Advanced Purification Technology for High-Purity Cariprazine Manufacturing
Advanced Purification Technology for High-Purity Cariprazine Manufacturing
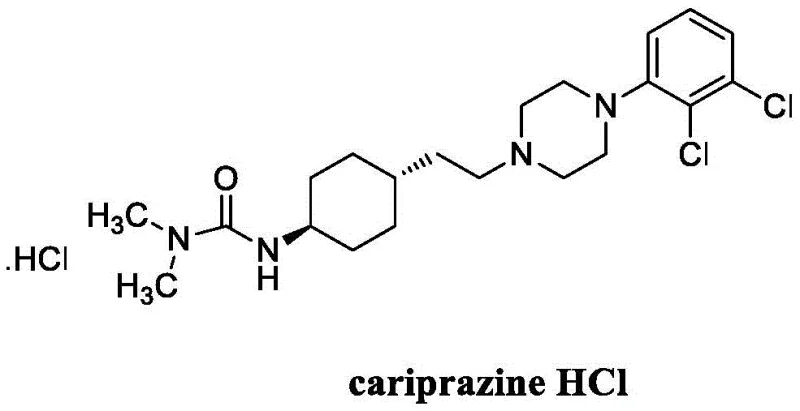
The pharmaceutical landscape for antipsychotic medications demands exceptionally high purity standards, particularly for complex molecules like Cariprazine hydrochloride, a potent D2 and D3 receptor partial agonist used in treating schizophrenia and bipolar disorder. As detailed in patent CN111320593B, a significant technological breakthrough has been achieved in the post-synthesis refining of this critical active pharmaceutical ingredient (API). The invention introduces a sophisticated solvent engineering approach that effectively addresses the persistent challenge of removing specific dimeric impurities which traditionally compromise product quality. By optimizing the crystallization environment through a precise blend of dichloromethane, lower alcohols, and anti-solvents, this method ensures the production of Cariprazine with purity levels exceeding 99.5%. For global procurement teams and R&D directors, this represents a vital advancement in securing a reliable supply chain for high-quality psychiatric medications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Cariprazine has relied on coupling reactions between key intermediates, such as 2,3-dichlorophenyl piperazine and functionalized cyclohexyl derivatives. As illustrated in prior art like patent CN1829703, the conventional route involves the reductive amination of intermediates followed by acylation to form the final urea linkage. However, these synthetic pathways are prone to the formation of a stubborn by-product known as Impurity X, a dimeric structure that shares similar physicochemical properties with the target molecule. Traditional purification techniques, often relying solely on salt formation or simple recrystallization from single solvents, frequently fail to separate this impurity effectively. Consequently, the impurity enriches during the salification process, leading to off-specification batches that require repeated, yield-depleting recrystallization cycles. This inefficiency not only drives up manufacturing costs but also creates bottlenecks in production timelines, posing a significant risk to supply continuity for downstream drug manufacturers.
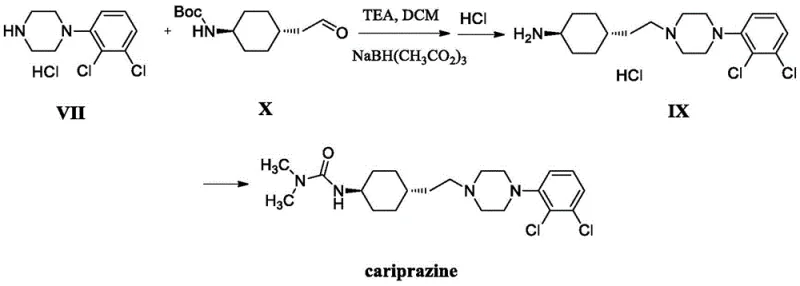
The Novel Approach
The refining method disclosed in CN111320593B offers a transformative solution by fundamentally altering the solubility dynamics during the crystallization phase. Instead of relying on brute-force purification, this novel approach employs a ternary solvent system designed to exploit the subtle solubility differences between the monomeric Cariprazine and its dimeric impurity. The process begins by dissolving the crude product in a mixture of methylene chloride and a C1-5 alcohol, creating a homogeneous phase at elevated temperatures. Subsequently, the controlled addition of an ester or ketone anti-solvent shifts the saturation point, inducing the selective precipitation of high-purity Cariprazine crystals while retaining the dimeric impurities in the mother liquor. This strategic manipulation of solvent polarity and temperature gradients allows for the efficient removal of Impurity X in a single processing step, drastically simplifying the workflow and enhancing the overall robustness of the manufacturing process.
Mechanistic Insights into Solvent-Engineered Crystallization
The efficacy of this purification strategy lies in the precise thermodynamic control exerted over the crystallization lattice formation. By utilizing dichloromethane as a primary solubilizer combined with polar protic solvents like ethanol or methanol, the system ensures complete dissolution of both the target API and the associated impurities at reflux temperatures. The critical mechanism occurs during the cooling and anti-solvent addition phase, where the solubility of the target Cariprazine decreases sharply relative to the dimeric Impurity X. The specific molecular geometry of Impurity X, which features a bis-urea linkage connecting two cyclohexyl-piperazine units, results in different intermolecular interaction energies compared to the monomer. The chosen solvent mixture disrupts the potential co-crystallization of these species, effectively trapping the impurity in the liquid phase. This selective exclusion is further enhanced by washing the filter cake with ethyl acetate, a solvent that maintains the impurity in solution while minimizing the loss of the purified product, thereby maximizing recovery rates.
