Advanced Green Synthesis of Cortisone Acetate for Commercial Scale-up and Cost Efficiency
Advanced Green Synthesis of Cortisone Acetate for Commercial Scale-up and Cost Efficiency
Introduction to Patent CN103724386A and Technological Breakthroughs
The pharmaceutical industry is constantly seeking robust manufacturing pathways that balance high purity with environmental sustainability, a challenge vividly addressed in the technical disclosure of patent CN103724386A. This pivotal document outlines a revolutionary preparation method for cortisone acetate, a critical adrenal cortex hormone drug used extensively in treating hypocortisolism and serving as a vital intermediate for other steroid derivatives. The core innovation lies in a strategic shift from traditional, hazardous oxidation agents to a greener, more cost-effective sodium hypochlorite system. By fundamentally re-engineering the oxidation step, this technology not only enhances the overall yield from a historical baseline of approximately 70% to over 88% but also eliminates the generation of toxic chromium-containing wastewater. For global procurement and R&D teams, this represents a significant opportunity to optimize the supply chain for high-purity pharmaceutical intermediates while adhering to increasingly stringent environmental regulations. The method leverages hydrocortisone as a starting material, utilizing a seamless one-pot strategy that minimizes intermediate isolation steps, thereby reducing solvent consumption and operational complexity. This report analyzes the technical merits and commercial implications of this process for stakeholders aiming to secure a reliable cortisone acetate supplier.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of cortisone acetate has relied heavily on oxidation processes utilizing chromic anhydride, a reagent known for its strong oxidizing power but equally notorious for its severe environmental and safety drawbacks. In traditional workflows, chromic anhydride is often paired with manganous chloride tetrahydrate as a retarder to control the reaction, yet the aggressive nature of the oxidant frequently leads to over-oxidation of the steroid nucleus, specifically attacking the delta-4(5) double bond and the 17-alpha-hydroxy group. This lack of selectivity results in the formation of structurally fractured by-products, which not only depresses the final yield but also complicates the purification process, requiring extensive downstream processing to meet pharmacopoeia standards. Furthermore, the environmental burden of this legacy technology is immense, as the resulting effluent contains high concentrations of hexavalent and trivalent chromium ions, both of which are highly toxic and carcinogenic. Managing this hazardous waste imposes substantial costs on manufacturers, involving complex treatment protocols to ensure compliance with environmental discharge limits, thus eroding the profit margins associated with cost reduction in pharmaceutical intermediates manufacturing. Additionally, alternative green oxidants like molecular oxygen or hydrogen peroxide have faced adoption barriers due to the requirement for expensive noble metal catalysts or complex phase-transfer systems that are difficult to recycle.
The Novel Approach
In stark contrast to these legacy constraints, the novel approach detailed in the patent introduces a streamlined oxidation protocol that utilizes sodium hypochlorite, a commercially abundant and economically favorable oxidizing agent. This method ingeniously bypasses the need for isolating the intermediate hydrocortisone acetate after the initial acetylation step, allowing the oxidation to proceed directly within the acetylation reaction liquid. By employing sodium bromide as a catalyst in conjunction with the hypochlorite, the system achieves high selectivity for the alcohol oxidation without degrading the sensitive steroid backbone. This direct conversion strategy significantly simplifies the operational workflow, reducing the number of unit operations and minimizing the exposure of personnel to hazardous materials. The elimination of chromium-based reagents means that the wastewater profile is drastically improved, removing the liability associated with heavy metal disposal and aligning the production process with modern green chemistry principles. Moreover, the avoidance of expensive catalysts like TEMPO, which are difficult to recover in steroid applications due to solubility mismatches, further enhances the economic viability of this route. This technological leap provides a compelling value proposition for supply chain heads focused on reducing lead time for high-purity pharmaceutical intermediates while ensuring long-term operational sustainability.
Mechanistic Insights into Sodium Hypochlorite-Bromide Catalyzed Oxidation
The chemical elegance of this process lies in the in-situ generation of active oxidizing species within a homogeneous phase that accommodates both the lipophilic steroid substrate and the hydrophilic oxidant. The reaction initiates with the acetylation of hydrocortisone using glacial acetic acid and acetic anhydride, catalyzed by barium acetate, which protects the hydroxyl groups and prepares the molecule for selective oxidation. Following this, the introduction of sodium hypochlorite and sodium bromide into the acidic reaction medium facilitates the formation of hypobromous acid or related active bromine species, which act as the true oxidizing agents for the secondary alcohol at the C-11 position. This mechanism ensures that the oxidation proceeds under mild conditions, typically between 10°C and 15°C, preventing the thermal degradation often seen with harsher oxidants. The solvent system, primarily composed of the acetic acid from the previous step, provides sufficient solubility for the steroid intermediate, negating the need for additional phase-transfer catalysts that often complicate work-up procedures. This precise control over reaction kinetics allows for the preservation of the delta-4(5) double bond, a critical structural feature for the biological activity of cortisone acetate, thereby ensuring a high-quality impurity profile.
