces the environmental footprint of the manufacturing site. Waste streams are easier to treat, and the overall E-factor (mass of waste per mass of product) is improved compared to traditional resolution methods. This alignment with sustainability goals is increasingly important for partnerships with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric oxidation technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this route into their existing production portfolios.
Q: What is the primary advantage of this asymmetric oxidation method over traditional resolution?
A: Unlike traditional resolution which theoretically limits yield to 50%, this direct asymmetric oxidation method utilizes chiral oxidants to convert thioethers directly into the desired enantiomer, significantly improving atom economy and potential yield.
Q: Can the chiral oxidants used in this process be recycled?
A: Yes, the patent explicitly highlights that the chiral camphor-based oxidants are inexpensive, readily available, and can be recovered and reused, which drastically reduces the overall cost of goods for large-scale manufacturing.
Q: What level of enantiomeric excess (ee) can be achieved with this technology?
A: The process initially yields products with moderate to high ee values (e.g., 70-80%), which can be further upgraded to optical purity (>95% ee) through a simple salt formation and recrystallization step, avoiding complex chromatographic separations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Sulfoxide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the asymmetric oxidation technologies described in CN101323609A for the production of high-value PPI intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including precise enantiomeric excess measurements via chiral HPLC, guaranteeing that every batch meets the highest international standards.
We invite you to collaborate with our technical team to explore how this cost-effective and scalable synthesis route can be tailored to your specific product needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis, specific COA data for our reference standards, and comprehensive route feasibility assessments. Let us help you secure a competitive edge in the global pharmaceutical market through superior chemical innovation and reliable supply chain execution.
