Optimizing Benzbromarone Production: A Technical Analysis of the Novel THP-Protection Route
Optimizing Benzbromarone Production: A Technical Analysis of the Novel THP-Protection Route
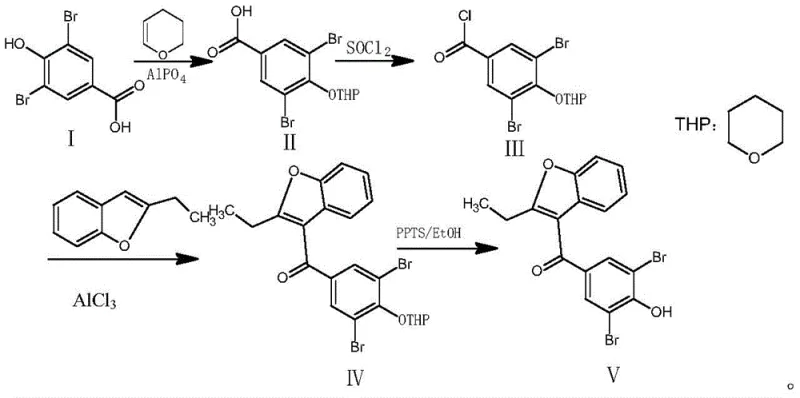
The pharmaceutical industry's demand for high-purity uricosuric agents continues to drive innovation in process chemistry, particularly for established drugs like Benzbromarone. Patent CN111533718B introduces a significant methodological advancement in the synthesis of this potent gout medication, addressing long-standing inefficiencies in traditional manufacturing routes. By shifting from conventional acetyl protection to a robust tetrahydropyranyl (THP) protection strategy, this technology offers a streamlined pathway that enhances both yield and product integrity. For R&D directors and procurement specialists evaluating supply chain resilience, understanding the mechanistic nuances of this patent is critical for securing a reliable benzbromarone intermediate supplier. The disclosed method not only mitigates the formation of stubborn impurities but also optimizes the use of common reagents, presenting a compelling case for cost reduction in API manufacturing without compromising on quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Benzbromarone has relied heavily on acetyl protection strategies to mask the phenolic hydroxyl group of the starting material, 3,5-dibromo-4-hydroxybenzoic acid. While conceptually straightforward, this approach suffers from inherent chemical liabilities that plague large-scale production. A primary defect is the lack of selectivity during the protection phase, where the acetylating agent inevitably reacts with the carboxylic acid moiety to form mixed anhydrides or esters, creating byproducts that are difficult to separate. Furthermore, the resulting acetyl ester is chemically fragile; during the subsequent acyl chlorination and Friedel-Crafts acylation steps, the ester linkage is prone to premature hydrolysis or chlorination at the hydroxyl position. These side reactions generate a complex impurity profile that necessitates rigorous and costly purification protocols, ultimately dragging down the overall yield and extending the production cycle time significantly.
The Novel Approach
In stark contrast, the methodology outlined in CN111533718B employs dihydropyran (DHP) to install a tetrahydropyranyl (THP) ether protecting group, a strategic modification that fundamentally alters the reaction landscape. This novel approach leverages the steric bulk and chemical stability of the THP group to shield the phenolic oxygen effectively throughout the harsh electrophilic substitution conditions. By utilizing aluminum phosphate as a mild catalyst for the initial protection step, the process achieves exceptional conversion rates while avoiding the over-reaction issues seen with acetyl chloride. The stability of the THP ether ensures that the subsequent conversion to the acid chloride and the aluminum trichloride-mediated coupling proceed with minimal interference, preserving the structural integrity of the intermediate. This robustness translates directly into a cleaner reaction profile, allowing for simpler workup procedures and higher isolated yields of the final active pharmaceutical ingredient.
Mechanistic Insights into THP Protection and Friedel-Crafts Acylation
The core innovation of this synthesis lies in the precise orchestration of protective group chemistry and Lewis acid catalysis. The initial protection step utilizes dihydropyran in the presence of aluminum phosphate, which acts as a solid acid catalyst to facilitate the addition of the alcohol to the enol ether double bond. This mechanism proceeds through an oxocarbenium ion intermediate, which is selectively attacked by the phenolic oxygen of the dibromo-benzoic acid. Unlike liquid acids that might promote polymerization of the dihydropyran, the heterogeneous nature of aluminum phosphate allows for easy removal via filtration, simplifying the downstream processing. Following protection, the conversion to the acid chloride is mediated by thionyl chloride with a catalytic amount of dimethylformamide (DMF). The DMF activates the thionyl chloride to form the highly reactive Vilsmeier-Haack reagent, ensuring rapid and complete chlorination of the carboxyl group at mild temperatures between 30°C and 35°C, thereby preventing thermal degradation of the sensitive THP group.
The subsequent Friedel-Crafts acylation represents the most critical bond-forming event in the sequence, requiring meticulous control to ensure regioselectivity and prevent polyacylation. The reaction couples the activated acid chloride with 2-ethylbenzofuran using aluminum trichloride as the Lewis acid promoter. Operating at cryogenic temperatures between -5°C and 0°C is paramount; this thermal constraint suppresses the formation of polymeric byproducts from the electron-rich benzofuran ring and directs the acylation specifically to the 3-position. Finally, the deprotection step utilizes pyridine p-toluenesulfonate (PPTS) in ethanol. PPTS is a mild organic acid that selectively cleaves the acid-labile THP ether without affecting the ketone linkage or the bromine substituents. This gentle deprotection mechanism avoids the harsh acidic or basic conditions typically required for ester hydrolysis, resulting in a final product with a purity exceeding 98% as confirmed by HPLC analysis, demonstrating superior impurity control compared to legacy methods.
