Advanced Synthetic Route for D3-Salbutamol: Enabling High-Precision Food Safety Analysis
The escalating demand for ultra-sensitive detection methods in food safety monitoring has necessitated the development of highly stable isotope-labeled internal standards, particularly for beta-agonists like salbutamol. Patent CN109678707B discloses a robust and efficient synthetic methodology for producing deuterium-labeled D3-salbutamol, a critical reference material for quantifying salbutamol residues in animal-derived food products via gas chromatography-mass spectrometry (GC-MS). Unlike traditional approaches that struggle with regioselective deuteration, this invention utilizes ethyl salicylate as a cost-effective starting material, navigating through a logical four-step sequence comprising Friedel-Crafts acylation, alpha-bromination, tert-butyl amination, and finally, a specialized deuterated reduction. This strategic pathway not only ensures the precise placement of deuterium atoms at the benzylic position but also achieves exceptional chemical purity exceeding 98% and isotopic abundance greater than 98%, addressing the stringent accuracy requirements of modern analytical laboratories.
For R&D directors and procurement specialists in the fine chemical sector, the availability of such high-fidelity isotopic standards is paramount for validating analytical methods and ensuring regulatory compliance. The synthesis described in the patent represents a significant technological leap, moving away from less suitable precursors like salicylaldehyde which complicate the specific introduction of deuterium at the target methylene group. By establishing a reliable supply chain for D3-salbutamol, manufacturers can support the global food safety infrastructure, enabling labs to distinguish between endogenous compounds and contaminants with unparalleled precision. This report delves into the mechanistic nuances and commercial viability of this novel route, highlighting its potential as a benchmark for isotope-labeled intermediate production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of salbutamol and its analogues has often relied on salicylaldehyde as the primary building block, a route that presents significant challenges when attempting to synthesize specific deuterium-labeled isotopologues like D3-salbutamol. The inherent reactivity of the aldehyde group in salicylaldehyde makes it difficult to selectively introduce deuterium atoms exclusively at the benzylic position without causing isotopic scrambling or requiring prohibitively expensive fully deuterated starting materials. Furthermore, conventional pathways often involve multiple protection and deprotection steps to manage the reactivity of the phenolic hydroxyl group and the amine functionality, leading to lower overall yields and increased waste generation. These inefficiencies translate into higher production costs and longer lead times, creating bottlenecks for suppliers aiming to meet the growing demand for certified reference materials in the food safety and pharmaceutical industries.
The Novel Approach
The innovative route detailed in patent CN109678707B circumvents these historical limitations by employing ethyl salicylate as the foundational substrate, allowing for a more controlled and linear assembly of the molecular architecture. The process initiates with a regioselective Friedel-Crafts acylation to install the acetyl group para to the phenolic hydroxyl, followed by a precise alpha-bromination that activates the side chain for subsequent amination. 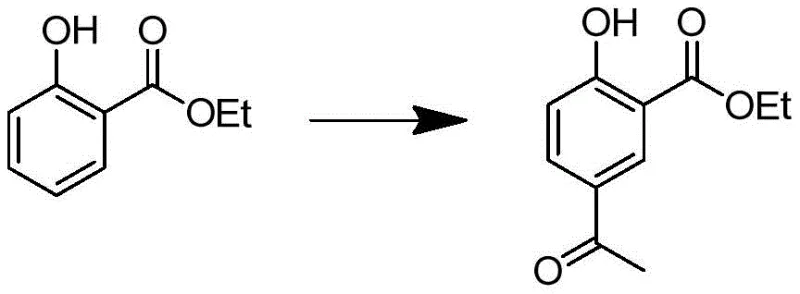 This sequence effectively builds the carbon skeleton before the critical isotopic labeling step, ensuring that the expensive deuterated reagents are utilized with maximum atom economy in the final reduction stage. By deferring the introduction of deuterium to the last step, the method minimizes isotopic dilution and simplifies the purification process, resulting in a product with superior isotopic enrichment and chemical homogeneity compared to earlier generation synthesis strategies.
