Revolutionizing Vitamin A Intermediate Production: A Noble-Metal-Free Catalytic Strategy
Revolutionizing Vitamin A Intermediate Production: A Noble-Metal-Free Catalytic Strategy
The global demand for high-purity Vitamin A acetate and its derivatives continues to drive innovation in fine chemical manufacturing, specifically regarding the efficiency of key intermediate synthesis. Patent CN113233979B introduces a groundbreaking preparation method for 4-acetoxyl-2-methyl-2-butenal (commonly known as C5 aldehyde), a critical building block in the C15+C5 synthetic route developed by major industry players like BASF. This technology addresses long-standing inefficiencies in the double bond isomerization step, traditionally a bottleneck characterized by low yields and reliance on costly noble metal catalysts. By leveraging a synergistic catalyst system comprising a cobalt acid complex and a metal chloride, this invention achieves high reaction speeds and superior selectivity without the economic burden of palladium or rhodium. For procurement leaders and R&D directors seeking a reliable pharmaceutical intermediates supplier, this patent represents a pivotal shift towards more sustainable and cost-effective manufacturing protocols that can be scaled from pilot plants to multi-ton commercial production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of C5 aldehyde has been plagued by significant technical and economic hurdles across four primary routes. The dimethoxyacetone route, while established, involves hazardous nitrated compounds and suffers from a lengthy process with low total yield, creating safety liabilities and high operational costs. Similarly, the ethylene oxide route faces challenges with the instability of acetoxyacetaldehyde intermediates and poor selectivity due to self-condensation side reactions. Perhaps most critically, the widely adopted butenediol route, particularly the variation developed by Roche, relies heavily on noble metal catalysts like palladium for the double bond isomerization step. As illustrated in the traditional pathways below, these legacy methods often struggle with conversion rates hovering around 60% and selectivity issues that necessitate complex recycling loops and extensive purification, driving up the cost of goods sold (COGS) for downstream vitamin manufacturers.
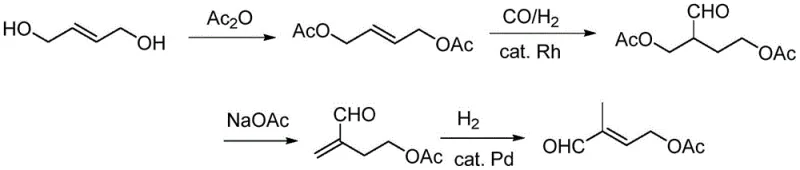
The Novel Approach
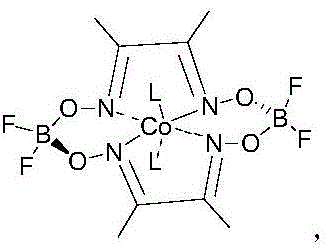
The methodology disclosed in CN113233979B fundamentally reengineers the isomerization step by replacing expensive noble metals with an earth-abundant cobalt-based system. Instead of using palladium, the process utilizes a specific cobalt acid complex, Co(dmgBF₂)₂L₂, in conjunction with a metal chloride promoter. This novel approach not only eliminates the volatility associated with precious metal pricing but also dramatically improves the chemical efficiency of the transformation. The reaction proceeds under moderate hydrogen pressure and temperature conditions, converting 4-acetoxyl-2-methylbutanal directly into the target C5 aldehyde with exceptional precision. By shifting away from the constraints of the traditional Roche route, this method offers a streamlined pathway that enhances overall throughput and simplifies the supply chain for high-purity pharmaceutical intermediates, making it an attractive option for large-scale industrial adoption.
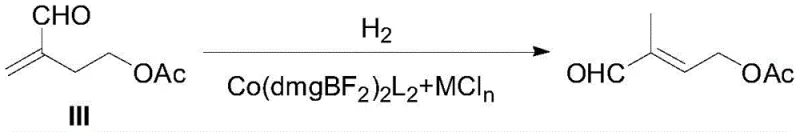
Mechanistic Insights into Cobalt-Chloride Synergistic Catalysis
The core innovation of this technology lies in the sophisticated interplay between the cobalt acid complex and the added metal chloride, which functions as a Lewis acid. In traditional radical-mediated isomerization processes using cobalt alone, the reaction often suffers from slow kinetics and significant side reactions, such as radical polymerization and unwanted hydrogenation, which can account for up to 20% of byproduct formation. The introduction of the metal chloride, preferably stannous chloride or ferric chloride, coordinates with the carbonyl group of the substrate. This coordination stabilizes the transient radical intermediate formed during the hydrogen atom transfer process. By enhancing the stability of this intermediate, the catalyst system effectively suppresses the competing polymerization pathways that typically degrade yield. Furthermore, the increased concentration of the stabilized radical species accelerates the overall reaction rate, allowing the process to reach maximum conversion in significantly less time compared to non-promoted cobalt systems.
