Advanced Synthesis of Sofosbuvir Intermediate via Lewis Acid Catalyzed Reduction
The global demand for direct-acting antiviral agents, particularly for the treatment of Hepatitis C Virus (HCV), has necessitated the development of robust and scalable synthetic routes for key nucleoside analogs. Central to this therapeutic class is Sofosbuvir, a breakthrough molecule that has revolutionized HCV management. The efficient production of its critical intermediates remains a focal point for process chemists aiming to secure supply chains. Patent CN110669085B discloses a significant technological advancement in the preparation of the Sofosbuvir intermediate ((2R,3R,4R)-3-(benzoyloxy)-4-fluoro-5-hydroxy-4-methyltetrahydrofuran-2-yl)benzoate, designated herein as Formula I. This innovation addresses long-standing bottlenecks in nucleoside synthesis by introducing a selective reduction protocol that operates under mild conditions.
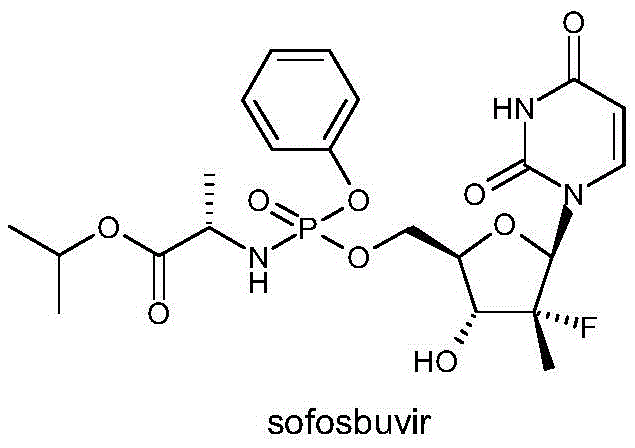
As a reliable pharmaceutical intermediate supplier, understanding the structural nuances of the final active pharmaceutical ingredient is crucial for optimizing precursor synthesis. The molecule features a complex stereocenter arrangement that must be preserved throughout the manufacturing process to ensure biological efficacy. The intermediate described in this patent serves as a pivotal building block, linking the sugar moiety to the phosphoramidate prodrug section. By refining the synthesis of this specific tetrahydrofuran derivative, manufacturers can significantly enhance the overall throughput of the final drug substance, thereby supporting the continuous availability of life-saving medications for patients worldwide.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this specific nucleoside intermediate has been plagued by harsh reaction conditions and cumbersome purification protocols. Prior art, such as the route disclosed in WO2010135569A, typically relies on the use of Red-Al (sodium bis(2-methoxyethoxy)aluminum hydride) in toluene solutions. This conventional approach necessitates cryogenic reaction temperatures, often as low as -40°C, to attempt to control the reduction selectivity. Such extreme thermal requirements impose substantial energy costs and demand specialized refrigeration equipment, creating a barrier to efficient commercial scale-up of complex pharmaceutical intermediates.
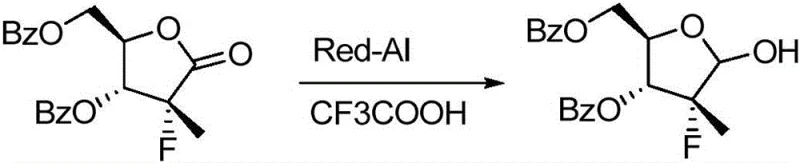
Furthermore, the use of aluminum-based hydrides generates significant quantities of aluminum salt by-products. These salts have a notorious tendency to adsorb the organic product, making isolation extremely difficult and leading to substantial material loss during elution. Consequently, the crude product invariably requires purification via column chromatography to achieve acceptable purity levels. This reliance on chromatographic separation is a major bottleneck in industrial chemistry, as it consumes vast amounts of silica gel and organic solvents, drastically increasing both the environmental footprint and the production timeline while limiting the overall reaction yield to approximately 78%.
The Novel Approach
In stark contrast, the methodology presented in Patent CN110669085B offers a transformative solution by replacing hazardous aluminum hydrides with a synergistic combination of borohydrides and Lewis acids in an organic amine medium. This novel system allows the reduction of the lactone precursor (Formula II) to proceed effectively at temperatures ranging from -10°C to 40°C, with optimal results observed between 10°C and 30°C. This shift to near-ambient conditions not only enhances operational safety but also simplifies the engineering requirements for reactor systems, facilitating cost reduction in API manufacturing.
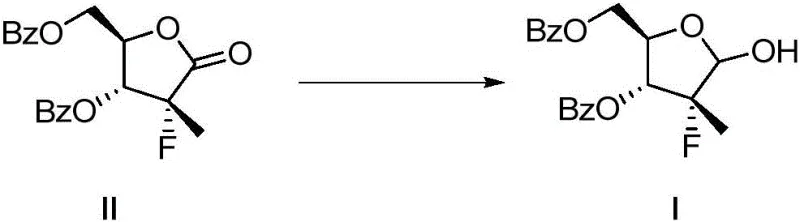
Perhaps most critically, this new chemical strategy exhibits exceptional selectivity, effectively suppressing the formation of over-reduced by-products that plague simpler borohydride reductions. The result is a crude reaction mixture of such high quality that it bypasses the need for column chromatography entirely. Instead, the product can be isolated through standard aqueous workup procedures involving extraction and acid washing. This streamlined downstream processing enables the recovery of Formula I with purity exceeding 99% and yields reaching up to 90%, representing a substantial improvement in material efficiency and process economics compared to legacy methods.
