Advanced Photoredox Strategy for Scalable 1,1-Diarylalkane Pharmaceutical Intermediates
Introduction to Next-Generation 1,1-Diarylalkane Synthesis
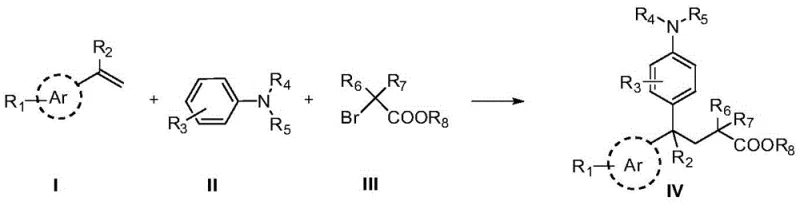
The strategic construction of 1,1-diarylalkane scaffolds represents a critical challenge and opportunity in modern medicinal chemistry, particularly given their prevalence in bioactive molecules targeting autoimmune diseases, cancer, and inflammation. According to third-party patent data CN108675935B, a groundbreaking preparation method has been developed that leverages visible-light photoredox catalysis to assemble these complex structures from readily available starting materials. This innovative approach utilizes para-substituted vinyl compounds, N,N-disubstituted anilines, and 2-bromocarboxylic acid esters as primary building blocks, reacting them under mild, environmentally benign conditions to yield high-purity 1,1-diarylalkane derivatives. For R&D directors and procurement specialists alike, this technology signals a shift away from energy-intensive thermal processes toward sustainable, light-driven synthesis that maintains rigorous quality standards while optimizing resource utilization.
The significance of this method extends beyond mere academic interest; it addresses the pressing industrial need for reliable pharmaceutical intermediate supplier capabilities that can deliver complex scaffolds with consistent purity and efficiency. By operating at room temperature and utilizing visible light, the process inherently reduces the thermal load on reaction systems, minimizing the formation of thermal degradation byproducts that often complicate downstream purification. This patent outlines a versatile platform where the electronic and steric properties of the substrates can be finely tuned, allowing for the generation of diverse chemical libraries essential for drug discovery pipelines. The integration of earth-abundant metal co-catalysts further enhances the economic viability of the route, making it a compelling candidate for cost reduction in pharmaceutical intermediate manufacturing without compromising on the structural integrity required for subsequent biological testing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,1-diarylalkane compounds has relied heavily on the use of donor-acceptor (D-A) cyclopropanes as key precursors, a strategy that, while effective, introduces significant logistical and safety burdens to the manufacturing process. Traditional protocols often necessitate stringent reaction conditions, including elevated temperatures or the use of strong Lewis acids, which can limit the functional group tolerance and lead to the decomposition of sensitive moieties present in advanced intermediates. Furthermore, the preparation of D-A cyclopropanes themselves is often a multi-step sequence involving hazardous reagents, thereby increasing the overall step count and reducing the cumulative yield of the final active pharmaceutical ingredient. From a supply chain perspective, reliance on such specialized and potentially unstable starting materials creates vulnerabilities, as any disruption in the supply of these niche precursors can halt entire production lines. Additionally, the removal of heavy metal residues or acidic byproducts from these conventional reactions often requires extensive workup procedures, generating substantial chemical waste and increasing the environmental footprint of the synthesis.
The Novel Approach
In stark contrast, the novel photoredox-mediated strategy described in the patent data offers a streamlined, direct assembly of the 1,1-diarylalkane core through a radical addition mechanism that bypasses the need for pre-functionalized cyclopropane intermediates. By employing a dual catalytic system consisting of a ruthenium-based photocatalyst and a copper salt co-catalyst, the reaction proceeds efficiently under ambient temperature and visible light irradiation, typically using blue LEDs. This mildness allows for the preservation of delicate functional groups such as esters, ethers, and halides, which might otherwise be compromised under harsher thermal conditions. The use of simple styrene derivatives and anilines as starting materials significantly broadens the scope of accessible chemical space, enabling the rapid exploration of structure-activity relationships. Moreover, the operational simplicity of the new method—requiring only standard glassware and LED light sources—facilitates easier commercial scale-up of complex pharmaceutical intermediates, as it eliminates the need for specialized high-pressure or high-temperature equipment often associated with traditional cross-coupling reactions.
