Advanced Synthetic Route for D3-Salbutamol: Scalable Production for Food Safety Standards
Advanced Synthetic Route for D3-Salbutamol: Scalable Production for Food Safety Standards
The global demand for precise analytical standards in food safety monitoring has never been more critical, particularly for detecting beta-agonists like salbutamol in animal-derived products. Patent CN109678707B introduces a groundbreaking synthetic methodology for producing deuterium-labeled D3-salbutamol, a vital internal standard for gas chromatography-mass spectrometry (GC-MS) analysis. This innovative approach addresses the historical challenges of synthesizing isotopically labeled compounds with high specific activity and chemical purity. By utilizing ethyl salicylate as a robust starting material, the process ensures that the deuterium atoms are strategically incorporated at the benzylic positions, maximizing the stability and detection accuracy of the final standard. The resulting compound exhibits exceptional isotopic abundance, making it an indispensable tool for regulatory compliance and residue tracking in the agricultural and food industries.
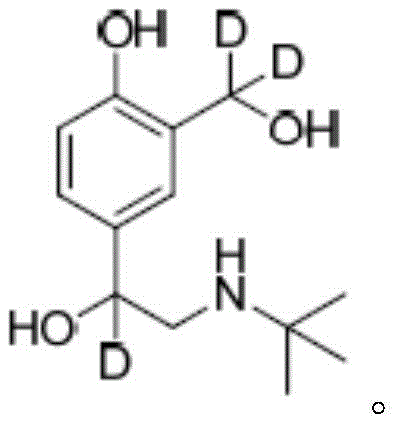
This technical breakthrough represents a significant leap forward for manufacturers of high-purity reference materials, offering a pathway that balances synthetic efficiency with rigorous quality control. The method circumvents the limitations of previous routes that relied on less suitable precursors like salicylaldehyde, which often failed to deliver the precise labeling pattern required for modern analytical instrumentation. By shifting the synthetic strategy to a four-step sequence involving acylation, bromination, amination, and selective reduction, the patent establishes a new benchmark for reliability in isotopic standard production. For procurement specialists and supply chain managers, this translates to a more secure source of critical reagents that are essential for maintaining the integrity of food safety testing protocols worldwide.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of salbutamol and its analogues has predominantly relied on salicylaldehyde as the primary building block, a route that presents significant drawbacks when attempting to introduce specific isotopic labels. The inherent reactivity of the aldehyde group often complicates the selective introduction of deuterium atoms at the desired benzylic positions without affecting other parts of the molecule. Furthermore, traditional pathways frequently suffer from poor regioselectivity during the formation of the side chain, leading to complex mixtures of impurities that are difficult and costly to separate. These inefficiencies not only drive up the cost of goods sold but also pose risks to the supply continuity of high-purity standards needed for sensitive analytical work. The reliance on multiple protection and deprotection steps in older methodologies further extends lead times and reduces overall process throughput.
The Novel Approach
In stark contrast, the novel approach detailed in the patent leverages ethyl salicylate to construct the molecular scaffold with superior control over functional group placement. This strategy initiates with a highly efficient Friedel-Crafts acylation, establishing the carbon backbone necessary for subsequent transformations while maintaining the integrity of the phenolic hydroxyl group. 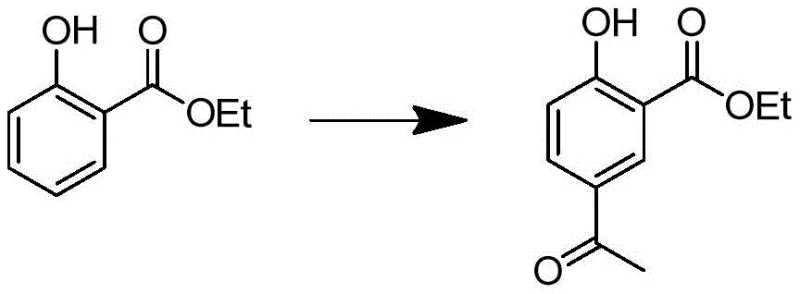 By deferring the introduction of the isotopic label to the final reduction step, the process minimizes the loss of expensive deuterated reagents and ensures that the label is incorporated exactly where it is needed for mass spectrometric differentiation. This streamlined logic eliminates unnecessary synthetic detours, allowing for a more direct and economically viable production cycle. The use of readily available industrial chemicals such as acetyl chloride and tert-butylamine further enhances the commercial attractiveness of this route, making it an ideal candidate for large-scale manufacturing of fine chemical intermediates.
