Advanced Hydrocortisone Synthesis: Overcoming Impurity Challenges via Hypochlorite Treatment and Resin Hydrolysis
The pharmaceutical industry continuously seeks robust manufacturing pathways for critical corticosteroids like hydrocortisone, balancing high purity requirements with environmental sustainability. A significant breakthrough in this domain is detailed in Chinese Patent CN114075258A, which discloses a novel preparation method utilizing a hypochlorite impurity removal reaction coupled with an IRA-400 resin hydrolysis step. This technical advancement addresses long-standing challenges in steroid synthesis, specifically the difficulty of removing structurally similar impurities and the reliance on toxic halogenating agents. By shifting from complex multi-step halogenation sequences to a streamlined purification and hydrolysis protocol, this method achieves a total mass yield exceeding 75% and a final product purity greater than 99.5%. For global supply chain leaders and R&D directors, this represents a pivotal shift towards greener, more efficient pharmaceutical intermediates manufacturing that aligns with modern regulatory standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of hydrocortisone has been plagued by cumbersome reaction sequences and severe environmental constraints. Traditional routes, such as those disclosed in earlier patents, often commence with 17-hydroxy-4,9-diene-pregna-3,20-dione and proceed through a arduous series of bromination, debromination, iodination, substitution, and hydrolysis steps. These pathways are not only operationally intensive but also rely heavily on elemental iodine, a reagent known for its high toxicity and unfavorable environmental profile. Furthermore, alternative routes starting from anecortave acetate have demonstrated critical flaws; while they attempt to simplify the skeleton construction, they frequently generate persistent impurities that share structural homology with the target molecule. As illustrated in the reaction scheme below, these impurities are notoriously difficult to separate using standard refining techniques, creating a bottleneck that restricts large-scale industrial production and compromises the quality of the final active pharmaceutical ingredient.
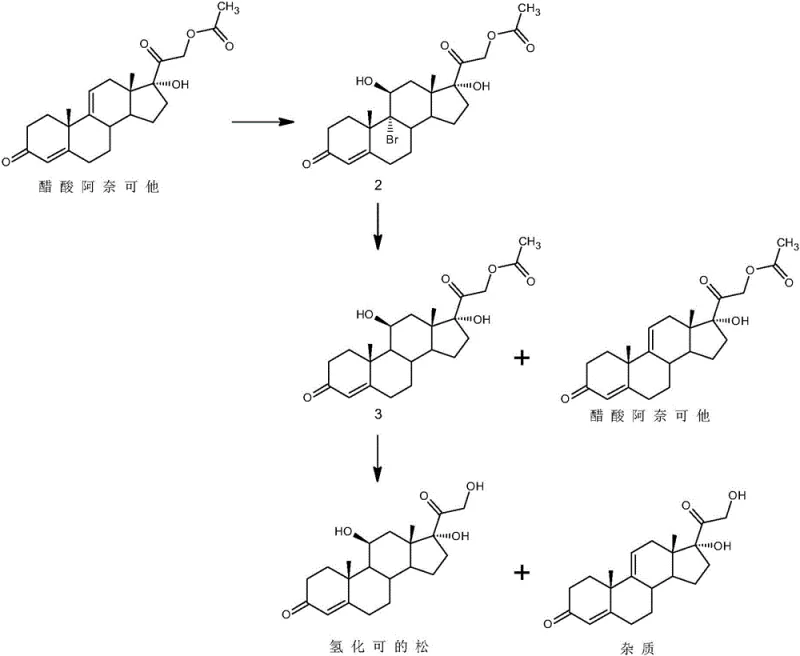
The Novel Approach
In stark contrast to these legacy methods, the technology described in CN114075258A introduces a paradigm shift by focusing on the purification of crude hydrocortisone acetate followed by mild enzymatic-mimetic hydrolysis. Instead of building the molecule through hazardous halogenation, this approach takes a crude debrominated substance (hydrocortisone acetate) and subjects it to a controlled hypochlorite treatment. This step effectively converts refractory impurities into removable derivatives without damaging the core steroid structure. Subsequently, the process employs IRA-400 resin for hydrolysis, replacing harsh liquid acids or bases with a solid-phase catalyst. This innovation drastically simplifies the workflow, eliminating the need for toxic iodine reagents entirely and reducing the generation of hazardous waste, thereby offering a superior solution for cost reduction in pharmaceutical manufacturing while ensuring consistent product quality.