Mechanistic Insights into Photoredox-Catalyzed Radical Addition
The underlying mechanism of this transformation involves a sophisticated interplay between photo-excited species and radical intermediates, initiated by the absorption of visible light by the ruthenium photocatalyst, typically [Ru(bipy)3]Cl2·6H2O. Upon irradiation, the photocatalyst enters an excited state capable of engaging in single-electron transfer (SET) processes with the 2-bromocarboxylic acid ester substrate. This interaction generates a reactive carbon-centered radical species along with an oxidized photocatalyst, which is subsequently regenerated through a redox cycle involving the copper co-catalyst and the amine substrate. The copper salt, preferably CuCl, plays a pivotal role in mediating the radical addition to the vinyl double bond, ensuring high regioselectivity and preventing uncontrolled polymerization of the styrene derivative. This precise control over the radical lifecycle is crucial for maintaining high product purity, as it minimizes the formation of homocoupling byproducts or oligomers that are common pitfalls in free-radical chemistry. The presence of a base, such as potassium carbonate, further assists in neutralizing acidic byproducts and facilitating the turnover of the catalytic cycle, ensuring that the reaction proceeds to completion with minimal accumulation of intermediates.
From an impurity control perspective, the room temperature operation is a decisive factor in achieving the high purity specifications demanded by regulatory bodies. Thermal reactions often suffer from non-selective activation of bonds, leading to a complex impurity profile that requires costly and time-consuming chromatographic separations. In this photoredox system, the energy input is highly specific to the electronic transitions of the catalyst, meaning that only the intended bond activations occur, leaving other parts of the molecule untouched. This chemoselectivity is particularly advantageous when synthesizing intermediates for drugs like sertraline analogs or omexifene derivatives, where the presence of trace impurities can have significant toxicological implications. The ability to monitor the reaction progress easily via TLC or GC-MS allows for real-time optimization, ensuring that the reaction is quenched at the point of maximum conversion before any secondary degradation pathways can become dominant. This level of process control translates directly into a more robust and predictable manufacturing workflow, reducing the risk of batch failures and ensuring consistent quality across different production runs.
How to Synthesize 1,1-Diarylalkane Derivatives Efficiently
The practical execution of this synthesis is designed to be straightforward and adaptable to various laboratory and pilot plant settings, leveraging standard equipment to achieve high efficiency. The process begins with the careful charging of a Schlenk sealed tube reactor with the requisite molar ratios of the para-substituted vinyl compound, the N,N-disubstituted aniline, and the 2-bromocarboxylic acid ester, typically in a 1:2:2 ratio to drive the equilibrium towards product formation. Following the addition of the catalytic system comprising the ruthenium complex and copper chloride, along with the inorganic base and acetonitrile solvent, the reaction vessel is sealed and purged with an inert gas such as argon or nitrogen to prevent oxygen quenching of the excited photocatalyst. The mixture is then subjected to irradiation from a 3W to 5W blue LED light source while being stirred at room temperature, a condition that is maintained until monitoring indicates complete consumption of the starting material, usually within a few hours. Detailed standardized synthesis steps are provided in the guide below.
- Charge a Schlenk sealed tube reactor with para-substituted vinyl compounds, N,N-disubstituted anilines, and 2-bromocarboxylic acid esters alongside the photocatalyst and copper salt.
- Add the organic solvent and base under an inert atmosphere, then irradiate the mixture with a blue LED light source at room temperature while monitoring progress.
- Upon completion, concentrate the reaction mixture under reduced pressure and purify the crude residue via silica gel column chromatography to isolate the target product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photoredox technology presents a compelling value proposition centered on operational resilience and long-term cost efficiency. The shift from thermal to photochemical activation fundamentally alters the energy profile of the synthesis, removing the need for energy-intensive heating mantles or cryogenic cooling systems, which translates to a significant reduction in utility costs over the lifespan of the production campaign. Furthermore, the reliance on visible light and earth-abundant copper co-catalysts mitigates the supply risks associated with volatile precious metal markets, ensuring a more stable and predictable cost structure for raw materials. The simplicity of the reaction setup also means that existing manufacturing infrastructure can often be repurposed for this chemistry with minimal capital expenditure, accelerating the time-to-market for new drug candidates. By streamlining the synthetic route and reducing the number of purification steps required, the overall throughput of the facility can be increased, effectively reducing lead time for high-purity pharmaceutical intermediates and allowing companies to respond more agilely to market demands.
