Advanced Synthesis of Betamethasone Sodium Phosphate: A Breakthrough in Moisture Control and Scalability
Advanced Synthesis of Betamethasone Sodium Phosphate: A Breakthrough in Moisture Control and Scalability
The pharmaceutical industry constantly seeks robust synthetic routes for critical corticosteroid derivatives, particularly for emergency medications like Betamethasone Sodium Phosphate. Patent CN110964075B introduces a transformative preparation method that addresses longstanding challenges regarding product stability, moisture content, and purification efficiency. This technical insight report analyzes the novel process disclosed in the patent, which utilizes a unique organic acid sodium salt exchange mechanism to eliminate water generation during salification. For R&D directors and procurement specialists, this innovation represents a significant leap forward in producing high-purity API intermediates with superior physical properties. By shifting away from traditional inorganic base neutralization, the process ensures the final active pharmaceutical ingredient meets stringent pharmacopoeia standards for moisture and related substances, thereby enhancing shelf-life and therapeutic reliability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Betamethasone Sodium Phosphate has been plagued by inefficiencies inherent in traditional salification techniques. As illustrated in prior art such as US Patent 3564028 and CN101397319A, conventional routes often rely on multi-step sequences involving mesylation and iodination or direct phosphorylation followed by neutralization with inorganic bases like sodium hydroxide or sodium bicarbonate. 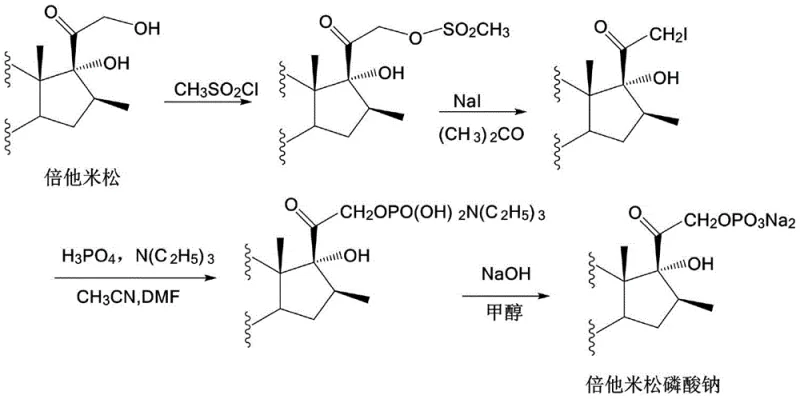 . The fundamental flaw in using inorganic bases is the stoichiometric generation of water as a byproduct during the acid-base neutralization reaction. Since Betamethasone Sodium Phosphate is highly hygroscopic, this internally generated water becomes entrapped within the crystal lattice, leading to moisture contents as high as 10-15%. Removing this water requires prolonged, high-temperature drying, which paradoxically accelerates the degradation of the thermally sensitive steroid molecule, resulting in lower purity and compromised stability.
. The fundamental flaw in using inorganic bases is the stoichiometric generation of water as a byproduct during the acid-base neutralization reaction. Since Betamethasone Sodium Phosphate is highly hygroscopic, this internally generated water becomes entrapped within the crystal lattice, leading to moisture contents as high as 10-15%. Removing this water requires prolonged, high-temperature drying, which paradoxically accelerates the degradation of the thermally sensitive steroid molecule, resulting in lower purity and compromised stability.
The Novel Approach
The methodology outlined in patent CN110964075B circumvents these issues through a strategic redesign of the salification step. Instead of employing inorganic alkalis, the inventors utilize organic acid sodium salts, such as sodium acetate or sodium formate, to convert the phosphate ester into its sodium salt form. 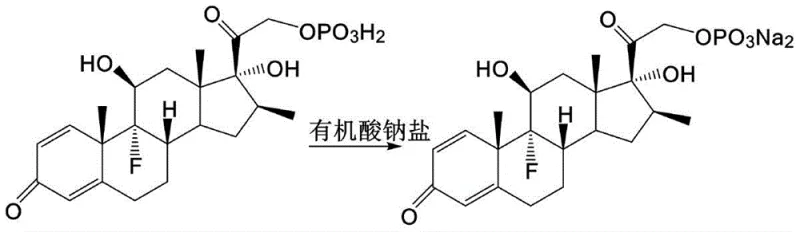 . This substitution is chemically profound because the reaction between the phosphate acid and the organic acid salt proceeds without producing water, effectively eliminating the primary source of contamination. Furthermore, the process incorporates a specialized recrystallization step using a mixed solvent system of ethers and alkanes. This modification alters the crystal habit of the intermediate from a fine powder to distinct granular crystals, which dramatically improves filtration characteristics and reduces the surface area available for moisture absorption, ultimately yielding a product with moisture content consistently below 2%.
