Advanced Asymmetric Reduction Strategy for High-Purity Formoterol Key Intermediates
Introduction to High-Efficiency Formoterol Intermediate Synthesis
The pharmaceutical industry continuously seeks robust methodologies for synthesizing critical asthma and COPD medications, specifically targeting the efficient production of beta-2 sympathomimetics. Patent CN110627673B, published in April 2023, introduces a groundbreaking preparation method for the key formoterol intermediate, (R)-N-(2-(benzyloxy)-5-(2-bromo-1-hydroxyethyl)phenyl)formamide, designated as Formula I. This compound serves as a pivotal building block in the manufacture of Formoterol fumarate, a widely prescribed drug for relaxing tracheal smooth muscle. The structural integrity and stereochemical purity of this intermediate are paramount for the efficacy and safety of the final API. 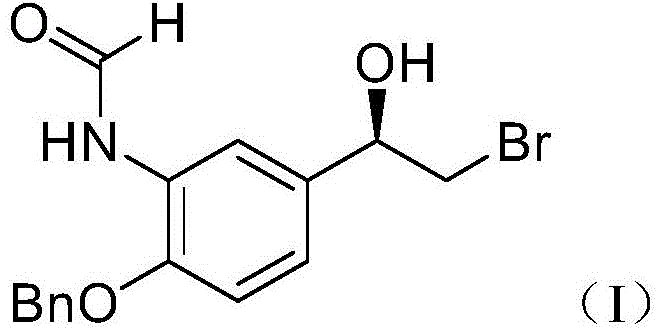 The disclosed technology addresses long-standing challenges in chiral synthesis by employing a highly selective asymmetric reduction strategy. By leveraging a specific indeno-oxazole borane catalyst, the process ensures exceptional enantioselectivity while maintaining operational simplicity. For R&D directors and procurement specialists, this patent represents a significant shift away from wasteful resolution techniques toward a more sustainable, high-yield manufacturing paradigm that aligns with modern green chemistry principles and stringent regulatory requirements for impurity control.
The disclosed technology addresses long-standing challenges in chiral synthesis by employing a highly selective asymmetric reduction strategy. By leveraging a specific indeno-oxazole borane catalyst, the process ensures exceptional enantioselectivity while maintaining operational simplicity. For R&D directors and procurement specialists, this patent represents a significant shift away from wasteful resolution techniques toward a more sustainable, high-yield manufacturing paradigm that aligns with modern green chemistry principles and stringent regulatory requirements for impurity control.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral alcohol intermediates for formoterol has been plagued by inefficiencies that hinder large-scale commercial viability. Traditional approaches often relied on the resolution of racemates using chiral columns, a technique that is not only prohibitively expensive due to the cost of chiral stationary phases but also inherently wasteful, generating up to 50% of the unwanted enantiomer which must be discarded. Alternative kinetic resolution methods, while slightly reducing costs, still suffer from complicated production processes and poor atom economy, failing to meet the sustainability goals of modern pharmaceutical manufacturing. Furthermore, existing chemical synthesis routes reported in prior literature often struggle to achieve the necessary chiral purity, typically capping at around 97% to 99% even after extensive purification. These limitations result in higher production costs, extended lead times, and potential supply chain bottlenecks, making them unsuitable for the high-volume demands of the global respiratory drug market. The inability to consistently produce high-purity intermediates without complex downstream processing remains a critical pain point for generic API manufacturers seeking cost-effective solutions.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in CN110627673B utilizes a direct asymmetric reduction pathway that fundamentally transforms the production landscape. The process begins with the conversion of 1-(4-(benzyloxy)-3-nitrophenyl)-2-bromoethanone (Formula II) into the chiral alcohol intermediate (Formula III) using a specialized borane catalyst system. 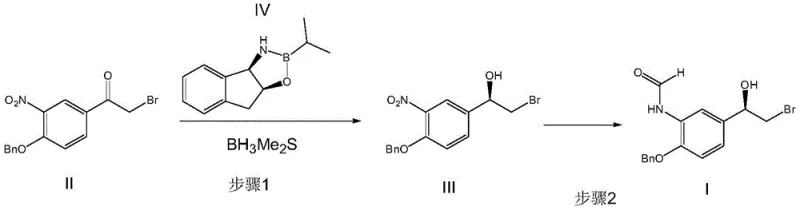 This is followed by a streamlined one-pot sequence involving nitro reduction and formylation to yield the target Formula I. This methodology eliminates the need for chiral separation entirely, thereby maximizing atom economy and ensuring that nearly all starting material is converted into the desired therapeutic enantiomer. The integration of nitro reduction and formylation into a single pot significantly reduces solvent usage, energy consumption, and processing time. For a reliable pharmaceutical intermediate supplier, adopting this route means transitioning from a low-yield, high-waste operation to a high-efficiency process capable of delivering superior quality at a fraction of the traditional operational overhead, directly addressing the need for cost reduction in API manufacturing.
