Advanced Synthesis of Pranoprofen: Overcoming Cryogenic Limitations for Commercial Scalability
The pharmaceutical industry continuously seeks robust synthetic pathways that balance high purity with operational efficiency, particularly for non-steroidal anti-inflammatory drugs (NSAIDs) like Pranoprofen. Patent CN116041358A presents a significant technological breakthrough in the preparation of this tricyclic propionic acid derivative, addressing long-standing challenges associated with traditional rearrangement reactions. Historically, the synthesis of Pranoprofen has been plagued by the necessity for严苛 cryogenic conditions and the persistent formation of structurally similar impurities that complicate downstream purification. This new methodology leverages advanced sulfonation activating reagents, such as p-toluenesulfonyl chloride, methanesulfonic anhydride, and trifluoromethanesulfonic anhydride, to facilitate a smoother rearrangement under alkaline conditions. By shifting the reaction paradigm, the inventors have successfully reduced the generation of byproducts, specifically targeting the notorious Alkene Impurity A, which is notoriously difficult to remove via standard crystallization techniques. Furthermore, the process dramatically relaxes the stringent temperature control requirements, moving from deep sub-zero environments to much more manageable thermal ranges. This evolution represents a critical step forward for manufacturers aiming to optimize cost reduction in pharmaceutical intermediates manufacturing while ensuring the highest standards of product quality and regulatory compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Conventional synthetic routes for Pranoprofen, such as those disclosed in Japanese Patent JP1992288081A, rely heavily on the use of anhydrous aluminum chloride and subsequent rearrangement steps involving sulfonyl chlorides or thionyl chloride. These legacy processes impose severe operational constraints, most notably the requirement to maintain reaction temperatures at or below -20°C to prevent runaway side reactions. Such cryogenic conditions demand specialized refrigeration equipment and result in substantial energy overheads, creating a bottleneck for commercial scale-up of complex pharmaceutical intermediates. Moreover, even with strict temperature adherence, these methods frequently suffer from chlorination elimination side reactions that generate Pranoprofen Alkene Impurity A. This specific impurity possesses a chemical structure remarkably similar to the target molecule, rendering it extremely resistant to removal through standard washing or recrystallization protocols. Consequently, manufacturers often face diminished overall yields and increased processing times as they attempt to purge these stubborn contaminants to meet pharmacopoeial standards. The reliance on hazardous reagents like thionyl chloride also introduces additional safety and environmental handling concerns that further complicate the supply chain logistics.
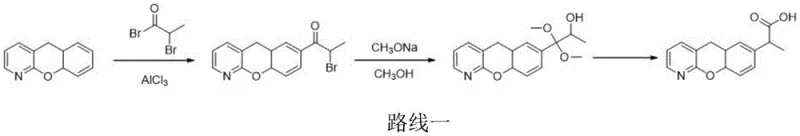
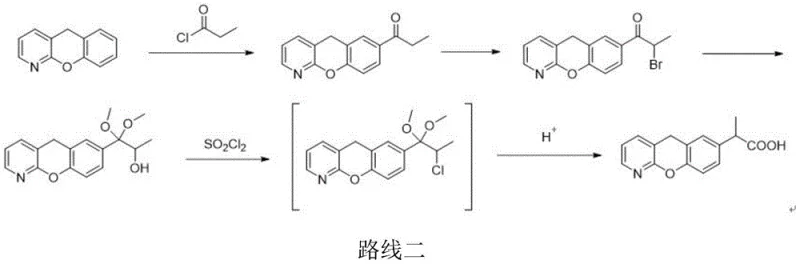
The Novel Approach
In stark contrast to these cumbersome legacy methods, the novel approach detailed in the patent utilizes a sophisticated sulfonation activation strategy that fundamentally alters the reaction kinetics and thermodynamics. By employing activating reagents like trifluoromethanesulfonic anhydride in the presence of organic bases such as pyridine or triethylamine, the reaction proceeds efficiently at temperatures ranging from -20°C to 50°C, with a preferred window of -5°C to 30°C. This significant widening of the operational temperature window eliminates the need for expensive cryogenic cooling, thereby simplifying the reactor setup and reducing utility costs. The new pathway facilitates a cleaner rearrangement that inherently suppresses the elimination mechanisms responsible for forming Alkene Impurity A. As demonstrated in the experimental data, this method achieves superior purity profiles, with examples showing purity levels reaching 99.96% and impurity A content dropping to negligible levels of 0.01%. This represents a transformative improvement for any entity acting as a reliable pharmaceutical intermediates supplier, as it ensures a more consistent and high-quality output. The streamlined workflow, which involves a direct activation followed by hydrolysis, reduces the number of unit operations and minimizes solvent usage, aligning perfectly with modern green chemistry principles.