By deferring the introduction of the isotopic label to the final reduction step, the process minimizes the loss of expensive deuterated reagents and ensures that the label is incorporated exactly where it is needed for mass spectrometric differentiation. This streamlined logic eliminates unnecessary synthetic detours, allowing for a more direct and economically viable production cycle. The use of readily available industrial chemicals such as acetyl chloride and tert-butylamine further enhances the commercial attractiveness of this route, making it an ideal candidate for large-scale manufacturing of fine chemical intermediates.
Mechanistic Insights into the Four-Step Deuteration Strategy
The core of this synthetic innovation lies in the precise orchestration of electrophilic aromatic substitution and nucleophilic displacement reactions to build the salbutamol skeleton. The initial acylation step utilizes a Lewis acid catalyst, typically anhydrous aluminum trichloride, to activate acetyl chloride, generating a highly reactive acylium ion that attacks the para-position of the ethyl salicylate ring. This regioselectivity is driven by the strong ortho/para-directing effect of the phenolic hydroxyl group, ensuring that the acetyl moiety is installed exclusively at the 5-position relative to the ester. Following this, the alpha-bromination of the resulting ketone is achieved using elemental bromine or copper bromide, a transformation that activates the side chain for nucleophilic attack by converting the methyl group into a reactive bromomethyl group. This activation is crucial for the subsequent amination step, where tert-butylamine displaces the bromide to form the characteristic amino-ketone intermediate.
The culmination of the synthesis is the stereoselective reduction of the ketone and ester functionalities using deuterated reducing agents, which is the defining step for creating the D3-labeled product. 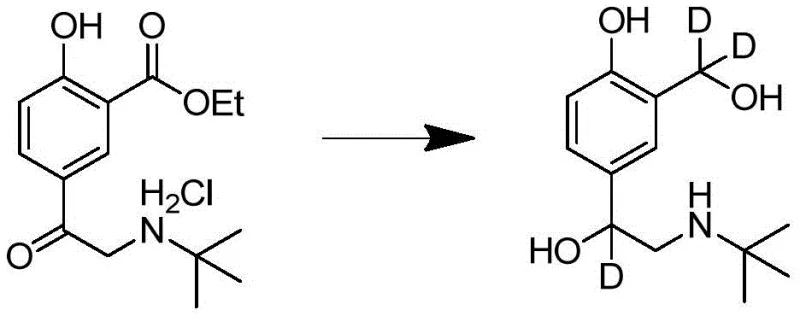 The patent highlights the use of sodium borodeuteride activated by boron trifluoride or lithium aluminum deuteride to effect this transformation. Mechanistically, the hydride (or rather deuteride) source delivers deuterium ions to the carbonyl carbons, simultaneously reducing the ketone to a secondary alcohol and the ester to a primary alcohol, both enriched with deuterium. This dual reduction capability is essential for achieving the tri-deuterated structure required for the internal standard. The careful control of reaction temperature and stoichiometry during this phase ensures that the isotopic abundance remains above 98%, preventing isotopic dilution that would compromise the analytical utility of the final compound.
The patent highlights the use of sodium borodeuteride activated by boron trifluoride or lithium aluminum deuteride to effect this transformation. Mechanistically, the hydride (or rather deuteride) source delivers deuterium ions to the carbonyl carbons, simultaneously reducing the ketone to a secondary alcohol and the ester to a primary alcohol, both enriched with deuterium. This dual reduction capability is essential for achieving the tri-deuterated structure required for the internal standard. The careful control of reaction temperature and stoichiometry during this phase ensures that the isotopic abundance remains above 98%, preventing isotopic dilution that would compromise the analytical utility of the final compound.
How to Synthesize D3-Salbutamol Efficiently
The synthesis of this high-value isotopic standard requires strict adherence to the optimized reaction conditions outlined in the patent to ensure maximum yield and purity. The process is designed to be operationally simple, avoiding the need for exotic equipment or hazardous high-pressure conditions, which facilitates its adoption in standard fine chemical manufacturing facilities. Detailed below is the structural framework for executing this synthesis, emphasizing the critical control points for each transformation. For laboratory technicians and process engineers, understanding the nuances of workup and purification at each stage is paramount to achieving the reported >98% chemical purity. The following guide serves as a high-level overview of the procedural flow, with specific quantitative parameters available in the full technical documentation.
- Perform Friedel-Crafts acylation of ethyl salicylate with acetyl chloride using anhydrous AlCl3 to form ethyl 2-hydroxy-5-acetylbenzoate.
- Conduct alpha-bromination of the acetyl group using liquid bromine or copper bromide to generate the bromoacetyl intermediate.
- React the bromo-intermediate with tert-butylamine followed by acidification to yield the amino-ketone hydrochloride salt.
