Revolutionizing Amino Alcohol Production: A Safe, One-Pot Reduction Strategy for Commercial Scale
Revolutionizing Amino Alcohol Production: A Safe, One-Pot Reduction Strategy for Commercial Scale
The landscape of fine chemical synthesis is constantly evolving, driven by the need for safer, more efficient, and cost-effective methodologies. Patent CN1075055C introduces a transformative approach to the reduction of amino acids and their derivatives into valuable amino alcohols, addressing critical bottlenecks in traditional synthetic routes. This technology leverages a ingenious "single-vessel" strategy that combines esterification and reduction into a seamless workflow, eliminating the need for intermediate isolation. For R&D directors and process chemists, this represents a significant leap forward in operational simplicity and safety profile. The method utilizes readily available alkali metal borohydrides without the need for hazardous activators, ensuring that chiral centers are preserved with high fidelity throughout the transformation. As a leading manufacturer, we recognize the immense potential of this patented route to streamline the supply chain for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the reduction of amino acids to amino alcohols has been fraught with significant technical and safety challenges that hinder large-scale adoption. Traditional protocols often rely on lithium aluminum hydride (LiAlH4), a powerful but notoriously dangerous reducing agent that requires strictly anhydrous conditions and specialized handling equipment due to its pyrophoric nature. Furthermore, alternative methods involving sodium borohydride typically necessitate the use of activators such as iodine or Lewis acids, which introduce additional impurities and complicate the downstream purification process. Perhaps most critically, conventional two-step approaches mandate the isolation and drying of the ester intermediate, a time-consuming operation that drastically increases solvent usage, labor costs, and overall cycle time. These inefficiencies create substantial barriers for procurement managers seeking reliable sources of complex chiral building blocks at competitive price points.
The Novel Approach
In stark contrast, the methodology outlined in CN1075055C offers a streamlined solution that bypasses these historical limitations through a clever integration of reaction steps. The core innovation lies in the ability to perform the reduction directly on the crude esterification mixture without isolating the intermediate ester. By employing unactivated alkali metal or alkaline earth metal borohydrides, such as sodium borohydride, the process avoids the need for hazardous activators while maintaining high reaction efficiency. This "one-pot" design not only simplifies the operational workflow but also significantly enhances the safety profile of the manufacturing process, making it ideal for commercial scale-up. The result is a robust synthetic route that delivers high yields of amino alcohols while minimizing waste generation and reducing the dependency on expensive, specialized reagents.
Mechanistic Insights into One-Pot Esterification and Borohydride Reduction
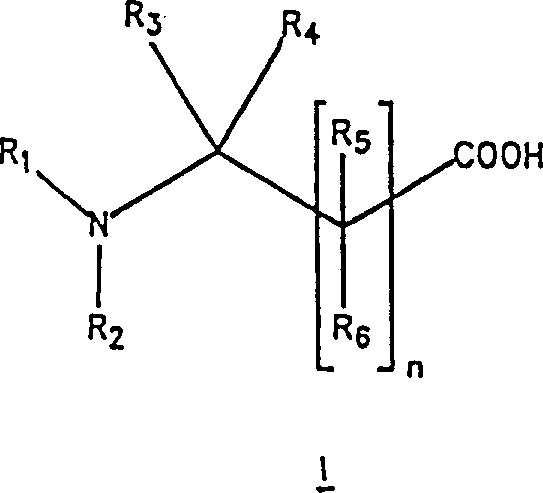
The mechanistic elegance of this process begins with the acid-catalyzed esterification of the amino acid or its derivative in a lower alcohol solvent, such as ethanol or methanol. Under reflux conditions, the carboxylic acid group is converted into an ester in situ, generating a reaction mixture that contains the activated substrate ready for reduction. Crucially, the reaction conditions are tuned to ensure that the amino group remains protected or compatible, preventing unwanted side reactions. Following esterification, the mixture is treated directly with a borohydride reducing agent. Unlike traditional methods that require activated hydrides to reduce esters efficiently, this patent demonstrates that unactivated borohydrides can effectively reduce the ester functionality when applied directly to the reaction matrix. This unexpected reactivity allows for the conversion of the ester to the primary alcohol functionality with remarkable selectivity.
From a stereochemical perspective, the preservation of chirality is paramount for the production of bioactive intermediates used in drug discovery and asymmetric synthesis. The mild conditions employed in this reduction protocol prevent the epimerization or racemization of sensitive chiral centers adjacent to the reaction site. This is achieved by avoiding strong bases or high temperatures that could facilitate enolization or other racemization pathways. The resulting amino alcohols retain their optical purity, making them suitable precursors for the synthesis of photoactive compounds, chiral catalysts, and peptide isosteres. For quality control teams, this inherent stability translates to simpler analytical validation and higher confidence in the consistency of the final product specifications across different batches.