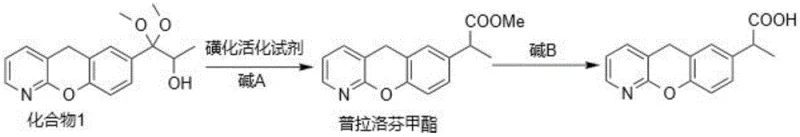
Mechanistic Insights into Sulfonation Activation and Rearrangement
The core innovation of this synthesis lies in the precise selection of the sulfonating activating reagent, which dictates the stability of the intermediate and the trajectory of the rearrangement. In traditional methods using thionyl chloride, the intermediate chloro-species is prone to beta-elimination under basic or thermal stress, leading directly to the formation of the alkene double bond found in Impurity A. However, the use of anhydrides like methanesulfonic anhydride or trifluoromethanesulfonic anhydride generates a different activated species, likely a sulfonate ester, which exhibits greater stability against elimination. This stability allows the rearrangement to proceed via the desired migration pathway without competing degradation reactions. The presence of a mild organic base, such as N,N-diisopropylethylamine or pyridine, serves to scavenge the acidic byproducts generated during activation without inducing the harsh deprotonation that triggers elimination. This delicate balance of reactivity ensures that the carbon skeleton remains intact while the functional groups are correctly positioned for the final hydrolysis step. For R&D directors, understanding this mechanistic nuance is crucial for troubleshooting and optimizing the process further, as it highlights the importance of reagent purity and base selection in maintaining the integrity of the tricyclic system.
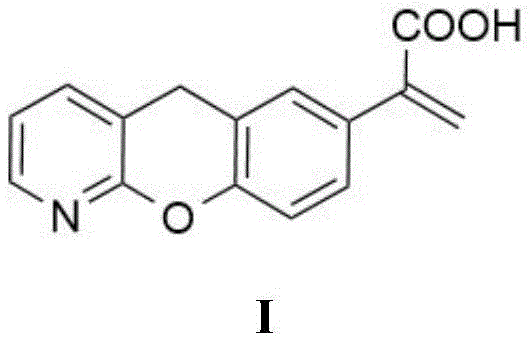
Furthermore, the control of Impurity A is not merely a matter of yield optimization but is critical for meeting stringent regulatory specifications for ophthalmic NSAIDs. The structural similarity of Impurity A to Pranoprofen means that standard chromatographic or crystallization techniques often fail to separate them effectively, leading to potential batch rejections. The new mechanism effectively shuts down the formation pathway of this impurity at the source, rather than attempting to remove it post-formation. By operating at milder temperatures (0°C to 10°C in preferred embodiments), the kinetic energy available for the elimination reaction is insufficient to overcome the activation barrier, whereas the desired rearrangement proceeds readily. This kinetic selectivity is the key to achieving the reported purity of >99.9%. Additionally, the hydrolysis step using sodium hydroxide in methanol/water mixtures is robust and forgiving, allowing for complete conversion of the methyl ester intermediate without epimerization or further degradation. This comprehensive control over the reaction landscape ensures that the final API intermediate possesses a clean impurity profile, significantly reducing the burden on quality control laboratories and accelerating the release of finished drug products.
How to Synthesize Pranoprofen Efficiently
The synthesis of Pranoprofen via this novel sulfonation activation route offers a practical and scalable solution for industrial manufacturing. The process begins with the dissolution of the precursor Compound 1 in a dry aprotic solvent, typically dichloromethane, under an inert atmosphere to prevent moisture interference. The addition of the activating reagent is carefully controlled to manage exotherms, followed by a stirring period that allows the rearrangement to reach completion. Subsequent hydrolysis converts the intermediate ester into the final acid, which is isolated through a straightforward acidification and filtration sequence. This streamlined approach minimizes the need for complex chromatographic purifications, making it ideal for large-scale production. For detailed operational parameters, stoichiometry, and specific workup procedures, please refer to the standardized synthesis guide below.
- Dissolve the precursor Compound 1 and a base (such as triethylamine or pyridine) in a dry solvent like dichloromethane, then add a sulfonating activating reagent (e.g., trifluoromethanesulfonic anhydride) under inert gas protection at mild temperatures (-20 to 50°C) to form the methyl ester intermediate.