- Execute the final reduction using deuterated reducing agents like NaBD4/BF3 or LiAlD4 to install deuterium atoms and form D3-Salbutamol.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this synthetic route offers compelling advantages that directly address the pain points of cost volatility and supply chain fragility often associated with isotopic standards. By utilizing commodity chemicals like ethyl salicylate and acetyl chloride as starting materials, the process decouples production from the fluctuating prices of specialized pharmaceutical intermediates. This foundational shift in raw material selection significantly reduces the cost of goods sold, allowing for more competitive pricing structures without sacrificing quality. Furthermore, the elimination of precious metal catalysts, such as palladium or platinum, which are common in hydrogenation reactions, removes a major cost driver and supply risk factor. This substitution with abundant Lewis acids like aluminum trichloride ensures that production can continue uninterrupted even during global shortages of rare earth or precious metals.
- Cost Reduction in Manufacturing: The economic benefits of this route are derived primarily from the high atom economy of the Friedel-Crafts acylation and the simplicity of the purification steps. Unlike complex biocatalytic routes that require expensive enzymes and controlled fermentation environments, this chemical synthesis relies on robust, well-understood unit operations that are easily scaled. The ability to purify intermediates through simple recrystallization rather than preparative HPLC drastically lowers solvent consumption and waste disposal costs. Additionally, the high yield reported in the patent examples suggests that raw material waste is minimized, further contributing to a leaner and more cost-effective manufacturing profile. These factors combine to create a substantial margin improvement potential for manufacturers adopting this technology.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the widespread availability of the requisite reagents, which are produced globally by numerous chemical suppliers. This diversity of supply sources mitigates the risk of single-point failures that can plague specialized synthetic routes dependent on niche custom synthesis. The robustness of the reaction conditions, which tolerate ambient temperatures and standard atmospheric pressure in several steps, also means that production is not limited to facilities with specialized high-pressure infrastructure. Consequently, this flexibility allows for distributed manufacturing strategies, ensuring that inventory levels can be maintained consistently to meet the steady demand from food safety laboratories. The simplified logistics of handling non-hazardous solids and common solvents further streamline the inbound and outbound supply chain operations.
- Scalability and Environmental Compliance: The environmental footprint of this process is favorably positioned due to the avoidance of heavy metal contaminants that often require complex remediation steps. The use of standard organic solvents like dichloromethane and ethanol allows for established recovery and recycling protocols, aligning with modern green chemistry initiatives. Scalability is inherently supported by the batch-wise nature of the reactions, which can be linearly increased from pilot plant scales to multi-ton annual production capacities without fundamental changes to the chemistry. This ease of scale-up ensures that as regulatory requirements for food testing tighten and demand for standards increases, supply can be ramped up rapidly to meet market needs. The generation of manageable waste streams, primarily inorganic salts and recoverable organics, simplifies compliance with increasingly stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of D3-Salbutamol synthesized via this patented method. These insights are derived directly from the experimental data and claims presented in the intellectual property documentation, providing a factual basis for decision-making. Understanding these details is crucial for quality assurance teams and procurement officers who need to validate the suitability of this material for their specific analytical workflows. The answers reflect the current state of the art as defined by the patent holder, ensuring accuracy and relevance.
Q: What is the isotopic purity of the D3-Salbutamol produced via this method?
A: According to patent CN109678707B, the synthesized D3-Salbutamol achieves an isotopic abundance of over 98%, ensuring high accuracy for mass spectrometry detection in food safety applications.
Q: Why is ethyl salicylate preferred over salicylaldehyde for this synthesis?
A: Ethyl salicylate provides a more stable scaffold for introducing the deuterium label at the benzylic position during the final reduction step, whereas salicylaldehyde routes often struggle with specific deuteration patterns required for this internal standard.
Q: Can this synthesis be scaled for commercial supply of reference standards?
A: Yes, the route utilizes common industrial reagents like acetyl chloride and aluminum trichloride, avoiding expensive transition metals, which facilitates straightforward scale-up from laboratory grams to multi-kilogram commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable D3-Salbutamol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity isotopic standards play in safeguarding public health and ensuring regulatory compliance in the food industry. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistent quality. We are committed to delivering D3-Salbutamol that meets stringent purity specifications, leveraging our rigorous QC labs to verify isotopic abundance and chemical identity before every shipment. Our facility is equipped to handle the specific handling requirements of deuterated reagents, guaranteeing that the integrity of the label is maintained throughout the manufacturing and packaging process.
We invite you to contact our technical procurement team to discuss how our advanced synthesis capabilities can support your analytical testing programs. By partnering with us, you gain access to a Customized Cost-Saving Analysis that demonstrates how our efficient route can lower your overall reagent costs. We encourage you to request specific COA data and route feasibility assessments to verify that our material aligns perfectly with your method validation requirements. Let us be your trusted partner in securing a stable and high-quality supply of essential food safety reference standards.
