Advanced Manufacturing of Benzbromarone Intermediates via Optimized Friedel-Crafts Acylation
Introduction to Next-Generation Benzbromarone Manufacturing
The global demand for effective gout treatments continues to rise, placing immense pressure on the supply chains of key active pharmaceutical ingredients (APIs) like Benzbromarone. As regulatory standards tighten regarding impurity profiles and process safety, legacy synthetic routes are increasingly becoming liabilities for pharmaceutical manufacturers. Patent CN103242272A introduces a transformative approach to Benzbromarone synthesis that fundamentally restructures the production timeline to prioritize safety and purity. By shifting the introduction of the dibromo-substituted aromatic ring to the very beginning of the synthesis rather than the end, this methodology circumvents the formation of critical impurities that have historically plagued quality control departments. This technical insight report analyzes the strategic value of this patent for R&D directors seeking robust pathways and procurement leaders aiming for cost-effective, compliant sourcing strategies in the competitive anti-gout medication market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Benzbromarone has relied on pathways that introduce significant safety hazards and purity challenges, particularly those attempting late-stage functionalization. For instance, prior art such as the route described in WO2012048058 involves a perilous sequence utilizing chloroacetone, which decomposes to release toxic phosgene upon heating, followed by reductions using hydrazine and acylations in carbon disulfide. These processes not only expose workers to carcinogenic and corrosive substances but also suffer from poor atom economy and low yields, with some steps reporting efficiencies as low as 35%. Furthermore, performing bromination as the final step inevitably leads to the generation of mono-brominated and tri-brominated byproducts, necessitating expensive and yield-loss-inducing purification cycles to meet pharmacopoeia standards.
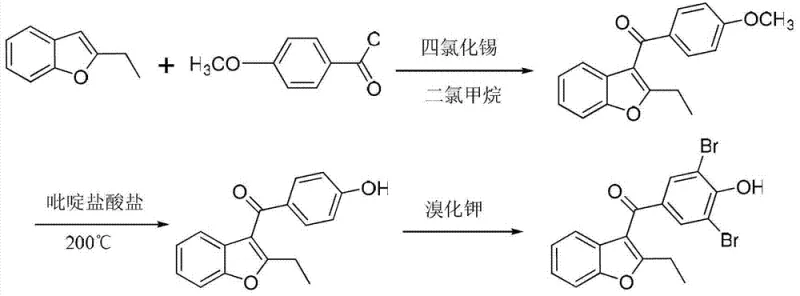
The Novel Approach
In stark contrast, the methodology disclosed in CN103242272A adopts a "protection-first" strategy that stabilizes the reactive phenolic hydroxyl group before the critical carbon-carbon bond-forming event. By starting with 3,5-dibromo-4-hydroxybenzoic acid, the bromine substitution pattern is pre-installed, effectively eliminating the risk of forming regio-isomeric impurities during the final stages. The process utilizes a mild acetylation to protect the phenol, followed by conversion to an acid chloride and a controlled Friedel-Crafts acylation with 2-ethylbenzofuran. This sequence avoids the use of elemental bromine entirely in the final steps and operates at significantly lower temperatures compared to the 200°C required in other alternative routes, thereby enhancing the thermal safety profile and simplifying the engineering controls required for reactor design.
Mechanistic Insights into Protected Friedel-Crafts Acylation
The core chemical innovation lies in the strategic use of the acetyl group as a temporary protecting mask for the phenolic oxygen. In traditional electrophilic aromatic substitutions, a free phenolic hydroxyl group can lead to unwanted O-acylation or polymerization side reactions, complicating the reaction mixture. By converting the starting material into 3,5-dibromo-4-acetoxybenzoyl chloride, the electron density of the aromatic ring is modulated, and the oxygen is rendered inert to the Lewis acid catalyst. This ensures that when the acid chloride reacts with 2-ethylbenzofuran in the presence of catalysts like aluminum trichloride or tin tetrachloride, the acylation occurs exclusively at the desired position on the benzofuran ring. The subsequent hydrolysis step is remarkably mild, utilizing aqueous sodium hydroxide at 40-45°C to cleave the ester bond, regenerating the free phenol without degrading the sensitive ketone linkage or the bromine substituents.
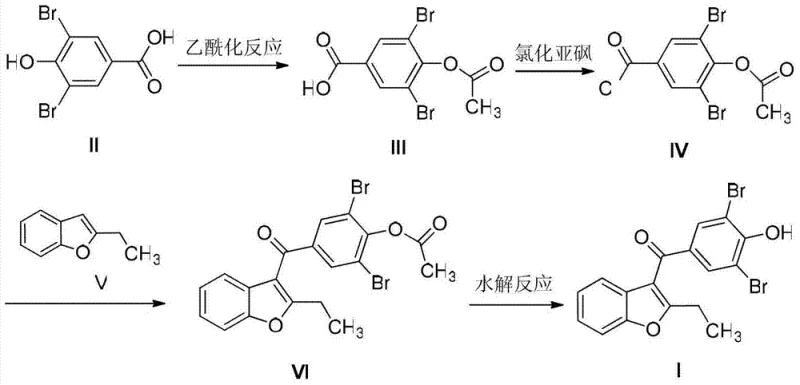
From an impurity control perspective, this mechanism offers a decisive advantage by decoupling the halogenation step from the final coupling step. In legacy processes, the electrophilic bromination of the final ketone is difficult to stop precisely at the di-bromo stage, often resulting in over-bromination or under-bromination. By utilizing a pre-brominated benzoic acid derivative, the stoichiometry of the halogen atoms is fixed before the molecule becomes complex. This "building block" approach ensures that the final API contains negligible amounts of the specific mono-bromo and tri-bromo impurities cited in the Chinese Pharmacopoeia, drastically reducing the burden on analytical teams and minimizing the batch rejection rates associated with out-of-specification impurity profiles.