This sequence effectively builds the carbon skeleton before the critical isotopic labeling step, ensuring that the expensive deuterated reagents are utilized with maximum atom economy in the final reduction stage. By deferring the introduction of deuterium to the last step, the method minimizes isotopic dilution and simplifies the purification process, resulting in a product with superior isotopic enrichment and chemical homogeneity compared to earlier generation synthesis strategies.
Mechanistic Insights into Deuterated Reduction and Functional Group Transformation
The cornerstone of this synthetic strategy lies in the final reduction step, where the simultaneous reduction of the ketone and ester functionalities is achieved using deuterated reducing agents such as sodium borodeuteride (NaBD4) activated by boron trifluoride (BF3) or lithium aluminum deuteride (LiAlD4). In the presence of a Lewis acid like BF3, the carbonyl oxygen of the ketone is coordinated, significantly increasing the electrophilicity of the carbonyl carbon and facilitating the nucleophilic attack by the deuteride ion. This mechanism ensures that the hydride (deuteride) transfer occurs specifically at the benzylic position, thereby installing two of the three deuterium atoms, while the third deuterium is incorporated during the reduction of the adjacent hydroxymethyl precursor or through exchange mechanisms controlled by the reaction conditions. 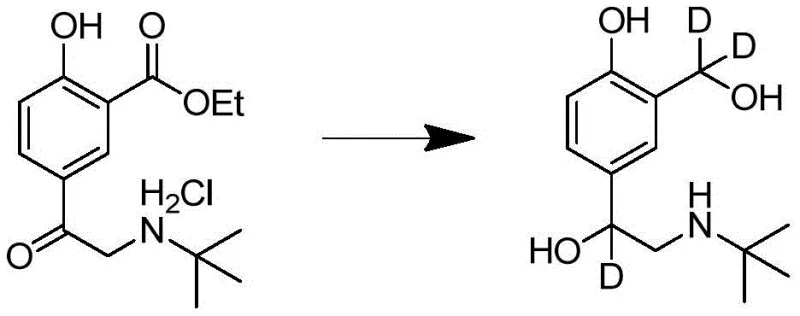 The careful control of temperature and stoichiometry during this phase is critical to prevent over-reduction or side reactions that could compromise the isotopic purity, demonstrating a sophisticated understanding of physical organic chemistry applied to industrial synthesis.
The careful control of temperature and stoichiometry during this phase is critical to prevent over-reduction or side reactions that could compromise the isotopic purity, demonstrating a sophisticated understanding of physical organic chemistry applied to industrial synthesis.
Furthermore, the preceding amination step exhibits excellent chemoselectivity, where the primary amine (tert-butylamine) displaces the alpha-bromine atom via an SN2 mechanism without affecting the ester or phenolic groups. The use of mild solvents like tetrahydrofuran or acetonitrile at room temperature facilitates this substitution while minimizing the formation of elimination byproducts. Following the amination, the conversion to the hydrochloride salt serves a dual purpose: it stabilizes the amine against oxidation and facilitates purification through crystallization, effectively removing unreacted amine and inorganic salts. This attention to intermediate stability and purification efficiency throughout the mechanistic pathway underscores the robustness of the process, making it highly suitable for the production of high-purity reference standards required for quantitative mass spectrometry applications.
How to Synthesize D3-Salbutamol Efficiently
The synthesis of D3-salbutamol via this patented route involves a carefully orchestrated sequence of reactions that balance reactivity with selectivity to ensure high yields and isotopic fidelity. The process begins with the acylation of ethyl salicylate, followed by bromination and amination to construct the core scaffold, culminating in the deuterated reduction that defines the product's identity. Each step has been optimized for scalability, utilizing common industrial solvents and reagents that are readily available in the global chemical market. For detailed operational parameters, including specific molar ratios, temperature profiles, and workup procedures, operators should refer to the standardized synthesis guidelines derived from the patent examples, which demonstrate reproducibility across different batch sizes.
- Perform Friedel-Crafts acylation of ethyl salicylate with acetyl chloride using anhydrous AlCl3 to form ethyl 2-hydroxy-5-acetylbenzoate.