Mechanistic Insights into Lewis Acid-Borohydride Synergistic Reduction
The core innovation of this process lies in the precise modulation of the reducing power of the borohydride species through coordination with a Lewis acid. In the absence of a Lewis acid, simple borohydrides like sodium borohydride or potassium borohydride often lack the necessary chemoselectivity, leading to the over-reduction of the lactone ring to the corresponding diol, a side reaction observed to generate up to 82% of unwanted by-products in comparative studies. However, when a Lewis acid such as zinc chloride (ZnCl2) or aluminum trichloride (AlCl3) is introduced into the organic amine solvent, it coordinates with the carbonyl oxygen of the substrate and potentially the hydride source.
This coordination creates a more controlled reactive species that favors the reduction of the lactone to the hemiacetal (lactol) state without further reducing the ring oxygen or affecting other sensitive functional groups like the benzoyl esters. The patent data indicates that the molar ratio of the substrate to the borohydride and Lewis acid is critical, with a preferred range of 1:(0.3~0.7):(0.3~0.7). This stoichiometric balance ensures that there is sufficient reducing potential to drive the reaction to completion while preventing the excess hydride availability that leads to ring opening. The organic amine solvent, such as N,N-dimethylaniline, plays a dual role as both a reaction medium and a stabilizer for the reactive intermediates, further contributing to the high fidelity of the transformation.
How to Synthesize Sofosbuvir Intermediate Efficiently
The implementation of this synthesis route requires careful attention to the order of addition and temperature control to maximize the benefits of the Lewis acid catalysis. The process begins by suspending the starting material, Formula II, in a suitable organic amine solvent. The reaction mixture is then cooled to a range of 0°C to 10°C before the sequential or simultaneous addition of the Lewis acid and the borohydride salt. Maintaining this initial low temperature during reagent addition is vital to manage the exotherm and prevent localized hot spots that could trigger side reactions.
- Suspend Formula II compound in an organic amine solvent such as N,N-dimethylaniline or triethylamine.
- Add Lewis acid (e.g., Zinc Chloride) and borohydride (e.g., Potassium Borohydride) at 0-10°C.
- Warm the mixture to 15-20°C, stir for 40-60 hours, then purify via extraction and acid wash without column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from Red-Al based chemistry to this Lewis acid-borohydride protocol offers profound strategic advantages beyond mere chemical elegance. The elimination of column chromatography is perhaps the most significant economic driver, as chromatographic purification is notoriously resource-intensive, requiring large volumes of high-purity solvents and disposable silica media. By replacing this step with a simple liquid-liquid extraction and acid wash, the process drastically reduces solvent consumption and waste generation, leading to substantial cost savings in raw material procurement and waste disposal fees.
- Cost Reduction in Manufacturing: The replacement of expensive and hazardous Red-Al with commodity chemicals like zinc chloride and potassium borohydride significantly lowers the direct material cost of goods sold. Furthermore, the ability to operate at ambient temperatures (10-30°C) rather than cryogenic conditions (-40°C) reduces energy consumption for cooling, while the simplified workup reduces labor hours and equipment occupancy time, collectively driving down the manufacturing overhead.
- Enhanced Supply Chain Reliability: The reagents utilized in this novel process are widely available bulk chemicals with stable supply chains, unlike specialized reducing agents that may face sourcing volatility. The robustness of the reaction, evidenced by high yields and tolerance to slight variations in conditions, ensures consistent batch-to-batch quality. This reliability minimizes the risk of production delays caused by failed batches or complex rework, ensuring a steady flow of high-purity pharmaceutical intermediates to downstream customers.
- Scalability and Environmental Compliance: The avoidance of aluminum salt sludge and the reduction in solvent usage align perfectly with modern green chemistry principles and increasingly stringent environmental regulations. The process generates less hazardous waste, simplifying compliance reporting and lowering the environmental burden. Additionally, the simplicity of the unit operations—suspension, stirring, and extraction—makes the technology highly transferable from pilot scale to multi-ton commercial production without the need for complex specialized equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and validation of this synthesis method. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear picture of the process capabilities and limitations for potential licensees or manufacturing partners.
Q: What is the primary advantage of this new reduction method over Red-Al?
A: The new method avoids hazardous Red-Al and cryogenic conditions (-40°C), operating instead at mild temperatures (10-30°C) with significantly higher yields up to 90%.
Q: Does this process require column chromatography for purification?
A: No, the process eliminates the need for column chromatography. High purity (>99%) is achieved through simple extraction and acid washing steps.
Q: Which Lewis acids are most effective for this transformation?
A: Zinc chloride and aluminum trichloride are preferred, with Zinc chloride showing optimal results in controlling selectivity and minimizing by-products.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sofosbuvir Intermediate Supplier
The technological advancements detailed in Patent CN110669085B represent a significant leap forward in the manufacturing of antiviral nucleoside precursors. At NINGBO INNO PHARMCHEM, we pride ourselves on staying at the forefront of such process innovations to deliver superior value to our global partners. As a dedicated CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory breakthroughs are seamlessly translated into industrial reality. Our facilities are equipped with stringent purity specifications and rigorous QC labs to guarantee that every batch of Sofosbuvir intermediate meets the highest international standards.
We invite pharmaceutical companies and generic manufacturers to collaborate with us to leverage this efficient synthesis route for their supply chains. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both the quality and cost-efficiency of your antiviral drug production.
