Revolutionizing Vitamin A Intermediate Production via Efficient 4-Acetoxyl-2-Methyl-2-Butenal Synthesis
The global demand for Vitamin A and its derivatives continues to drive innovation in the synthesis of critical C5 intermediates, specifically 4-acetoxyl-2-methyl-2-butenal, often referred to in the industry as pentacarbonaldehyde. A groundbreaking technical disclosure found in patent CN110143875B introduces a streamlined, high-efficiency preparation method that addresses long-standing bottlenecks in yield and cost. This novel approach leverages 4-hydroxybutyraldehyde as a foundational building block, initiating a concise three-step sequence involving methylenation, catalytic isomerization, and final acetylation. For R&D directors and procurement strategists, this patent represents a pivotal shift away from legacy processes that rely on scarce resources and complex purification protocols. By optimizing the reaction pathway to achieve a total yield exceeding 72%, this technology offers a robust framework for reliable pharmaceutical intermediate supplier networks aiming to secure stable production capacities for high-value vitamins and fine chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of pentacarbonaldehyde has been plagued by inefficient multi-step sequences that erode profit margins and complicate supply chain logistics. For instance, early methodologies described in US5453547 utilized dimethoxyacetone as a starting material, necessitating a lengthy reaction cascade involving acetylene addition, hydrogenation, and subsequent hydrolysis. As illustrated in the reaction scheme below, this traditional pathway suffers from a cumulative total yield of merely 35-45%, rendering it economically unsustainable for modern high-volume manufacturing.
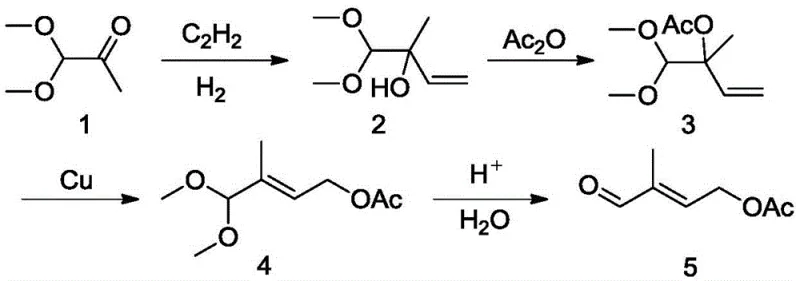
Furthermore, alternative routes such as those disclosed in US4124619 depend heavily on expensive noble metal catalysts like rhodium and palladium under high-pressure carbon monoxide conditions. These processes not only incur substantial capital expenditure for catalyst recovery systems but also introduce significant safety risks associated with high-pressure gas handling. Additionally, routes originating from isoprene or ethylene oxide often generate copious amounts of saline wastewater or involve extremely unstable intermediates that are difficult to control, leading to batch-to-batch variability and increased environmental compliance costs for agrochemical intermediate and pharma manufacturers alike.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the methodology outlined in CN110143875B presents a radically simplified synthetic architecture that prioritizes atom economy and operational simplicity. By selecting 4-hydroxybutyraldehyde—a common and inexpensive fine chemical intermediate—as the feedstock, the new process eliminates the need for hazardous acetylene gas or unstable epoxide ring-opening reactions. The core innovation lies in the strategic use of (N,N-dimethyl)methylene ammonium iodide to introduce the exocyclic methylene group, followed by a highly selective isomerization step. This approach drastically reduces the number of unit operations required, thereby minimizing solvent consumption and energy usage. The result is a streamlined workflow that enhances cost reduction in pharmaceutical intermediate manufacturing while delivering a product purity profile that meets the stringent specifications required for downstream Vitamin A synthesis.
Mechanistic Insights into Catalytic Isomerization and Esterification
The heart of this synthetic breakthrough is the precise control over the carbon skeleton rearrangement during the isomerization phase. The process begins with the reaction of 4-hydroxybutyraldehyde with (N,N-dimethyl)methylene ammonium iodide in the presence of a strong base system, typically comprising n-butyllithium and an organic amine such as diisopropylamine. This generates the key intermediate, 2-methylene-4-hydroxybutyraldehyde (Structure I), with high fidelity. Subsequently, this intermediate undergoes a catalytic isomerization to form 4-hydroxy-2-methyl-2-butenal (Structure II). The patent highlights the efficacy of transition metal carbonyl clusters, specifically Ru3(CO)12, in facilitating this double bond migration. Unlike phosphine-ligated catalysts which can be air-sensitive and costly, these carbonyl complexes offer robust activity at moderate temperatures (80-120°C), ensuring consistent conversion rates without the degradation issues seen in earlier palladium-catalyzed systems.
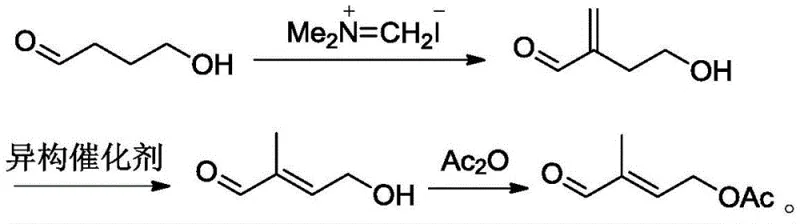
Following the isomerization, the final transformation involves the acetylation of the allylic alcohol moiety using acetic anhydride. This step is critical for stabilizing the molecule for subsequent condensation reactions in Vitamin A production. The reaction conditions are optimized to proceed at temperatures between 100°C and 120°C, ensuring complete conversion while minimizing side reactions such as polymerization or ether formation. Impurity control is maintained through rigorous vacuum rectification protocols, where fractions are collected at specific boiling points under reduced pressure (1 kPa). This meticulous attention to distillation parameters ensures that the final 4-acetoxyl-2-methyl-2-butenal (Structure III) is obtained with a purity exceeding 99%, effectively removing trace isomers and unreacted starting materials that could otherwise poison downstream catalysts in the final coupling steps.