How to Synthesize Amino Alcohols Efficiently
The practical implementation of this synthesis route is designed for ease of execution in standard chemical manufacturing facilities. The process initiates by suspending or dissolving the starting amino acid in a C1 to C5 alcohol, followed by the addition of a catalytic amount of acid such as sulfuric acid or p-toluenesulfonic acid. The mixture is heated to reflux to drive the esterification to completion, monitored typically by HPLC to ensure high conversion. Once the ester formation is confirmed, the reaction mixture is cooled, and excess solvent may be removed to concentrate the intermediate. Without any purification of the ester, a base may be added to neutralize residual acid, followed by the controlled addition of the borohydride reducing agent. The detailed standardized synthesis steps see the guide below.
- Dissolve the amino acid or derivative in a C1-C5 alcohol (e.g., ethanol) and add an acid catalyst like sulfuric acid, then reflux to form the ester intermediate in situ.
- Without isolating the ester, cool the reaction mixture and optionally concentrate it to remove excess solvent before neutralizing excess acid with a base.
- Add an unactivated alkali metal borohydride (e.g., NaBH4) directly to the mixture to reduce the ester to the corresponding amino alcohol, preserving chirality.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented technology offers compelling economic and logistical benefits that extend beyond simple yield improvements. The elimination of the intermediate isolation step fundamentally alters the cost structure of amino alcohol production by removing unit operations that are both capital and labor-intensive. By consolidating two distinct chemical transformations into a single vessel, manufacturers can significantly reduce the total processing time and the volume of solvents required, leading to a smaller environmental footprint and lower waste disposal costs. This efficiency gain is critical in a market where margin pressure is constant and the demand for sustainable manufacturing practices is increasing. Furthermore, the use of commodity chemicals like sodium borohydride and ethanol ensures a stable and predictable supply of raw materials, mitigating the risk of shortages associated with exotic reagents.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the removal of the isolation and drying stages for the ester intermediate. In traditional workflows, these steps consume significant amounts of energy, filtration media, and solvent, all of which contribute to the final cost of goods sold. By proceeding directly to reduction, the process eliminates these overheads entirely. Additionally, the substitution of expensive and hazardous lithium aluminum hydride with inexpensive sodium borohydride results in substantial raw material cost savings. The reduced stoichiometric requirement for the borohydride, as evidenced by the patent examples using near-stoichiometric amounts rather than large excesses, further enhances the economic viability of the route for large-scale production.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on widely available, non-specialized reagents. Sodium borohydride and common alcohols are produced globally in massive quantities, ensuring that production schedules are not held hostage by the availability of niche catalysts or activators. The simplified workflow also reduces the complexity of the manufacturing schedule, allowing for faster turnaround times and increased throughput capacity. This agility enables suppliers to respond more rapidly to fluctuations in market demand, ensuring consistent delivery of high-purity intermediates to downstream pharmaceutical customers. The robustness of the method against minor variations in reaction conditions further guarantees batch-to-batch consistency.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new safety and environmental challenges, but this one-pot method is inherently designed for scalability. The avoidance of pyrophoric reagents removes the need for specialized inert atmosphere equipment and rigorous safety protocols associated with lithium aluminum hydride, simplifying facility requirements. From an environmental standpoint, the reduction in solvent usage and the generation of benign byproducts align with green chemistry principles. The process minimizes the release of hazardous waste streams, facilitating easier compliance with increasingly stringent environmental regulations. This makes the technology not only economically attractive but also socially responsible, appealing to stakeholders focused on sustainable development goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this reduction technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the method's capabilities and limitations. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers highlight the balance between operational simplicity and chemical performance that defines this innovative approach.
Q: Why is the one-pot method superior to traditional lithium aluminum hydride reduction?
A: Traditional methods using lithium aluminum hydride require strictly anhydrous conditions and pose significant safety risks due to pyrophoric nature. The patented one-pot method uses safer, unactivated sodium borohydride in alcoholic solvents, eliminating the need for hazardous reagents and complex isolation steps.
Q: Does this reduction process cause racemization of chiral centers?
A: No, the process is specifically designed to preserve chiral integrity. By avoiding harsh activation conditions and utilizing mild reduction temperatures, the method ensures high optical purity, which is critical for producing photoactive compounds and resolving racemic mixtures.
Q: What are the cost benefits of not isolating the ester intermediate?
A: Eliminating the isolation and drying of the ester intermediate significantly reduces processing time, solvent consumption, and labor costs. This 'single-vessel' approach streamlines the workflow, leading to substantial overall cost reduction in amino alcohol manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Amino Alcohols Supplier
At NINGBO INNO PHARMCHEM, we understand that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent literature can be reliably reproduced on an industrial scale. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of amino alcohol meets the exacting standards required for pharmaceutical applications. Our infrastructure is designed to handle complex chemistries safely and efficiently, providing a secure foundation for your supply chain.
We invite you to explore how this advanced reduction technology can optimize your production costs and enhance your product quality. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific amino acid reduction needs. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate the tangible benefits of partnering with us. Let us help you secure a reliable supply of high-performance intermediates that drive your innovation forward.