- Execute alpha-bromination of the acetyl group using liquid bromine or copper bromide to generate the bromo-ketone intermediate.
- Conduct nucleophilic substitution with tert-butylamine followed by acidification to yield the amino-ketone hydrochloride salt.
- Reduce the ketone and ester functionalities using a deuterated reducing agent like NaBD4/BF3 or LiAlD4 to introduce deuterium labels.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain and procurement perspective, this synthetic route offers compelling advantages by leveraging commodity chemicals and minimizing the reliance on exotic or custom-synthesized starting materials. The use of ethyl salicylate, a widely produced fine chemical, as the starting material significantly reduces raw material costs and mitigates supply risk compared to routes requiring specialized deuterated aldehydes. Moreover, the operational simplicity of the reaction steps, many of which proceed at ambient temperature or mild heating, lowers energy consumption and reduces the complexity of reactor requirements, thereby enhancing the overall cost-efficiency of the manufacturing process. These factors collectively contribute to a more resilient supply chain capable of responding rapidly to fluctuations in demand for food safety testing reagents.
- Cost Reduction in Manufacturing: The strategic design of the synthesis pathway eliminates the need for expensive fully deuterated starting materials by introducing the isotopic label in the final step, maximizing the utility of costly deuterated reagents. This late-stage functionalization approach drastically reduces the material cost per gram of the final product, as the bulk of the molecular weight is built using inexpensive, non-labeled commodities. Additionally, the purification methods described, such as recrystallization and simple aqueous workups, avoid the need for preparative HPLC or extensive column chromatography on a large scale, further driving down processing costs and solvent waste disposal expenses.
- Enhanced Supply Chain Reliability: By relying on a four-step linear synthesis with high-yielding transformations, the process minimizes the accumulation of impurities that often plague longer synthetic routes, ensuring consistent batch-to-batch quality. The robustness of the chemistry allows for flexible manufacturing schedules, as the intermediates are stable and can be stocked if necessary, providing a buffer against sudden spikes in demand for D3-salbutamol standards. This reliability is crucial for maintaining the continuity of supply for regulatory bodies and testing laboratories that depend on these standards for daily operations.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability potential due to the use of standard unit operations like filtration, distillation, and crystallization, which are easily transferred from laboratory to pilot and commercial scales. The avoidance of heavy metal catalysts and the use of relatively benign solvents align with green chemistry principles, simplifying environmental compliance and waste treatment protocols. This environmental compatibility not only reduces regulatory burdens but also enhances the corporate sustainability profile of the manufacturer, a key consideration for modern procurement strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of D3-salbutamol synthesized via this novel method. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on the capabilities and limitations of the technology. Understanding these aspects is essential for stakeholders evaluating the suitability of this material for their specific analytical or research needs.
Q: Why is ethyl salicylate preferred over salicylaldehyde for D3-salbutamol synthesis?
A: Ethyl salicylate allows for the construction of the carbon skeleton prior to isotopic labeling. Using salicylaldehyde makes it difficult to specifically introduce deuterium atoms at the benzylic position without affecting other parts of the molecule, whereas the ketone reduction strategy ensures precise D3 labeling.
Q: What is the achieved isotopic abundance and chemical purity?
A: The patented method reports achieving a chemical purity of over 98% and an isotopic abundance exceeding 98%, which meets the rigorous requirements for mass spectrometry internal standards in food safety analysis.
Q: Can this process be scaled for commercial production?
A: Yes, the process utilizes common industrial solvents like dichloromethane and ethanol, and avoids extremely harsh conditions. The purification steps rely on recrystallization and standard extraction, which are highly scalable compared to complex chromatographic separations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable D3-Salbutamol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity isotopic standards play in ensuring food safety and public health, and we are committed to delivering D3-salbutamol that meets the highest industry benchmarks. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to verify both chemical purity and isotopic abundance, guaranteeing that every batch performs reliably in your GC-MS assays.
We invite you to collaborate with us to optimize your supply chain for reference standards and benefit from our technical expertise in isotope-labeled compound synthesis. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term strategic goals in the food safety and analytical chemistry sectors.