Mechanistic Insights into Hypochlorite-Mediated Purification and Resin Hydrolysis
The core chemical ingenuity of this patent lies in the selective reactivity of hypochlorous acid under buffered conditions. In the first stage, sodium hypochlorite is introduced into a weakly acidic system maintained by glacial acetic acid. Under these specific pH conditions, hypochlorous acid acts as a selective oxidant that targets the specific functional groups present in the stubborn impurities—likely unsaturated bonds or susceptible hydroxyl configurations found in byproducts like anecortave acetate derivatives. This reaction transforms these lipophilic, hard-to-separate impurities into more polar or soluble species that can be easily washed away or left in the mother liquor during recrystallization. Crucially, the reaction conditions (15-40°C) are mild enough to preserve the integrity of the hydrocortisone acetate backbone, preventing over-oxidation or degradation of the valuable API intermediate.
Following purification, the hydrolysis mechanism leverages the unique properties of the IRA-400 strong base anion exchange resin. Unlike traditional homogeneous hydrolysis which requires stoichiometric amounts of strong bases and generates significant salt waste, the resin provides a heterogeneous catalytic surface. The process operates at low temperatures (-10°C to 10°C), which kinetically favors the hydrolysis of the C21-acetate ester while suppressing side reactions such as epimerization or dehydration. The resin captures the acetic acid byproduct, driving the equilibrium forward and simplifying the workup to a mere filtration step. This mechanistic elegance ensures that the final hydrocortisone product emerges with exceptional purity (>99.5%) and minimal contamination, validating the process as a highly reliable method for producing high-purity pharmaceutical intermediates suitable for sensitive therapeutic applications.
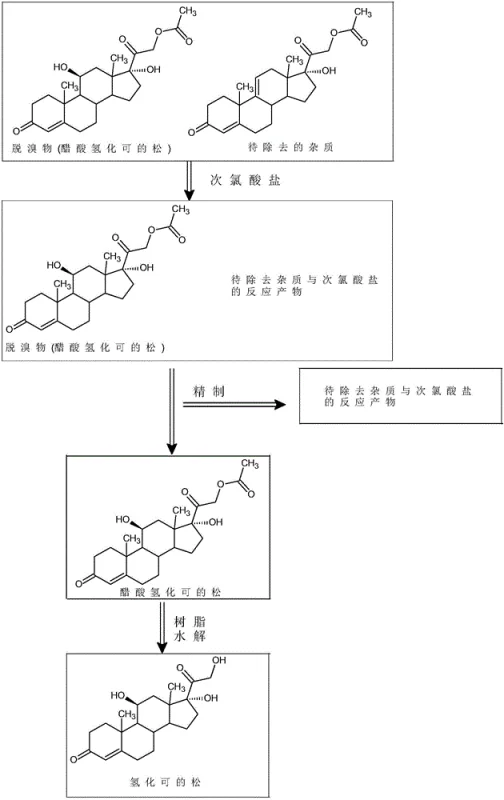
How to Synthesize Hydrocortisone Efficiently
The implementation of this synthesis route requires precise control over solvent systems and reagent addition sequences to maximize yield and purity. The process begins with the dissolution of crude hydrocortisone acetate in a optimized mixed solvent system, typically comprising dichloromethane and an alcohol such as methanol or isopropanol. The subsequent addition of glacial acetic acid creates the necessary buffer capacity before the introduction of the sodium hypochlorite solution. Following the oxidation and quenching steps, the refined acetate undergoes the critical resin hydrolysis. For a comprehensive understanding of the specific operational parameters, including exact solvent ratios and temperature ramps, please refer to the standardized synthesis guide provided below.
- Dissolve crude hydrocortisone acetate in a mixed organic solvent (dichloromethane/alcohol), add glacial acetic acid, and treat with sodium hypochlorite at 15-40°C to oxidize impurities.