- Cost Reduction in Manufacturing: The elimination of expensive palladium catalysts and the replacement of specialized cyclopropane precursors with commodity chemicals like styrenes and anilines drives down the direct material costs substantially. Additionally, the mild reaction conditions reduce the wear and tear on reactor vessels and associated equipment, lowering maintenance overheads and extending the operational life of the manufacturing assets. The high atom economy of the radical addition process ensures that a greater proportion of the input mass is converted into valuable product, minimizing waste disposal fees and maximizing the yield per batch. These factors combine to create a leaner, more cost-effective production model that enhances the overall profitability of the pharmaceutical intermediate supply chain without sacrificing quality.
- Enhanced Supply Chain Reliability: Sourcing strategies are greatly improved by the use of widely available starting materials that are produced on a multi-ton scale by the global chemical industry, reducing the risk of shortages that can plague niche reagent markets. The robustness of the photocatalytic system, which tolerates a wide range of substituents and functional groups, allows for flexibility in supplier selection, as slight variations in raw material grades can often be accommodated without impacting the final product quality. This flexibility builds a more resilient supply chain capable of withstanding geopolitical or logistical disruptions. Moreover, the ability to perform the reaction at room temperature simplifies storage and transportation requirements for the reaction mixture, reducing the logistical complexity and cost associated with temperature-controlled shipping of intermediates between different processing sites.
- Scalability and Environmental Compliance: The transition from batch to continuous flow processing is naturally facilitated by photochemical reactions, offering a clear pathway for scaling production from grams to tons while maintaining precise control over light exposure and residence time. This scalability is crucial for meeting the growing demand for complex intermediates in the generic and branded drug markets. Environmentally, the process aligns with green chemistry principles by utilizing non-toxic solvents like acetonitrile which can be recovered and recycled, and by avoiding the generation of hazardous heavy metal waste streams. The reduced energy consumption and lower waste output contribute to a smaller carbon footprint, helping manufacturers meet increasingly stringent environmental regulations and corporate sustainability goals, thereby enhancing the brand reputation and market competitiveness of the final pharmaceutical products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photoredox synthesis platform, drawing directly from the experimental data and scope defined in the patent literature. Understanding these nuances is essential for R&D teams evaluating the feasibility of integrating this chemistry into their existing workflows and for procurement officers assessing the long-term viability of the supply source. The answers provided reflect the proven capabilities of the method as demonstrated in the optimization studies and substrate scope evaluations, offering a realistic view of its potential and limitations in a commercial setting.
Q: What are the key advantages of this photoredox method over traditional D-A cyclopropane routes?
A: Unlike conventional methods requiring donor-acceptor cyclopropanes and harsh conditions, this visible-light protocol operates at room temperature with broader substrate tolerance and eliminates the need for expensive transition metals like palladium.
Q: Can this synthesis be scaled up for commercial production?
A: Yes, the patent data indicates successful scale-up trials maintaining high yields. The use of standard LED lighting and common solvents like acetonitrile facilitates easy adaptation to flow chemistry or large batch reactors.
Q: Is the photocatalyst recoverable to reduce long-term costs?
A: The methodology supports the use of robust ruthenium-based photocatalysts which, combined with earth-abundant copper co-catalysts, offers a cost-effective profile suitable for repeated cycles in industrial settings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,1-Diarylalkane Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced photoredox methodologies in accelerating the development of next-generation therapeutics. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with specialized photochemical reactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 1,1-diarylalkane intermediate delivered meets the highest international standards. We are committed to leveraging our technical expertise to optimize these novel routes, providing our clients with a competitive edge through superior quality and consistent supply reliability.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis strategy can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this photoredox platform for your pipeline. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your drug development programs. Let us be your partner in turning complex chemical challenges into commercial successes.
