Advanced Catalytic De-enrichment Technology for High-Purity Pharmaceutical Intermediates
Advanced Catalytic De-enrichment Technology for High-Purity Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries constantly face the challenge of managing stereochemistry during the synthesis of active pharmaceutical ingredients (APIs) and their precursors. A significant bottleneck in chiral synthesis is the generation of unwanted enantiomers during resolution processes, which often leads to substantial material loss and increased production costs. Addressing this critical inefficiency, patent CN101052604A introduces a sophisticated de-enrichment method for enantiomerically enriched substrates, specifically targeting compounds containing carbon-heteroatom bonds where the carbon acts as a chiral center. This technology provides a robust pathway to convert enriched isomeric compositions back into racemic or de-enriched mixtures, thereby enabling the recycling of valuable chiral building blocks. By leveraging advanced transition metal catalysis, this process offers a strategic solution for manufacturers seeking to optimize yield and reduce waste in the production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the management of unwanted enantiomers in chiral synthesis has been a costly and environmentally burdensome endeavor. In standard resolution protocols, such as enzymatic or chemical kinetic resolution, only a maximum theoretical yield of 50% of the desired enantiomer is obtained, leaving the other half as an undesired isomer. Historically, this unwanted fraction was often discarded or subjected to harsh, non-selective chemical treatments that could degrade the molecular scaffold or require extensive purification steps to remove toxic byproducts. Furthermore, conventional racemization methods frequently rely on extreme pH conditions, high temperatures, or stoichiometric amounts of strong bases, which are incompatible with sensitive functional groups commonly found in complex drug molecules. These limitations not only inflate the cost of goods sold (COGS) due to raw material wastage but also complicate the supply chain by necessitating larger initial batches to secure sufficient quantities of the final active isomer.
The Novel Approach
The methodology outlined in patent CN101052604A represents a paradigm shift by employing a catalytic system to achieve mild and selective de-enrichment. Instead of discarding the unwanted isomer, this novel approach utilizes a transition metal catalyst in the presence of optional reaction promoters to equilibrate the enantiomeric ratio. The process operates under relatively mild conditions, typically ranging from -20°C to +110°C, and can utilize the substrate itself as a solvent or employ common organic solvents like toluene or 2-propanol. By facilitating a reversible transfer of hydrogen or utilizing specific hydrogen donors and acceptors, the catalyst effectively scrambles the stereochemical information at the chiral center without compromising the integrity of the rest of the molecule. This capability transforms what was once considered waste into a reusable feedstock, dramatically enhancing the overall efficiency of chiral synthesis routes and providing a reliable pharmaceutical intermediate supplier with a distinct competitive advantage in resource utilization.
Mechanistic Insights into Transition Metal-Catalyzed Racemization
The core of this de-enrichment technology lies in the precise engineering of the catalyst system, which preferably comprises a transition metal catalyst and an optional ligand. The patent specifies that Group VIII metals, particularly Ruthenium, Rhodium, and Iridium, are highly effective for this transformation. The catalytic cycle likely involves the formation of a metal-hydride species that interacts with the chiral substrate, temporarily removing the stereochemical definition through a planar intermediate (such as a ketone or thione in the case of alcohols and sulfides) before re-delivering the hydrogen in a non-stereoselective manner. The choice of ligand is critical; the patent highlights the use of amines, alcohols, and sulfides, with specific preference for chiral amino alcohols and diamines that can modulate the electronic and steric environment around the metal center. For instance, ligands such as norephedrine, 1-amino-2-indanol, and various substituted ethylenediamines are explicitly mentioned as effective co-catalysts that stabilize the active metal species and enhance reaction rates.
