Advanced Crystallization Technology for Betamethasone Sodium Phosphate: Enhancing Purity and Drying Efficiency
Advanced Crystallization Technology for Betamethasone Sodium Phosphate: Enhancing Purity and Drying Efficiency
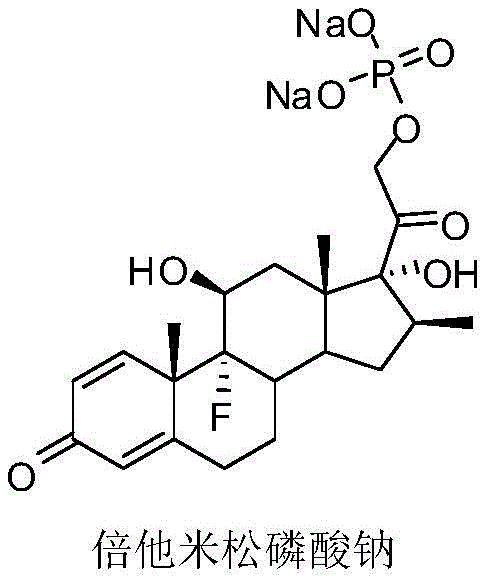
The pharmaceutical industry continuously seeks robust manufacturing processes that can meet the escalating global demand for critical corticosteroids, particularly in the wake of recent health crises that have highlighted the necessity for effective anti-inflammatory agents. Patent CN113773359A introduces a significant technological advancement in the preparation and separation of Betamethasone Sodium Phosphate, a vital water-soluble derivative of glucocorticoid Betamethasone used extensively for immune disease treatment and shock management. This innovation addresses long-standing challenges in the downstream processing of this high-value active pharmaceutical ingredient (API) by replacing traditional ketone-based crystallization solvents with a more efficient ester-based system. The method ensures not only exceptional chemical purity but also superior physical properties such as particle size distribution and moisture content, which are critical parameters for downstream formulation and tableting processes. By optimizing the solvent exchange from alcohol to ester, manufacturers can achieve a product that is significantly easier to dry and stabilize, thereby enhancing the overall reliability of the supply chain for this essential medication.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Betamethasone Sodium Phosphate has relied heavily on acetone as the primary solvent for the final crystallization and isolation steps following the salt formation reaction. While acetone is a common industrial solvent, its application in this specific context presents several detrimental physicochemical drawbacks that complicate large-scale manufacturing. The crystallization of the sodium phosphate salt from acetone tends to produce extremely fine particles with a high specific surface area, which inherently traps significant amounts of solvent and water within the crystal lattice. This phenomenon results in a product with high water content that is notoriously hygroscopic, meaning it readily absorbs moisture from the atmosphere during the separation and drying phases. Consequently, the drying process becomes energy-intensive and time-consuming, often requiring extended periods under vacuum to achieve acceptable residual solvent levels. Furthermore, the fine, powdery nature of the acetone-crystallized material leads to poor flowability, causing operational difficulties in filtration and centrifugation equipment, which can ultimately bottleneck production throughput and increase the risk of batch-to-batch variability.
The Novel Approach
The novel approach detailed in the patent data fundamentally shifts the crystallization paradigm by utilizing ester solvents, such as ethyl acetate or isopropyl acetate, to replace acetone in the pulping and isolation stages. This strategic solvent substitution leverages the specific solubility characteristics of the betamethasone sodium salt to induce a more controlled nucleation and crystal growth environment. When the alcohol solvent is removed and the ester is introduced, the product precipitates or undergoes a phase transformation that favors the formation of larger, more defined crystal structures with significantly lower surface energy. These larger crystals exhibit superior filtration characteristics, allowing for rapid and efficient separation of the mother liquor without the clogging issues associated with fine powders. Moreover, the ester-based crystals demonstrate markedly reduced hygroscopicity, meaning they retain less water and are far less prone to absorbing atmospheric moisture during handling. This results in a final product that is not only easier to dry to constant weight but also possesses enhanced physical stability, ensuring that the critical quality attributes of the API are maintained throughout its shelf life without the need for excessive desiccants or specialized packaging.