. This substitution is chemically profound because the reaction between the phosphate acid and the organic acid salt proceeds without producing water, effectively eliminating the primary source of contamination. Furthermore, the process incorporates a specialized recrystallization step using a mixed solvent system of ethers and alkanes. This modification alters the crystal habit of the intermediate from a fine powder to distinct granular crystals, which dramatically improves filtration characteristics and reduces the surface area available for moisture absorption, ultimately yielding a product with moisture content consistently below 2%.
Mechanistic Insights into Organic Acid Salt Exchange and Crystallization
The core mechanistic advantage of this process lies in the thermodynamics of the salt exchange reaction. When Betamethasone Phosphate reacts with an inorganic base like NaOH, the reaction is essentially a proton transfer that yields the sodium salt and water (H+ + OH- -> H2O). In contrast, reacting the phosphate with sodium acetate involves an equilibrium where the stronger phosphoric acid moiety displaces the weaker acetic acid from its salt. 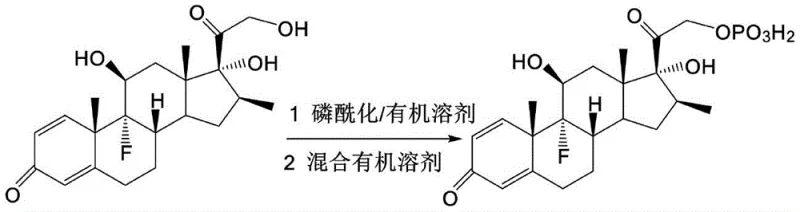 . Because no hydroxide ions are present to combine with protons, no water molecules are formed. This anhydrous environment is critical for maintaining the integrity of the phosphate ester bond, which is susceptible to hydrolysis in the presence of moisture and heat. The absence of water allows the reaction to proceed at mild temperatures (20-30°C), preserving the stereochemical integrity of the complex steroid skeleton and preventing the formation of degradation impurities that typically arise under harsh drying conditions.
. Because no hydroxide ions are present to combine with protons, no water molecules are formed. This anhydrous environment is critical for maintaining the integrity of the phosphate ester bond, which is susceptible to hydrolysis in the presence of moisture and heat. The absence of water allows the reaction to proceed at mild temperatures (20-30°C), preserving the stereochemical integrity of the complex steroid skeleton and preventing the formation of degradation impurities that typically arise under harsh drying conditions.
Furthermore, the purification mechanism relies on the differential solubility of the target compound in specific mixed organic solvents. The patent specifies a mixture of ethers (e.g., methyl tert-butyl ether) and alkanes (e.g., 1,2-dimethoxyethane). This solvent combination creates a polarity environment where the Betamethasone Phosphate is soluble at elevated temperatures (40-50°C) but precipitates efficiently upon cooling. This thermal gradient crystallization promotes the growth of larger, more ordered granular crystals rather than amorphous powders. Larger crystals possess a lower specific surface area, which inherently reduces their capacity to adsorb atmospheric moisture during downstream processing and storage. This physical modification is as crucial as the chemical purity, as it directly impacts the flowability and compressibility of the bulk drug substance, facilitating easier handling in subsequent formulation stages for injectable preparations.