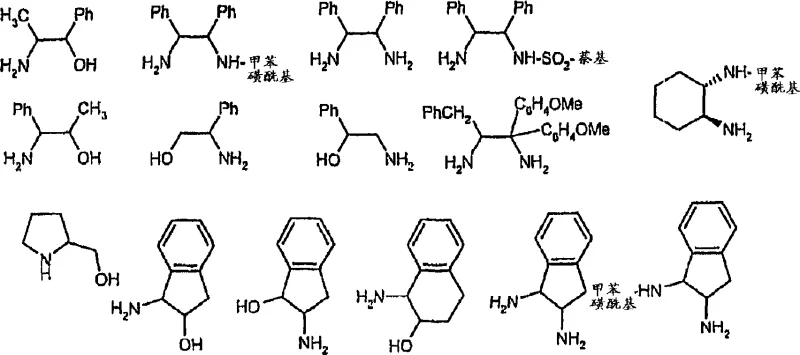
Furthermore, the structural complexity of the catalyst can be tuned to maximize activity and stability. The patent details specific transition metal halide complexes, such as those with the general formula MnXpYr, where M is the metal, X is a halide, and Y is a neutral coordinating group like a cyclopentadienyl ligand. Particularly preferred are Iridium complexes featuring pentamethylcyclopentadienyl (Cp*) ligands, which provide exceptional thermal stability and catalytic turnover. The interaction between the metal center and the substrate is further facilitated by reaction promoters, such as iodide salts (e.g., potassium iodide), which may assist in ligand exchange or stabilize cationic intermediates. The versatility of this system allows it to accommodate a wide range of substrates, including aryl alkyl carbinols and cyclic alcohols, making it a versatile tool for the commercial scale-up of complex polymer additives and fine chemicals where stereochemical purity is paramount.
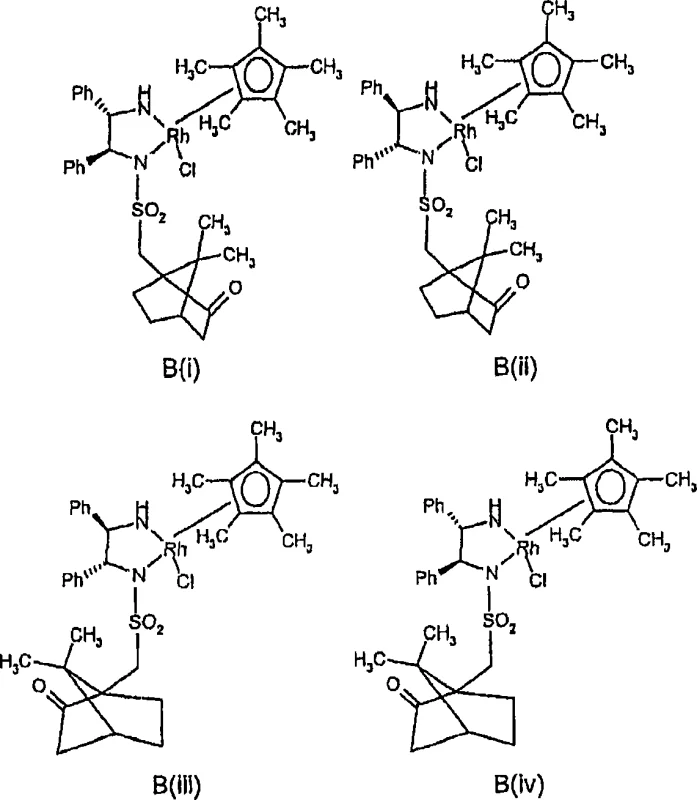
How to Synthesize De-enriched Chiral Substrates Efficiently
Implementing this de-enrichment protocol requires careful attention to the preparation of the catalyst and the control of reaction parameters to ensure optimal conversion. The process generally begins with the in situ or pre-formed combination of the transition metal precursor and the chosen ligand in a suitable solvent, followed by the addition of the enantiomerically enriched substrate. To drive the equilibrium towards the racemic state, the reaction may be conducted in the presence of a hydrogen donor, such as 2-propanol or formic acid, or a hydrogen acceptor like acetone, depending on the specific thermodynamic requirements of the substrate. The detailed standardized synthesis steps see the guide below, which outlines the precise molar ratios, temperature profiles, and workup procedures necessary to achieve consistent results in a laboratory or pilot plant setting.
- Prepare the catalyst system by combining a transition metal halide complex (e.g., Iridium or Rhodium based) with a suitable ligand such as an amino alcohol or diamine in a solvent like 2-propanol.
- Mix the enantiomerically enriched substrate (e.g., chiral alcohol) with the catalyst system and a reaction promoter such as potassium iodide or a base like potassium carbonate.