This is followed by a streamlined one-pot sequence involving nitro reduction and formylation to yield the target Formula I. This methodology eliminates the need for chiral separation entirely, thereby maximizing atom economy and ensuring that nearly all starting material is converted into the desired therapeutic enantiomer. The integration of nitro reduction and formylation into a single pot significantly reduces solvent usage, energy consumption, and processing time. For a reliable pharmaceutical intermediate supplier, adopting this route means transitioning from a low-yield, high-waste operation to a high-efficiency process capable of delivering superior quality at a fraction of the traditional operational overhead, directly addressing the need for cost reduction in API manufacturing.
Mechanistic Insights into Borane-Catalyzed Asymmetric Reduction
The core innovation of this synthesis lies in the precise stereochemical control exerted by the catalyst, (3aS-cis)-(-)-3,3a,8,8a-tetrahydro-2H-indeno[1,2-d]oxazole-2-isopropylborane (Formula IV). This chiral borane species facilitates the transfer of hydride from borane dimethyl sulfide to the carbonyl group of the ketone substrate with exceptional facial selectivity. The rigid bicyclic structure of the catalyst creates a well-defined chiral environment that effectively shields one face of the carbonyl group, forcing the hydride attack to occur exclusively from the opposite side. This mechanistic precision is what allows the process to achieve chiral purities exceeding 99.5% for the intermediate alcohol (III), a metric that is critical for preventing the formation of diastereomeric impurities in subsequent steps. The reaction is conducted under mild conditions, typically between -25 °C and 30 °C, which minimizes thermal degradation and side reactions such as debromination or over-reduction. Understanding this catalytic cycle is essential for R&D teams aiming to replicate or optimize the process, as the ratio of catalyst to substrate and the rate of borane addition are key variables influencing the enantiomeric excess.
Furthermore, the subsequent transformation of the nitro group to the formamide functionality is engineered to preserve this high optical purity. The one-pot nitro reduction utilizes heterogeneous catalysts like platinum carbon or platinum dioxide under controlled hydrogen pressure (0.1-2 MPa). By avoiding the isolation of the unstable amino intermediate, the process mitigates the risk of racemization or oxidation that often occurs during work-up procedures in multi-step syntheses. The immediate in-situ formylation using formic acid and acetic anhydride locks the amine into the stable formamide structure. This seamless transition from reduction to protection ensures that the final product maintains a chiral purity of greater than 99.8%, meeting the rigorous specifications required for clinical-grade materials. The ability to control impurity profiles through mechanistic design rather than relying solely on purification underscores the sophistication of this route, offering a robust solution for the commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize (R)-N-(2-(benzyloxy)-5-(2-bromo-1-hydroxyethyl)phenyl)formamide Efficiently
Implementing this synthesis requires careful attention to reaction parameters to maximize yield and stereoselectivity. The process is divided into two primary stages: the initial asymmetric reduction to establish the chiral center, followed by the functional group interconversion to complete the molecular architecture. Operators must ensure strict temperature control during the borane addition phase, as exothermic events can compromise enantioselectivity. The use of anhydrous solvents such as tetrahydrofuran or toluene is critical to prevent premature hydrolysis of the borane reagent. Following the reduction, the one-pot nitro reduction step demands precise hydrogen pressure management to ensure complete conversion without affecting the benzyloxy protecting group. The detailed standardized synthesis steps below outline the specific stoichiometric ratios and operational sequences required to achieve the reported >75% total yield and >99.8% chiral purity.
- Perform asymmetric reduction of 1-(4-(benzyloxy)-3-nitrophenyl)-2-bromoethanone (II) using (3aS-cis)-(-)-3,3a,8,8a-tetrahydro-2H-indeno[1,2-d]oxazole-2-isopropylborane (IV) and borane dimethyl sulfide to obtain chiral alcohol (III).
- Conduct nitro reduction of compound (III) using a platinum catalyst under hydrogen pressure, followed immediately by formylation with formic acid and acetic anhydride.