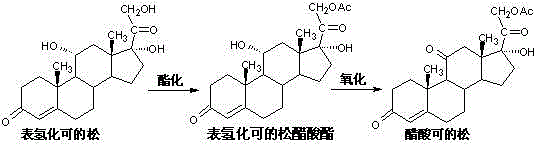
Furthermore, the impurity control mechanism is inherently robust due to the specificity of the bromide-catalyzed hypochlorite oxidation. Unlike chromic anhydride, which can indiscriminately attack various electron-rich sites on the steroid nucleus, this system targets the allylic alcohol functionality with high fidelity. The subsequent purification step involves a decolorization and recrystallization process using a mixed solvent system of methanol and dichloromethane, which effectively removes trace organic impurities and residual salts. The result is a final product with a content exceeding 99.0%, comfortably meeting the rigorous specifications of the Chinese Pharmacopoeia and international standards. For R&D directors, understanding this mechanistic pathway is crucial for validating the scalability of the process, as it demonstrates that high purity is achievable without resorting to complex chromatographic separations. The ability to maintain structural integrity while achieving such high conversion rates underscores the feasibility of this route for the commercial scale-up of complex steroid intermediates, offering a reliable alternative to older, more polluting technologies.
How to Synthesize Cortisone Acetate Efficiently
The synthesis of cortisone acetate via this green oxidation pathway is designed for operational simplicity and high throughput, making it an ideal candidate for industrial adoption. The process begins with the precise weighing of hydrocortisone and its dissolution in glacial acetic acid, followed by the controlled addition of acetic anhydride and barium acetate catalyst to drive the acetylation to completion at moderate temperatures. Once the acetylation is confirmed, the reaction mixture is cooled, and the oxidizing solution containing sodium hypochlorite and sodium bromide is added dropwise to manage the exotherm and ensure selective oxidation.
- Perform acetylation of hydrocortisone using glacial acetic acid and acetic anhydride with barium acetate catalyst at 20-35°C.
- Without isolating the intermediate, add sodium hypochlorite and sodium bromide directly to the acetylation liquid for oxidation at 10-15°C.
- Quench the reaction in ice water, filter the crude product, and recrystallize using a methanol-dichloromethane mixed solvent system.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented technology translates into tangible strategic advantages that extend beyond mere technical performance. The primary benefit is the substantial cost savings achieved through the replacement of expensive and regulated chromic anhydride with low-cost sodium hypochlorite, a commodity chemical with a stable global supply. This shift not only reduces the raw material expenditure but also eliminates the significant overhead costs associated with the treatment and disposal of hazardous chromium waste, which can be a major financial burden in strict regulatory environments. Additionally, the simplified process flow, which avoids intermediate isolation and expensive catalyst recovery, reduces labor hours and energy consumption, further driving down the cost of goods sold. These efficiencies allow suppliers to offer more competitive pricing structures without compromising on quality, providing a buffer against market volatility in raw material costs.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and toxic chromium reagents removes the need for specialized waste treatment infrastructure, leading to significantly reduced operational expenditures. By avoiding the use of non-recyclable catalysts like TEMPO, the process minimizes material loss and reduces the frequency of reactor cleaning and maintenance. The higher overall yield of over 88% means that less starting material is required to produce the same amount of final product, maximizing the utility of every kilogram of hydrocortisone input. These factors combine to create a leaner manufacturing model that is resilient to price fluctuations in the specialty chemical market.
- Enhanced Supply Chain Reliability: Utilizing widely available reagents such as sodium hypochlorite and sodium bromide ensures that production is not held hostage by the supply constraints of niche or imported catalysts. The robustness of the reaction conditions, which tolerate a range of temperatures and concentrations, reduces the risk of batch failures and ensures consistent output volumes. This reliability is critical for maintaining continuous supply to downstream pharmaceutical manufacturers who depend on just-in-time delivery models. Furthermore, the reduced environmental footprint simplifies the permitting process for manufacturing sites, securing long-term operational licenses and preventing supply disruptions due to regulatory crackdowns.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the successful transition from laboratory to pilot scales without the need for complex engineering controls. The absence of toxic heavy metals in the effluent stream simplifies wastewater management, allowing facilities to meet increasingly strict environmental discharge standards with ease. This compliance capability is a key differentiator in the global market, where customers are increasingly auditing their suppliers for sustainability credentials. The ability to produce high-purity cortisone acetate with minimal environmental impact positions the manufacturer as a preferred partner for eco-conscious pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green synthesis method, derived directly from the patent's experimental data and comparative analysis. These insights are intended to clarify the operational benefits and quality assurances associated with this advanced manufacturing route.
Q: How does this method improve environmental compliance compared to traditional chromic anhydride oxidation?
A: This method replaces toxic chromic anhydride with sodium hypochlorite, completely eliminating the discharge of hazardous Cr6+ and Cr3+ wastewater, thereby significantly reducing environmental treatment costs and regulatory risks.
Q: What is the expected purity and yield advantage of this novel oxidation route?
A: The patented process achieves a total recovery rate of over 88%, which is substantially higher than the conventional 70% yield, while maintaining product content above 99.0% meeting pharmacopoeia standards.
Q: Does this process require expensive phase-transfer catalysts or TEMPO?
A: No, the process utilizes the solubility properties of the acetylation liquid itself, removing the need for expensive, non-recyclable TEMPO catalysts or additional phase-transfer agents, simplifying the supply chain.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cortisone Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes like the one described in patent CN103724386A to meet the evolving demands of the global pharmaceutical market. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this green oxidation technology are fully realized in large-scale manufacturing. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of cortisone acetate meets the highest international standards for potency and impurity profiles. We are committed to leveraging our technical expertise to deliver cost-effective solutions that enhance your supply chain resilience.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis method can be integrated into your sourcing strategy. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the potential economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your partnership with NINGBO INNO PHARMCHEM is built on a foundation of transparency, quality, and shared success in the competitive landscape of fine chemical intermediates.