- Heat the reaction mixture to a temperature range of -20°C to +110°C, optionally adding a hydrogen donor or acceptor to facilitate the transfer hydrogenation and racemization process.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic de-enrichment technology translates directly into tangible operational improvements and risk mitigation. By enabling the recycling of unwanted isomers, manufacturers can significantly reduce the volume of raw materials required to produce a fixed amount of final API, leading to substantial cost savings in purchasing and inventory management. This efficiency gain is particularly critical for high-value chiral intermediates where the starting materials are expensive or difficult to source. Moreover, the ability to internally recycle waste streams reduces dependency on external suppliers for additional batches of racemic starting material, thereby enhancing supply chain reliability and insulating the production schedule from market volatility or logistics disruptions.
- Cost Reduction in Manufacturing: The elimination of waste disposal costs and the reduction in raw material consumption are the primary drivers for cost optimization in this process. By converting the unwanted 50% of resolved material back into usable feedstock, the effective yield of the overall synthetic route is nearly doubled without the need for new starting materials. This qualitative improvement in atom economy means that the cost per kilogram of the final product is drastically lowered, as the amortized cost of the initial chiral resolution is spread over a much larger quantity of usable material. Additionally, the mild reaction conditions reduce energy consumption compared to traditional thermal racemization methods, further contributing to lower utility costs and a smaller environmental footprint.
- Enhanced Supply Chain Reliability: Integrating this recycling step creates a more resilient supply chain by decoupling production capacity from the immediate availability of fresh racemic precursors. In scenarios where the supply of a specific chiral building block is constrained, the ability to regenerate the racemate from process waste ensures continuity of operations. This internal loop minimizes the risk of production stoppages due to raw material shortages and allows for more flexible inventory planning. Furthermore, the use of robust transition metal catalysts that can potentially be recovered or reused adds another layer of stability to the manufacturing process, ensuring that lead times for high-purity pharmaceutical intermediates remain consistent and predictable.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing standard unit operations and common solvents that are easily handled in large-scale reactors. The avoidance of stoichiometric hazardous reagents simplifies waste treatment and aligns with increasingly stringent environmental regulations regarding chemical discharge. By reducing the total mass of chemical waste generated per unit of product, manufacturers can lower their compliance costs and improve their sustainability metrics. This green chemistry approach not only meets regulatory standards but also appeals to downstream customers who are prioritizing environmentally responsible sourcing, thus strengthening the market position of the supplier in the global fine chemicals sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this de-enrichment technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the scope and applicability of the method for potential partners and technical stakeholders. Understanding these nuances is essential for evaluating the feasibility of integrating this process into existing manufacturing workflows.
Q: What is the primary advantage of this de-enrichment method over traditional disposal?
A: The primary advantage is the ability to recycle unwanted isomers from chiral resolutions back into usable racemic starting materials, significantly reducing raw material waste and improving overall process atom economy without requiring harsh chemical conditions.
Q: Which transition metals are most effective for this catalytic process?
A: According to the patent data, Group VIII transition metals are preferred, with Ruthenium, Rhodium, and Iridium showing particularly high efficacy. Iridium complexes, especially those with cyclopentadienyl ligands, are highlighted as most preferred for achieving efficient racemization.
Q: Can this process be scaled for industrial production of intermediates?
A: Yes, the process is designed for scalability, utilizing standard reaction conditions and commercially available catalysts. The method supports large-scale operations by allowing for the recycling of feedstock and minimizing the need for complex separation steps typically associated with chiral purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Alcohols Supplier
The technological advancements described in patent CN101052604A underscore the importance of having a manufacturing partner with deep expertise in chiral chemistry and process optimization. NINGBO INNO PHARMCHEM stands ready to leverage these insights to deliver superior quality intermediates to the global market. As a dedicated CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our commitment to quality is backed by stringent purity specifications and rigorous QC labs that monitor every batch to guarantee consistency and compliance with international pharmacopeial standards.
We invite you to collaborate with us to explore how this de-enrichment technology can be tailored to your specific synthesis needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis to quantify the potential economic benefits for your specific product line. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our advanced catalytic capabilities can drive efficiency and value for your organization.
