Advanced Manufacturing of Pranoprofen: A Technical Breakthrough for Global API Supply Chains
Advanced Manufacturing of Pranoprofen: A Technical Breakthrough for Global API Supply Chains
The pharmaceutical industry continuously seeks robust synthetic routes for non-steroidal anti-inflammatory drugs (NSAIDs) that balance high purity with economic efficiency. A significant advancement in this domain is detailed in patent CN115611908A, which discloses a novel preparation method for Pranoprofen, a potent analgesic and antirheumatic agent. This technology addresses critical bottlenecks in existing manufacturing protocols by integrating phase transfer catalysis with a sophisticated ultrasonic-assisted crystallization technique. For R&D directors and procurement managers overseeing the supply of high-purity pharmaceutical intermediates, this patent represents a pivotal shift towards more reliable and cost-effective production methodologies. By optimizing the rearrangement step and refining the final purification, the process ensures a superior impurity profile and enhanced overall yield, making it an attractive candidate for commercial scale-up in the competitive generic drug market.
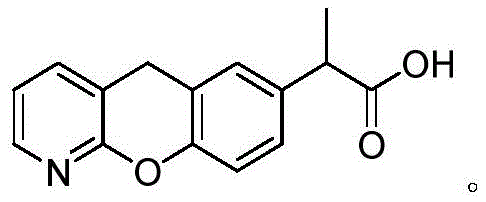
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Pranoprofen has relied on methods such as those published by Jinrong et al., which typically involve a Friedel-Crafts acylation followed by hydrolysis and acidification. While functional, these legacy processes often struggle with significant inefficiencies, particularly during the rearrangement and purification stages. Traditional acidification and rearrangement steps frequently result in incomplete conversions and the formation of difficult-to-remove byproducts, leading to crude products with suboptimal purity. Furthermore, standard crystallization techniques using ethanol often fail to adequately separate these impurities, necessitating multiple recrystallization cycles that drastically reduce the final yield. In comparative studies referenced in the background art, yields for the final active pharmaceutical ingredient often hovered around 48.5%, representing a substantial loss of valuable starting materials and increased waste generation. These inefficiencies translate directly into higher production costs and longer lead times for supply chain managers.
The Novel Approach
The methodology outlined in CN115611908A introduces a transformative two-pronged strategy to overcome these historical deficits. First, it incorporates trimethylbenzyl ammonium chloride as a specialized phase transfer catalyst during the critical sulfonyl chloride rearrangement step. This specific quaternary ammonium salt is selected not merely for its phase transfer capabilities but for its unique steric properties, which help regulate the reaction pathway and minimize side reactions. Second, the patent details an innovative crystallization protocol where the dissolved crude product is split into two fractions. A small portion is cooled and subjected to ultrasonic treatment to generate precise seed nuclei, which are then introduced into the main heated batch. This controlled nucleation ensures uniform crystal growth and superior exclusion of impurities. The result is a streamlined process that delivers Pranoprofen with significantly improved yield and purity metrics compared to the prior art, effectively solving the long-standing issues of low recovery rates in commercial manufacturing.
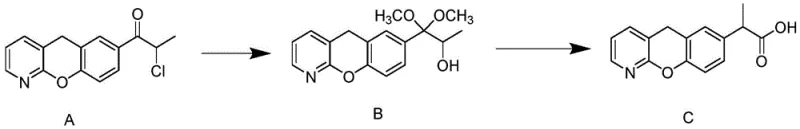
Mechanistic Insights into Phase Transfer Catalyzed Rearrangement
The core chemical innovation lies in the second step of the synthesis, where the dimethyl acetal intermediate (Compound B) undergoes acidification and rearrangement to form the propionic acid side chain. In this stage, the reaction mixture comprises dichloromethane, triethylamine, and the crucial additive, trimethylbenzyl ammonium chloride. Mechanistically, the phase transfer catalyst facilitates the transport of ionic species across the interface between the organic and aqueous phases, thereby increasing the collision frequency between reactants. However, the distinct advantage of the trimethylbenzyl group over simpler alkyl chains is its steric bulk. This bulkiness appears to modulate the transition state of the sulfonyl chloride acylation, favoring the desired rearrangement pathway while sterically hindering the formation of polymeric or oligomeric impurities. This precise control over the reaction microenvironment allows the process to proceed efficiently at room temperature followed by mild heating, avoiding the harsh conditions that typically degrade sensitive heterocyclic structures like the benzopyrano-pyridine core found in Pranoprofen.
Following the reaction, the workup procedure is designed to maximize recovery while maintaining high purity standards. The reaction mixture is quenched into ice water, and the pH is carefully adjusted to an alkaline range of 11-12 using sodium hydroxide. This step converts the acidic product into its water-soluble salt form, allowing for the extraction of neutral organic impurities into the dichloromethane layer. Subsequently, the aqueous layer is re-acidified to a pH of 4-5, precipitating the Pranoprofen crude product. This acid-base extraction sequence is highly effective at removing non-acidic byproducts and residual starting materials. The combination of the specialized catalyst and this rigorous purification workup ensures that the crude material entering the final crystallization step is already of high quality, reducing the burden on the final purification stage and contributing to the overall high yield of the process.