- Isolate the final product (R)-N-(2-(benzyloxy)-5-(2-bromo-1-hydroxyethyl)phenyl)formamide (I) through solvent removal and pulping in toluene.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the technology described in CN110627673B offers transformative economic and logistical benefits. The shift from resolution-based methods to direct asymmetric synthesis fundamentally alters the cost structure of producing formoterol intermediates. By eliminating the disposal of 50% of the material as the wrong enantiomer, the effective cost of raw materials is drastically reduced, leading to substantial cost savings in the overall manufacturing budget. Additionally, the simplification of the workflow—specifically the combination of nitro reduction and formylation into a single vessel—reduces the requirement for intermediate isolation equipment, solvent volumes, and labor hours. This streamlining translates directly into a shorter production cycle, allowing manufacturers to respond more agilely to market demand fluctuations. The robustness of the reaction conditions, which do not require cryogenic temperatures or ultra-high pressures, further enhances the reliability of the supply chain by reducing the risk of batch failures due to equipment limitations.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the exceptional atom economy achieved through asymmetric catalysis. Unlike kinetic resolution, which inherently caps the maximum theoretical yield at 50%, this method utilizes the entire starting material load to generate the desired chiral product. This efficiency means that for every kilogram of raw material purchased, nearly double the amount of useful intermediate is produced compared to traditional methods. Furthermore, the removal of chiral chromatography steps eliminates the need for expensive chiral columns and the associated solvent recovery costs. The one-pot nature of the second step also minimizes solvent consumption and waste treatment expenses, contributing to a leaner, more cost-effective production model that significantly lowers the cost of goods sold (COGS) for the final API.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by complex processes that rely on scarce reagents or fragile reaction conditions. This patented method utilizes commercially available catalysts and standard hydrogenation equipment, reducing dependency on specialized proprietary technologies that might face supply bottlenecks. The mild reaction temperatures (-25 °C to 30 °C) allow the process to be run in standard glass-lined or stainless steel reactors without the need for specialized cryogenic infrastructure, making it easier to transfer technology between different manufacturing sites. This flexibility ensures that production can be scaled or shifted geographically without significant capital expenditure, thereby securing the supply chain against regional disruptions. The high yield and purity also reduce the likelihood of batch rejections, ensuring a steady flow of qualified material to downstream API synthesis units.
- Scalability and Environmental Compliance: As regulatory bodies increasingly scrutinize the environmental footprint of pharmaceutical manufacturing, this process offers a compliant pathway for expansion. The high atom economy inherently reduces the volume of chemical waste generated per unit of product, aligning with green chemistry metrics. The avoidance of chlorinated solvents in favor of toluene or THF, combined with the reduced solvent load from the one-pot procedure, simplifies waste stream management and lowers the cost of environmental compliance. The process is explicitly designed for industrial mass production, meaning it can be scaled from pilot plant quantities to multi-ton annual production capacities with minimal re-optimization. This scalability ensures that as the demand for respiratory medications grows, the supply of this critical intermediate can expand in tandem without compromising quality or environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. They are derived from the specific advantageous effects and experimental data presented in the patent documentation. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this method into their existing production lines. The answers highlight the specific improvements in purity and yield that differentiate this technology from prior art.
Q: What is the chiral purity achieved by this new synthesis method?
A: The method described in patent CN110627673B achieves a chiral purity of greater than 99.5% for the intermediate alcohol (III) and greater than 99.8% for the final formoterol key intermediate (I), significantly exceeding previous literature standards.
Q: How does this process improve atom economy compared to traditional resolution?
A: Unlike traditional chiral column separation or kinetic resolution which discard up to 50% of the material as the unwanted enantiomer, this asymmetric reduction utilizes the starting material efficiently with almost no invalid enantiomer generation, drastically improving atom economy.
Q: Can this process be scaled for industrial production?
A: Yes, the patent explicitly states the method is suitable for industrial mass production due to its mild reaction conditions (-25 to 30 °C), simple operation, and the elimination of complex purification steps like chiral chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Formoterol Intermediate Supplier
The technological advancements outlined in patent CN110627673B demonstrate the immense potential for optimizing the supply of critical respiratory drug intermediates. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such innovative laboratory protocols into robust commercial realities. As a dedicated CDMO partner, we bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from development to market. Our facilities are equipped with state-of-the-art hydrogenation reactors and stringent purity specifications are maintained through our rigorous QC labs, guaranteeing that every batch of (R)-N-(2-(benzyloxy)-5-(2-bromo-1-hydroxyethyl)phenyl)formamide meets the highest international standards. We understand that consistency is key in the pharmaceutical supply chain, and our commitment to quality assurance ensures that you receive a high-purity pharmaceutical intermediate that supports the efficacy of your final drug product.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. Our team is ready to provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this asymmetric reduction route for your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data from our recent pilot runs and to discuss route feasibility assessments tailored to your manufacturing needs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable pharmaceutical intermediate supplier committed to driving efficiency, quality, and innovation in the global healthcare market.