How to Synthesize Betamethasone Sodium Phosphate Efficiently
Implementing this synthesis route requires precise control over reaction parameters, particularly temperature and solvent composition, to maximize yield and purity. The process begins with the phosphorylation of Betamethasone at cryogenic temperatures to ensure regioselectivity at the 21-hydroxyl position, followed by the critical recrystallization and salt exchange steps described above. Operators must adhere strictly to the specified molar ratios and solvent volumes to achieve the reported high-purity profiles. The following guide outlines the standardized operational procedure derived from the patent examples, ensuring reproducibility and compliance with quality standards.
- Perform phosphorylation of Betamethasone using pyrophosphoryl chloride or phosphorus oxychloride at -40°C to -20°C in THF or acetone.
- Recrystallize the crude phosphate ester in a mixed organic solvent system comprising ethers and alkanes to obtain granular crystals with low moisture.
- React the purified phosphate ester with an organic acid sodium salt (e.g., sodium acetate) in methanol to form the final sodium salt without generating water.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers substantial logistical and economic benefits driven by process simplification and quality enhancement. The elimination of water generation during the salification step fundamentally changes the downstream processing requirements. Traditional methods necessitate extensive drying cycles, often lasting days, to reduce moisture to acceptable levels, which consumes significant energy and ties up production equipment. By contrast, the new method produces a low-moisture product that requires only short-duration drying, drastically reducing energy consumption and increasing equipment turnover rates. This efficiency gain translates directly into lower manufacturing costs and a reduced carbon footprint, aligning with modern green chemistry initiatives and sustainability goals.
- Cost Reduction in Manufacturing: The shift to organic acid sodium salts eliminates the need for expensive and energy-intensive drying protocols associated with removing stoichiometric water. Additionally, the use of cheap and easily obtained raw materials, such as sodium acetate and common organic solvents, reduces the overall bill of materials. The high yield and minimal impurity profile mean less material is lost to waste streams or reprocessing, further optimizing the cost structure for large-scale production of this critical hormone intermediate.
- Enhanced Supply Chain Reliability: The robustness of the crystallization process ensures consistent product quality, reducing the risk of batch failures due to out-of-specification moisture or purity levels. The formation of granular crystals improves the physical handling properties of the material, making it less prone to caking or clumping during storage and transport. This physical stability minimizes losses during logistics and ensures that the material arrives at the formulation site in optimal condition, thereby securing the continuity of supply for finished dosage forms like injectables.
- Scalability and Environmental Compliance: The process operates under mild conditions without the need for hazardous reagents or extreme temperatures, simplifying the engineering controls required for scale-up. The avoidance of water generation reduces the volume of wastewater requiring treatment, easing the burden on environmental management systems. Furthermore, the simplified workflow with fewer purification steps enhances the overall throughput capacity, allowing manufacturers to respond more agilely to market demand fluctuations for this essential anti-inflammatory medication.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this patented synthesis method. These answers are derived directly from the experimental data and technical disclosures within patent CN110964075B, providing clarity on the operational advantages and chemical principles involved. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for commercial manufacturing.
Q: Why is organic acid sodium salt preferred over inorganic bases for salification?
A: Using inorganic bases like sodium hydroxide generates equivalent water during the reaction, leading to high moisture content (10-15%) and hygroscopicity in the final product. Organic acid salts avoid water generation, ensuring low moisture (<2%) and superior stability.
Q: How does the new recrystallization method improve product quality?
A: The patented method uses a specific mixed solvent system of ethers and alkanes. This transforms the product from a powder into granular crystals, significantly reducing related substances to below 0.10% and lowering moisture content to under 2%.
Q: What are the optimal reaction conditions for the phosphorylation step?
A: The phosphorylation reaction should be conducted at low temperatures between -40°C and -20°C using solvents like tetrahydrofuran or acetone. This ensures high conversion rates while minimizing degradation of the steroid backbone.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Betamethasone Sodium Phosphate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the production of life-saving medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex synthetic routes like the one described in CN110964075B can be successfully translated from the laboratory to the plant. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Betamethasone Sodium Phosphate meets the highest international standards for moisture content, related substances, and crystal form.
We invite pharmaceutical partners to collaborate with us to leverage this advanced technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing capabilities can enhance your product quality while reducing overall production costs.