- Subject the resulting pranoprofen methyl ester intermediate to hydrolysis using a strong base such as sodium hydroxide or potassium hydroxide to cleave the ester group and yield the final Pranoprofen acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthesis route translates into tangible strategic advantages that extend beyond simple chemical yield. The elimination of extreme low-temperature requirements removes a significant barrier to entry for many contract manufacturing organizations, as it negates the need for specialized cryogenic reactors and the associated high energy consumption. This shift allows for greater flexibility in selecting manufacturing partners and reduces the overall cost of goods sold (COGS) by lowering utility overheads. Furthermore, the drastic reduction in difficult-to-remove impurities means that fewer batches are rejected due to out-of-specification results, thereby enhancing supply continuity and reliability. The simplified purification process also reduces the volume of solvents and reagents required per kilogram of product, contributing to a smaller environmental footprint and lower waste disposal costs. These factors collectively create a more resilient and cost-effective supply chain for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The transition from cryogenic conditions (-20°C) to mild temperatures (-5°C to 30°C) results in substantial energy savings by eliminating the need for continuous deep-freeze cooling. Additionally, the suppression of Alkene Impurity A reduces the need for extensive re-crystallization cycles or preparative chromatography, which are both time-consuming and solvent-intensive processes. The use of commercially available and relatively inexpensive activating reagents further optimizes the raw material cost structure. By streamlining the workflow and reducing the number of processing steps, manufacturers can achieve a significantly lower cost per kilogram, allowing for more competitive pricing in the global market without compromising on quality margins.
- Enhanced Supply Chain Reliability: The robustness of the new method against temperature fluctuations ensures consistent batch-to-batch quality, which is critical for maintaining long-term supply agreements with major pharmaceutical companies. The reduced risk of batch failure due to impurity excursions means that production schedules are less likely to be disrupted by unexpected reworks or investigations. Moreover, the reagents used, such as trifluoromethanesulfonic anhydride and common organic bases, are widely available from multiple global suppliers, mitigating the risk of raw material shortages. This diversification of the supply base enhances the overall security of the supply chain, ensuring that reducing lead time for high-purity pharmaceutical intermediates becomes a achievable reality rather than just a goal.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, as the thermal management requirements are easily met with standard industrial jacketed reactors rather than specialized cryogenic vessels. This ease of scale-up facilitates a smoother transition from pilot plant trials to multi-ton commercial production, accelerating time-to-market for new drug formulations. From an environmental perspective, the reduction in solvent usage and the avoidance of hazardous chlorinating agents like thionyl chloride align with increasingly strict global environmental regulations. The simpler workup procedure generates less hazardous waste, simplifying disposal and reducing the environmental compliance burden on the manufacturing facility, thus supporting sustainable chemical manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced Pranoprofen synthesis method. These answers are derived directly from the technical specifications and comparative data presented in the patent literature, providing a clear overview of the process capabilities. Understanding these details is essential for stakeholders evaluating the feasibility of adopting this technology for their own production lines or sourcing strategies. The focus is on clarifying the operational benefits and the specific improvements over prior art methods.
Q: How does the new sulfonation method improve upon traditional low-temperature rearrangement processes?
A: Traditional methods utilizing sulfonyl chloride or thionyl chloride require strict cryogenic conditions (below -20°C) to suppress side reactions. The novel sulfonation activation method described in CN116041358A operates effectively at significantly milder temperatures (preferably -5 to 30°C), drastically reducing energy consumption and equipment complexity while minimizing the formation of hard-to-remove alkene impurities.
Q: What is the primary advantage regarding impurity control in this synthesis route?
A: The primary advantage is the substantial reduction of Pranoprofen Alkene Impurity A. Conventional routes often generate this structurally similar byproduct via elimination side reactions, which is extremely difficult to purge. The new activation strategy using reagents like trifluoromethanesulfonic anhydride fundamentally alters the reaction pathway to suppress this elimination, achieving purities exceeding 99.9%.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is highly scalable. By eliminating the need for extreme low-temperature control (-20°C), the method simplifies thermal management in large reactors. The use of common solvents like dichloromethane and standard bases, combined with a robust workup procedure involving pH adjustment and recrystallization, ensures consistent quality and safety from kilogram to multi-ton scales.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pranoprofen Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the global pharmaceutical market. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the sulfonation activation route for Pranoprofen can be seamlessly transferred to industrial scale. We are committed to delivering products with stringent purity specifications, leveraging our rigorous QC labs to verify that every batch meets or exceeds the highest international standards. Our infrastructure is designed to handle complex organic syntheses with precision, guaranteeing supply continuity and quality consistency for our partners.
We invite you to collaborate with us to explore how this optimized synthesis route can benefit your specific project requirements. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating the economic advantages of switching to this superior method. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a reliable supply of high-quality Pranoprofen for your future endeavors.