How to Synthesize Pranoprofen Efficiently
The synthesis of Pranoprofen via this patented route involves three distinct operational stages that can be seamlessly integrated into existing multipurpose reactor setups. The process begins with the formation of the acetal intermediate using sodium methoxide, followed by the catalytic rearrangement, and concludes with the ultrasonic-assisted crystallization. Each step is optimized for safety and scalability, utilizing standard solvents and reagents available in the global chemical supply chain. The following guide outlines the standardized operational parameters derived from the patent examples, providing a clear roadmap for technical teams aiming to implement this high-efficiency route. For detailed standard operating procedures and specific equipment requirements, please refer to the technical guidelines below.
- React Compound A with sodium methoxide in methanol at 55-60°C to form the dimethyl acetal intermediate (Compound B).
- Perform acidification and rearrangement of Compound B using sulfonyl chloride in dichloromethane, utilizing trimethylbenzyl ammonium chloride as a phase transfer catalyst.
- Purify the crude product by dissolving in ethanol-water, splitting the solution, generating seed crystals via ultrasonics in the smaller fraction, and inducing crystallization in the main batch.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers compelling economic and logistical benefits that extend beyond simple yield improvements. The primary advantage lies in the drastic simplification of the purification workflow. By achieving higher purity in the crude stage through effective phase transfer catalysis and controlled crystallization, manufacturers can significantly reduce the number of recrystallization cycles required. This reduction directly correlates to lower solvent consumption, decreased energy usage for heating and cooling, and reduced labor hours, all of which contribute to substantial cost savings in the final cost of goods sold (COGS). Furthermore, the robustness of the process minimizes the risk of batch failures due to out-of-specification purity, ensuring a more predictable and reliable supply of the active ingredient for downstream formulation.
- Cost Reduction in Manufacturing: The implementation of trimethylbenzyl ammonium chloride eliminates the need for expensive or hazardous reagents often used in older rearrangement protocols. By enhancing the reaction efficiency and selectivity, the process maximizes the conversion of raw materials into the final product, thereby reducing the cost per kilogram of Pranoprofen produced. Additionally, the high yield reduces the volume of waste solvent and chemical byproducts that require disposal, leading to further operational cost reductions and a smaller environmental footprint. The qualitative improvement in process efficiency means that facilities can produce more output with the same fixed asset base, improving capital utilization rates.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as methanol, dichloromethane, and ethanol ensures that the supply chain is not vulnerable to shortages of exotic or specialized reagents. The process conditions, ranging from 0°C to 65°C, are well within the operational limits of standard glass-lined or stainless steel reactors found in most contract manufacturing organizations (CMOs). This compatibility means that the technology can be transferred to multiple manufacturing sites globally without requiring significant capital investment in new infrastructure. Consequently, buyers can secure a more resilient supply network with multiple qualified sources capable of executing this standardized, robust protocol.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, the process is designed with scalability in mind. The use of a phase transfer catalyst allows for milder reaction conditions, reducing the thermal load on the reactor and minimizing the risk of runaway exotherms. The crystallization step, which avoids the use of large volumes of anti-solvents by utilizing a split-solution technique, reduces the total volume of effluent generated. This aligns with modern green chemistry principles and helps manufacturers meet increasingly stringent environmental regulations. The ability to scale from kilogram to multi-ton production while maintaining consistent quality makes this route ideal for meeting the growing global demand for NSAIDs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented Pranoprofen synthesis method. These answers are derived directly from the experimental data and technical disclosures within CN115611908A, providing clarity on the process capabilities and advantages. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer and for procurement professionals assessing the long-term value proposition of this manufacturing route.
Q: How does the new method improve upon traditional Pranoprofen synthesis yields?
A: The patented method introduces trimethylbenzyl ammonium chloride as a phase transfer catalyst during the rearrangement step. Unlike conventional alkylammonium salts, its specific steric hindrance regulates the acylation rearrangement more effectively, preventing side reactions and significantly boosting the conversion rate compared to prior art methods which often suffer from low yields around 48%.
Q: What is the role of ultrasonic treatment in the purification process?
A: The process utilizes a unique split-solution crystallization technique where a small fraction of the dissolved product is subjected to ultrasonic treatment while cooling. This generates extremely fine and uniform crystal nuclei which, when reintroduced to the main hot solution, act as superior seeds. This controls the crystal growth kinetics, resulting in higher purity and easier filtration compared to standard cooling crystallization.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the route relies on common industrial solvents such as methanol, dichloromethane, and ethanol, and operates at moderate temperatures (0°C to 65°C). The elimination of complex transition metal catalysts and the use of robust phase transfer catalysis make the process highly amenable to scale-up from kilogram to multi-ton production without requiring exotic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pranoprofen Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to maintain competitiveness in the global pharmaceutical market. Our technical team has extensively analyzed the methodology described in CN115611908A and possesses the expertise to implement this phase transfer catalyzed process at an industrial scale. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless and efficient. Our facility is equipped with rigorous QC labs and stringent purity specifications that guarantee every batch of Pranoprofen meets the highest international pharmacopoeia standards, providing our partners with absolute confidence in product quality.
We invite pharmaceutical companies and generic drug manufacturers to collaborate with us to leverage this cutting-edge technology for their supply chains. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you optimize your Pranoprofen supply chain with a solution that delivers superior purity, enhanced yield, and long-term commercial reliability.