How to Synthesize Benzbromarone Efficiently
The implementation of this synthesis route requires precise control over reaction stoichiometry and temperature, particularly during the exothermic Friedel-Crafts acylation step. The process is divided into four distinct operational phases: protection, activation, coupling, and deprotection. Each phase utilizes common industrial solvents such as dichloromethane and ethanol, facilitating easy solvent recovery and recycling. The following guide outlines the standardized operational parameters derived from the patent examples, providing a roadmap for process engineers to translate this laboratory-scale success into commercial production while maintaining the high purity and yield characteristics demonstrated in the intellectual property.
- Acetylate 3,5-dibromo-4-hydroxybenzoic acid using acetic anhydride to protect the phenolic hydroxyl group.
- Convert the protected acid to its corresponding acid chloride using thionyl chloride.
- Perform Friedel-Crafts acylation with 2-ethylbenzofuran using a Lewis acid catalyst like aluminum trichloride.
- Hydrolyze the acetyl protecting group under mild alkaline conditions to yield the final Benzbromarone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route translates directly into reduced operational risk and enhanced cost predictability. By eliminating the need for handling elemental bromine and phosgene in the final manufacturing stages, facilities can significantly lower their expenditure on specialized safety equipment, hazardous waste disposal, and regulatory compliance monitoring. The avoidance of extreme high-temperature reactions also reduces energy consumption and extends the lifespan of reactor vessels, contributing to a lower total cost of ownership for the manufacturing asset. Furthermore, the high selectivity of the reaction minimizes the loss of valuable starting materials to byproduct formation, ensuring that raw material spend is efficiently converted into saleable product.
- Cost Reduction in Manufacturing: The elimination of late-stage bromination removes the need for complex purification processes required to separate difficult isomeric impurities, which traditionally consume significant solvent and chromatography resources. Additionally, the use of readily available Lewis acid catalysts and common solvents avoids the premium pricing associated with exotic reagents, leading to substantial savings in the bill of materials without compromising product quality.
- Enhanced Supply Chain Reliability: Because the process avoids the use of highly regulated toxic gases like phosgene, the supply chain is less vulnerable to disruptions caused by strict transportation regulations or supplier shutdowns due to safety incidents. The starting materials, such as 3,5-dibromo-4-hydroxybenzoic acid, are stable solids that can be stocked in bulk, providing a buffer against market volatility and ensuring continuous production schedules even during periods of raw material scarcity.
- Scalability and Environmental Compliance: The mild reaction conditions, operating primarily between 0°C and 45°C, make this process inherently easier to scale from pilot plants to multi-ton reactors without the need for specialized high-pressure or high-temperature infrastructure. The reduction in hazardous waste generation, specifically the absence of bromine-containing mother liquors from the final step, simplifies wastewater treatment protocols and aligns the manufacturing process with increasingly stringent global environmental, health, and safety (EHS) standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding the implementation of this optimized Benzbromarone synthesis pathway. These insights are derived directly from the comparative data and experimental results presented in the patent documentation, offering clarity on how this method resolves historical bottlenecks in production. Understanding these nuances is critical for technical teams evaluating the feasibility of technology transfer and for commercial teams negotiating supply agreements based on process robustness.
Q: How does this synthesis route improve impurity control compared to traditional methods?
A: Traditional methods often perform bromination as the final step, leading to difficult-to-remove mono- and tri-bromo impurities. This patented route utilizes 3,5-dibromo-4-hydroxybenzoic acid as a starting material, locking the bromine substitution pattern early in the synthesis and eliminating the generation of these specific regio-isomeric impurities.
Q: What are the safety advantages of this process for industrial scale-up?
A: This method eliminates the use of highly toxic phosgene and corrosive bromine in the final steps. Additionally, it avoids extreme reaction temperatures (such as 200°C required in other routes), operating instead at mild conditions between 0°C and 45°C, which significantly reduces operational risks and equipment stress.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is designed for industrialization. It uses common solvents like dichloromethane and ethanol, avoids complex purification steps for isomeric impurities, and demonstrates high yields in pilot examples, making it highly scalable for metric-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzbromarone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to safer, more efficient synthetic routes is essential for maintaining competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN103242272A are fully realized in practical, GMP-compliant manufacturing environments. We maintain stringent purity specifications and operate rigorous QC labs capable of detecting trace impurities at ppm levels, guaranteeing that every batch of Benzbromarone meets the highest international regulatory standards for API intermediates.
We invite you to collaborate with us to leverage this advanced technology for your supply chain needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing capabilities can enhance your product quality while reducing your overall procurement costs.