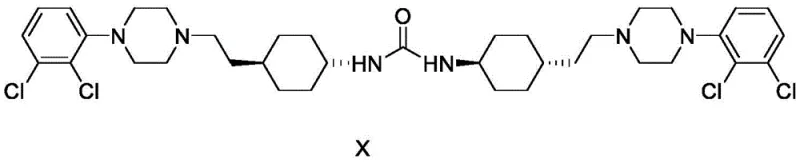
Furthermore, the control of crystallization kinetics plays a pivotal role in defining the final purity profile. The protocol specifies a controlled cooling ramp to 0-10°C followed by a sustained stirring period of 2 to 3 hours. This extended maturation time allows for Ostwald ripening, where smaller, less perfect crystals dissolve and redeposit onto larger, more stable crystal lattices that inherently exclude impurities. The result is a crystalline product with superior morphological consistency and significantly reduced surface adsorption of contaminants. From an analytical perspective, High-Performance Liquid Chromatography (HPLC) data confirms that this mechanism reduces the content of Impurity X from initial levels of roughly 0.35% in crude material down to trace levels of 0.02% to 0.04%. Such rigorous impurity control is essential for meeting the stringent regulatory requirements for CNS-active pharmaceutical ingredients, ensuring patient safety and therapeutic efficacy.
How to Synthesize High-Purity Cariprazine Efficiently
Implementing this refining protocol requires careful attention to solvent ratios and temperature profiles to replicate the high yields and purity demonstrated in the patent examples. The process is designed to be operationally simple, utilizing standard reactor equipment found in most fine chemical facilities. Operators must ensure that the initial dissolution is complete before introducing the anti-solvent to prevent premature nucleation of impure solids. The following guide outlines the standardized steps derived from the experimental data, providing a clear roadmap for process chemists to integrate this technology into their existing production lines for immediate quality improvements.
- Dissolve crude Cariprazine in a mixture of dichloromethane and C1-5 alcohol (e.g., ethanol) under heating and stirring.
- Add an ester solvent (e.g., ethyl acetate) or ketone solvent (e.g., acetone) to the clear solution to induce crystallization conditions.
- Cool the mixture to 0-10°C, stir for 2-3 hours, filter the crystals, wash with ethyl acetate, and dry under vacuum.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain executives, the adoption of this advanced refining method translates directly into tangible operational efficiencies and risk mitigation. The ability to consistently produce Cariprazine with purity levels above 99.5% eliminates the need for costly and time-consuming reprocessing loops that plague conventional manufacturing. By streamlining the purification into a single, high-yield crystallization step, manufacturers can significantly reduce the consumption of raw materials and utilities per kilogram of finished product. This efficiency gain is critical in a competitive market where margin compression is a constant pressure, allowing suppliers to offer more competitive pricing without compromising on quality standards. Moreover, the robustness of the process ensures a steady flow of qualified material, reducing the likelihood of batch failures that could disrupt downstream tablet formulation schedules.
- Cost Reduction in Manufacturing: The elimination of complex chromatographic purification or multiple recrystallization cycles leads to substantial cost savings in both labor and solvent consumption. By achieving high purity in a single pass, the process minimizes the volume of waste solvent generated, thereby lowering disposal costs and environmental compliance burdens. The use of common, commercially available solvents like dichloromethane, ethanol, and ethyl acetate ensures that raw material costs remain stable and predictable, avoiding the volatility associated with exotic reagents. This streamlined approach optimizes the overall cost of goods sold (COGS), providing a distinct economic advantage for large-scale production campaigns.
- Enhanced Supply Chain Reliability: The simplicity and reproducibility of this refining technique enhance the reliability of the supply chain by reducing process variability. With a method that consistently delivers yields over 80% and purity exceeding 99.5%, manufacturers can confidently commit to delivery schedules without the buffer stock typically required to account for failed batches. The scalability of the process means that production volumes can be ramped up rapidly to meet surges in demand for schizophrenia and bipolar disorder treatments. This reliability is paramount for pharmaceutical partners who require uninterrupted access to high-quality API intermediates to maintain their own regulatory filings and market presence.
- Scalability and Environmental Compliance: From an environmental and safety perspective, the process utilizes solvents that are well-understood and manageable within standard industrial hygiene frameworks. The reduction in processing steps inherently lowers the energy footprint of the manufacturing operation, aligning with modern green chemistry principles. The high efficiency of impurity removal reduces the load on wastewater treatment systems, as the mother liquor contains a concentrated but manageable amount of organic by-products. This facilitates easier compliance with increasingly strict environmental regulations, ensuring long-term operational sustainability and minimizing the risk of regulatory shutdowns due to effluent violations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this high-purity Cariprazine refining technology. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity for stakeholders evaluating this process for potential adoption or sourcing. Understanding these details is crucial for making informed decisions about process integration and supplier qualification.
Q: What is the primary impurity removed by this refining method?
A: The process specifically targets and removes 'Impurity X', a dimeric by-product that typically co-crystallizes with Cariprazine and is difficult to eliminate via standard salification processes.
Q: What purity levels can be achieved with this protocol?
A: Experimental data indicates that this refining method consistently achieves HPLC purity levels exceeding 99.5%, with Impurity X content reduced to approximately 0.02% - 0.04%.
Q: Is this purification method suitable for large-scale production?
A: Yes, the method utilizes common industrial solvents like dichloromethane, ethanol, and ethyl acetate, and relies on standard crystallization and filtration unit operations, making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cariprazine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the complexity of synthesizing and purifying molecules like Cariprazine requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to full-scale manufacturing is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify that every batch meets the highest international standards. Our commitment to quality assurance means that we can reliably deliver high-purity Cariprazine intermediates and APIs that satisfy the demanding requirements of global regulatory bodies.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs through the adoption of advanced processing technologies. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to evaluate how our refined manufacturing processes can enhance the stability and profitability of your pharmaceutical product portfolio. Let us be your strategic partner in delivering life-changing medications to patients worldwide.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