From an impurity control perspective, this mechanistic refinement is crucial for meeting the stringent quality standards required for vitamin synthesis. The suppression of hydrogenation byproducts reduces the formation of saturated aldehydes, which are difficult to separate from the target unsaturated product due to similar boiling points. Additionally, minimizing polymeric heavies ensures that the downstream distillation columns operate more efficiently with less fouling. The result is a cleaner reaction profile where selectivity for the desired C5 isomer exceeds 90%, a substantial improvement over the roughly 70% selectivity observed in earlier cobalt-only attempts. This level of control demonstrates a deep understanding of organometallic chemistry, providing R&D teams with a robust platform for cost reduction in pharmaceutical intermediates manufacturing by minimizing waste and maximizing raw material utilization.
How to Synthesize 4-Acetoxyl-2-Methyl-2-Butenal Efficiently
The complete synthetic sequence begins with commercially available 1,4-butenediol, undergoing acetylation to form the diacetate, followed by hydroformylation and decarboxylation to generate the isomerization precursor. The final critical step involves the cobalt-catalyzed isomerization under hydrogen pressure. The detailed standardized operating procedures, including precise molar ratios, solvent choices, and workup protocols necessary for replicating this high-yield process, are outlined below.
- Acetylation of 1,4-butenediol with acetic anhydride or acetic acid at 120°C to form 1,4-butenediol diacetate.
- Hydroformylation of the diacetate using a Rhodium catalyst under CO/H2 pressure to yield 2-formyl-1,4-butanediol diacetate.
- Decarboxylation to obtain 4-acetoxyl-2-methylbutanal, followed by isomerization using Co(dmgBF2)2L2 and stannous chloride under H2 pressure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this cobalt-based methodology offers tangible strategic benefits beyond simple chemical curiosity. The primary advantage is the decoupling of production costs from the volatile noble metal market. By eliminating the need for palladium or rhodium, manufacturers can secure a more predictable cost structure, insulating their operations from geopolitical supply shocks that frequently impact precious metal availability. Furthermore, the improved selectivity and reduced byproduct formation translate directly into lower downstream processing costs. Less energy is required for purification, and solvent consumption is reduced, contributing to a leaner, more efficient manufacturing footprint that aligns with modern sustainability goals.
- Cost Reduction in Manufacturing: The substitution of noble metals with inexpensive cobalt and tin salts drastically lowers the direct material cost of the catalyst system. Since the catalyst loading is low and the metals are abundant, the overall expenditure on catalytic materials is significantly reduced. Additionally, the higher selectivity means less raw material is wasted on side products, improving the effective yield per ton of input. This efficiency gain allows for substantial cost savings in the final product price, enhancing competitiveness in the global market for vitamin precursors.
- Enhanced Supply Chain Reliability: Relying on earth-abundant metals like cobalt and iron/tin ensures a stable supply of catalytic materials, unlike palladium which is subject to severe supply constraints. The robustness of the catalyst system also implies longer catalyst life and easier handling, reducing the frequency of reactor shutdowns for catalyst replacement. This reliability is critical for maintaining continuous production schedules and meeting tight delivery windows for downstream pharmaceutical clients who depend on just-in-time inventory models.
- Scalability and Environmental Compliance: The process operates at moderate temperatures and pressures, utilizing standard stainless steel reactors rather than specialized high-pressure equipment required for some alternative routes. The reduction in heavy metal waste simplifies effluent treatment, lowering the environmental compliance burden. The ability to scale this reaction from laboratory benchtop to multi-ton commercial production without significant re-engineering makes it an ideal candidate for rapid capacity expansion to meet surging market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cobalt-catalyzed isomerization technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy for technical due diligence.
Q: Why is the cobalt-chloride catalyst system superior to traditional palladium catalysts for C5 synthesis?
A: Traditional Roche routes rely on expensive noble metals like palladium, which suffer from low conversion rates (around 61%) and selectivity issues. The novel cobalt acid complex combined with a metal chloride (Lewis acid) stabilizes radical intermediates, significantly boosting selectivity to over 90% while eliminating the high cost of precious metals.
Q: What are the critical reaction conditions for the isomerization step?
A: The process operates efficiently at moderate temperatures between 40-60°C and hydrogen pressures of 30-60 atm. The use of aromatic solvents like toluene and specific ligands (such as THF or water) in the cobalt complex further optimizes reaction speed and yield.
Q: How does this method impact the purity profile of the final Vitamin A intermediate?
A: By suppressing competing radical polymerization and hydrogenation side reactions, this method reduces heavy byproduct formation from roughly 20% in prior art to approximately 5%. This results in a crude product that is easier to purify to the stringent 99% content required for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Acetoxyl-2-Methyl-2-Butenal Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced intermediates like 4-acetoxyl-2-methyl-2-butenal requires more than just a patent; it demands engineering excellence and rigorous quality assurance. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this cobalt-catalyzed route are fully realized in practice. Our facilities are equipped with state-of-the-art hydrogenation reactors and stringent purity specifications are maintained through our rigorous QC labs, guaranteeing that every batch meets the exacting standards required for Vitamin A synthesis.
We invite global partners to collaborate with us to leverage this innovative technology for their supply chains. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to discuss specific COA data and route feasibility assessments, ensuring that your transition to this next-generation manufacturing process is seamless, compliant, and economically advantageous.