How to Synthesize 4-Acetoxyl-2-Methyl-2-Butenal Efficiently
The implementation of this synthesis route requires careful attention to stoichiometry and temperature control across three distinct stages to maximize the reported >72% total yield. The process initiates with the generation of a lithiated amine base at cryogenic temperatures to ensure selective deprotonation, followed by the addition of the methylenating agent. The subsequent isomerization step demands an inert atmosphere and precise catalyst loading to prevent over-reduction or decomposition. Finally, the acetylation is driven to completion using a slight excess of acetic anhydride, followed by a neutralization and purification sequence. For detailed operational parameters, reagent grades, and specific equipment setups required to replicate these results, please refer to the standardized protocol below.
- React 4-hydroxybutyraldehyde with (N,N-dimethyl)methylene ammonium iodide in the presence of a base to generate 2-methylene-4-hydroxybutyraldehyde.
- Catalytically isomerize 2-methylene-4-hydroxybutyraldehyde using a Ruthenium or Palladium catalyst to obtain 4-hydroxy-2-methyl-2-butenal.
- Perform an esterification reaction between 4-hydroxy-2-methyl-2-butenal and acetic anhydride to yield the final product, 4-acetoxyl-2-methyl-2-butenal.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this patented methodology offers profound benefits for supply chain resilience and total cost of ownership. The primary driver of value is the substitution of exotic or hazardous starting materials with 4-hydroxybutyraldehyde, a commodity chemical available from multiple global suppliers. This diversification of the raw material base mitigates the risk of supply disruptions that often plague single-source precursor markets. Furthermore, the elimination of expensive organic phosphine reagents and high-loading noble metal catalysts directly translates to lower variable costs per kilogram of output. By removing the dependency on rhodium and specialized phosphine ligands, manufacturers can insulate their production budgets from the volatility of precious metal markets, ensuring more predictable pricing structures for long-term contracts.
- Cost Reduction in Manufacturing: The economic advantage of this process is anchored in its high atom efficiency and reduced catalyst burden. Traditional routes often require stoichiometric amounts of costly reagents or high loadings of palladium and rhodium, which necessitate complex recovery units to be economically viable. In contrast, this new route utilizes catalytic amounts of ruthenium carbonyls which are significantly more affordable and easier to handle. The simplification of the purification train, moving from complex chromatographic separations to standard vacuum distillation, further reduces utility costs and labor hours. Consequently, the overall manufacturing cost is substantially lowered, providing a competitive edge in the pricing of high-purity pharmaceutical intermediates without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of robust, non-hazardous reagents that do not require special transport permits or storage conditions. Unlike processes relying on acetylene gas or ethylene oxide, which pose significant logistical and safety challenges, the reagents in this synthesis are liquid or solid at ambient conditions. This ease of handling facilitates smoother inventory management and reduces the lead time for high-purity pharmaceutical intermediates by minimizing regulatory hurdles and safety inspections. Additionally, the high yield of the process means that less raw material is required to produce the same amount of finished goods, effectively increasing the throughput capacity of existing manufacturing facilities and reducing the frequency of raw material replenishment orders.
- Scalability and Environmental Compliance: Environmental sustainability is increasingly a prerequisite for maintaining a social license to operate in the fine chemical sector. This synthesis route generates significantly less wastewater and saline byproducts compared to the hypochlorite oxidation methods used in isoprene-based routes. The absence of heavy metal waste streams simplifies effluent treatment and reduces the burden on waste management infrastructure. Moreover, the process is inherently scalable; the reaction conditions are mild enough to be safely transferred from pilot plant to multi-ton commercial reactors without encountering the heat transfer or mixing limitations often associated with high-pressure carbonylation reactions. This scalability ensures that commercial scale-up of complex pharmaceutical intermediates can be achieved rapidly to meet surging market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on yield expectations, catalyst selection, and process safety. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing production portfolios.
Q: What are the primary advantages of this new synthesis route compared to traditional methods?
A: The new route utilizes 4-hydroxybutyraldehyde, a cheap and readily available starting material, avoiding the expensive phosphine and noble metal catalysts required in prior art. It achieves a total yield exceeding 72%, significantly higher than the 35-45% typical of conventional dimethoxyacetone-based routes, while generating less wastewater.
Q: Which catalysts are preferred for the isomerization step in this process?
A: The patent specifies several effective catalysts for the isomerization of 2-methylene-4-hydroxybutyraldehyde, including Ru3(CO)12, Pd(OAc)2, and RhCl3. Among these, Ru3(CO)12 is particularly preferred for its balance of activity and cost-effectiveness compared to rhodium-based alternatives.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is designed for scalability. It employs standard unit operations such as vacuum rectification and uses common solvents like toluene and tetrahydrofuran. The elimination of highly unstable intermediates found in ethylene oxide routes further enhances its safety and feasibility for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Acetoxyl-2-Methyl-2-Butenal Supplier
As the global pharmaceutical landscape evolves, the ability to access high-quality intermediates through efficient and sustainable pathways becomes a critical differentiator. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging deep expertise in process chemistry to bring innovative solutions like the CN110143875B route to commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial application is seamless. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 4-acetoxyl-2-methyl-2-butenal meets the exacting standards required for Vitamin A synthesis.
We invite procurement leaders and R&D directors to collaborate with us to explore how this advanced synthesis method can optimize your supply chain. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and security in your vitamin and fine chemical supply chains.