- Quench residual oxidant with sodium metabisulfite, concentrate, and recrystallize to obtain refined hydrocortisone acetate with >99.2% purity.
- Hydrolyze the refined acetate using IRA-400 resin in organic solvent at low temperature (-10°C to 10°C), then filter and crystallize to yield final hydrocortisone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this hypochlorite-resin methodology offers transformative economic and logistical benefits. By eliminating the reliance on elemental iodine and complex multi-step halogenation sequences, manufacturers can significantly reduce the costs associated with hazardous material handling, storage, and waste disposal. The simplification of the process flow directly translates to shorter production cycles and reduced equipment occupancy time, enhancing overall plant throughput. Furthermore, the use of common, commercially available reagents like sodium hypochlorite and macro-reticular resins ensures a stable supply chain, mitigating the risks associated with sourcing specialized or regulated chemicals. This robustness makes the method ideal for the commercial scale-up of complex pharmaceutical intermediates, ensuring consistent availability for downstream formulation.
- Cost Reduction in Manufacturing: The elimination of toxic iodine reagents removes the substantial financial burden of specialized waste treatment and environmental compliance fees. Additionally, the replacement of energy-intensive distillation or complex extraction steps with simple filtration during the resin hydrolysis phase drastically lowers utility consumption. The high yield (>75% total mass yield) minimizes raw material loss, ensuring that every kilogram of starting material contributes maximally to the final output, thereby driving down the cost per kilogram of the active ingredient.
- Enhanced Supply Chain Reliability: By utilizing widely available industrial chemicals such as sodium hypochlorite and standard ion-exchange resins, the process decouples production from the volatility of specialized reagent markets. The operational simplicity reduces the dependency on highly specialized technical operators, allowing for more flexible staffing and reduced training overheads. This accessibility ensures that production schedules remain uninterrupted, reducing lead time for high-purity pharmaceutical intermediates and securing a steady flow of materials for global distribution networks.
- Scalability and Environmental Compliance: The mild reaction conditions (near room temperature for purification and low temperature for hydrolysis) impose low thermal stress on reactor vessels, allowing for easy scaling from pilot batches to multi-ton commercial production without requiring exotic metallurgy. The absence of heavy metal catalysts and toxic halogens simplifies the environmental impact assessment, facilitating faster regulatory approvals in stringent markets. This eco-friendly profile aligns perfectly with corporate sustainability goals, positioning the manufacturer as a responsible partner in the global pharmaceutical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and validation of this synthesis route. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on impurity profiles and process robustness. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer.
Q: How does the hypochlorite treatment remove impurities without degrading hydrocortisone acetate?
A: Under weakly acidic conditions generated by glacial acetic acid, sodium hypochlorite forms hypochlorous acid. This species selectively reacts with difficult-to-remove impurities (such as anecortave acetate derivatives) to form polar or soluble byproducts that are easily separated during recrystallization, while the stable steroid backbone of hydrocortisone acetate remains intact.
Q: Why is IRA-400 resin preferred over traditional acid/base hydrolysis for the final step?
A: Traditional homogeneous acid or base hydrolysis often leads to side reactions and degradation of the sensitive steroid structure. IRA-400 resin provides a heterogeneous, mild alkaline environment that facilitates selective ester hydrolysis at low temperatures (-10°C), minimizing impurity generation and simplifying post-reaction filtration.
Q: What are the environmental advantages of this method compared to iodine-based routes?
A: This method eliminates the use of elemental iodine and toxic iodination reagents required in conventional multi-step syntheses. By avoiding heavy halogen waste and reducing the number of synthetic steps through direct purification, the process significantly lowers the environmental footprint and waste disposal costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hydrocortisone Supplier
The technological advancements outlined in CN114075258A underscore the potential for greener, more efficient steroid synthesis, a vision that NINGBO INNO PHARMCHEM is fully equipped to realize. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are seamlessly translated into industrial reality. Our facilities are outfitted with state-of-the-art reactors capable of handling sensitive low-temperature resin reactions and rigorous QC labs dedicated to maintaining stringent purity specifications, guaranteeing that every batch meets the highest international pharmacopoeia standards.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this advanced manufacturing capability. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