How to Synthesize Benzbromarone Efficiently
Implementing this optimized synthetic route requires adherence to specific operational parameters to maximize the benefits of the THP protection strategy. The process begins with the reflux of 3,5-dibromo-4-hydroxybenzoic acid in excess dihydropyran with aluminum phosphate, followed by a careful acyl chlorination using thionyl chloride and DIPEA as an acid scavenger. The critical coupling step demands strict temperature maintenance below 5°C during the addition of aluminum trichloride to manage the exotherm and ensure selectivity. The final deprotection is achieved under mild reflux in ethanol with PPTS, followed by crystallization from dichloroethane to secure high-purity crystals. For detailed standard operating procedures and safety guidelines regarding reagent handling and waste disposal, please refer to the technical execution guide below.
- Protect the hydroxyl group of 3,5-dibromo-4-hydroxybenzoic acid using dihydropyran and aluminum phosphate catalyst under reflux to form the THP ether intermediate.
- Convert the protected acid to the corresponding acid chloride using thionyl chloride and DMF catalyst in dichloromethane.
- Perform Friedel-Crafts acylation with 2-ethylbenzofuran using aluminum trichloride at low temperature (-5 to 0°C) to couple the aromatic rings.
- Remove the THP protecting group using pyridine p-toluenesulfonate (PPTS) in ethanol under reflux to yield crude Benzbromarone.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this THP-based synthesis route offers substantial strategic advantages for procurement managers and supply chain heads focused on continuity and margin improvement. The elimination of unstable acetyl intermediates reduces the risk of batch failures due to hydrolysis, thereby enhancing the predictability of production schedules. Furthermore, the simplified purification requirements—stemming from the reduced generation of chlorinated and acetylated byproducts—translate into significant operational efficiencies. By minimizing the number of recrystallization steps and chromatographic separations needed to meet pharmacopeial standards, manufacturers can drastically reduce solvent consumption and processing time. This streamlining of the workflow not only lowers the direct cost of goods sold but also alleviates pressure on waste management infrastructure, aligning production capabilities with increasingly stringent environmental regulations.
- Cost Reduction in Manufacturing: The transition to a THP protection strategy eliminates the need for expensive and hazardous reagents often required to correct side reactions in acetyl-based routes. By preventing the formation of carboxyl-acetylated byproducts, the process avoids the yield losses associated with separating these difficult impurities. Additionally, the use of aluminum phosphate as a reusable heterogeneous catalyst in the first step reduces the reliance on stoichiometric amounts of corrosive liquid acids, lowering raw material costs and neutralization expenses. The overall increase in yield across the four-step sequence means that less starting material is required to produce the same amount of final API, directly improving the cost-efficiency ratio for large-scale commercial batches.
- Enhanced Supply Chain Reliability: The robustness of the THP intermediate ensures greater stability during storage and transport between synthesis stages, reducing the risk of degradation that can disrupt just-in-time manufacturing models. The reagents employed, such as dihydropyran and thionyl chloride, are commodity chemicals with stable global supply chains, mitigating the risk of sourcing bottlenecks. Moreover, the high purity of the crude product obtained after deprotection reduces the dependency on specialized third-party purification services, allowing for more vertical integration and control over the supply timeline. This reliability is crucial for maintaining consistent inventory levels of this critical gout medication in the face of fluctuating market demand.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard unit operations like reflux, filtration, and distillation that are easily transferable from pilot plant to multi-ton production scales. The avoidance of heavy metal catalysts and the minimization of halogenated waste streams simplify the effluent treatment process, ensuring compliance with modern environmental discharge standards. The mild conditions of the final deprotection step also reduce energy consumption compared to high-temperature hydrolysis methods, contributing to a lower carbon footprint for the manufacturing facility. These factors collectively make the technology highly attractive for contract development and manufacturing organizations (CDMOs) seeking to expand their portfolio of sustainable API intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. They are derived from the specific experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical implications of adopting this technology. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing frameworks.
Q: Why is THP protection superior to acetyl protection for Benzbromarone synthesis?
A: Traditional acetyl protection often leads to unwanted acetylation of the carboxyl group and forms unstable esters that hydrolyze easily during subsequent steps. The THP (tetrahydropyranyl) group offers superior stability during the harsh Friedel-Crafts acylation conditions and prevents side reactions like hydroxyl chlorination, resulting in higher overall purity.
Q: What are the critical reaction conditions for the Friedel-Crafts step in this process?
A: The coupling of the acid chloride with 2-ethylbenzofuran requires strict temperature control between -5°C and 0°C using aluminum trichloride as the Lewis acid catalyst. Maintaining this low temperature is essential to minimize polymerization of the benzofuran ring and ensure high regioselectivity.
Q: How does this process impact the environmental footprint of production?
A: By eliminating the need for harsh hydrolysis steps required to remove acetyl groups and reducing the formation of chlorinated byproducts, this route simplifies waste treatment. The use of recyclable solvents like dichloromethane and ethanol, combined with high yields, significantly reduces the E-factor of the manufacturing process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzbromarone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of process robustness in the supply of high-value pharmaceutical intermediates like Benzbromarone. Our technical team has extensively analyzed the THP-protection route described in CN111533718B and possesses the expertise to implement this advanced chemistry at scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant environment. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch meets the exacting standards required for downstream API synthesis.
We invite procurement leaders and R&D directors to collaborate with us to leverage this superior synthetic technology for your supply chain. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us directly to obtain specific COA data and route feasibility assessments, ensuring that your transition to this optimized Benzbromarone supply source is seamless, compliant, and economically advantageous.