Mechanistic Insights into Solvent Exchange Crystallization
The core mechanism driving the success of this process lies in the precise manipulation of solubility parameters and intermolecular forces during the transition from the reaction medium to the crystallization medium. The initial reaction involves the neutralization of betamethasone phosphate with sodium hydroxide in a polar alcohol solvent like methanol, where the ionic character of the disodium phosphate group ensures high solubility. As the methanol is distilled off under reduced pressure, the system moves towards a state of supersaturation, but instead of crashing out immediately in a disordered fashion, the addition of the ester solvent modulates the dielectric constant of the medium. Ethyl acetate, being less polar than methanol but possessing specific hydrogen bond accepting capabilities, interacts with the hydrated sodium ions and the phosphate moiety in a way that slows down the nucleation rate while promoting crystal growth. This kinetic control prevents the rapid formation of micro-crystals and instead allows the molecules to arrange themselves into a more thermodynamically stable lattice structure. The result is a polymorph or crystal habit that minimizes the inclusion of solvent molecules within the lattice, directly addressing the issue of high residual volatiles that plagues the conventional acetone method.
From an impurity control perspective, this solvent exchange mechanism also acts as a powerful purification step that enhances the overall chemical profile of the API. The selective solubility of potential organic impurities in the ester solvent versus the product allows for a 'washing' effect during the pulping stage, where trace byproducts or unreacted starting materials remain in the solution while the pure product crystallizes out. The patent data indicates that this method consistently achieves HPLC purity levels exceeding 99.4%, demonstrating the efficacy of the ester pulping in rejecting impurities that might otherwise co-precipitate in a faster, less controlled acetone crash. Furthermore, the mild temperature conditions employed during the pulping phase, typically between 20°C and 30°C, prevent thermal degradation of the sensitive steroid backbone, preserving the integrity of the 9-alpha-fluoro and 16-beta-methyl substituents that are crucial for the drug's biological activity. This combination of thermodynamic stability and kinetic control ensures that the final product meets the stringent specifications required for parenteral or ophthalmic formulations where particulate matter and chemical purity are paramount.
How to Synthesize Betamethasone Sodium Phosphate Efficiently
The synthesis protocol outlined in the patent provides a clear and reproducible pathway for manufacturing high-quality Betamethasone Sodium Phosphate suitable for commercial production. The process begins with the dissolution of the phosphate precursor in methanol, followed by the controlled addition of sodium hydroxide at low temperatures to effect salt formation. Once the reaction is complete, the methanol is removed, and the critical step of adding the ester solvent is performed to induce crystallization. This sequence is designed to maximize yield while minimizing processing time and energy consumption. For detailed operational parameters, stoichiometry, and safety considerations, please refer to the standardized synthesis guide below which breaks down the procedure into actionable steps for process engineers.
- Dissolve betamethasone phosphate in a low-boiling alcohol solvent like methanol and react with sodium hydroxide at 0-10°C to form the sodium salt.
- Remove the alcohol solvent completely under reduced pressure to prepare the residue for the crystallization step.
- Add an ester solvent such as ethyl acetate to the residue, stir at 20-30°C to induce pulping and crystallization, then filter and dry.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this ester-based crystallization technology offers substantial strategic advantages that extend beyond mere technical performance metrics. The primary value proposition lies in the drastic simplification of the downstream processing workflow, which translates directly into operational cost savings and increased facility throughput. By eliminating the difficulties associated with drying hygroscopic fine powders, manufacturers can significantly reduce the cycle time per batch, allowing for more production runs within the same timeframe without requiring additional capital investment in drying equipment. This efficiency gain is critical in a market where the demand for corticosteroids can fluctuate rapidly, enabling suppliers to respond more agilely to procurement requests and maintain consistent inventory levels. Additionally, the use of common ester solvents like ethyl acetate, which are widely available and often less expensive than specialized ketones in certain regions, contributes to a more stable and predictable raw material cost structure, shielding the supply chain from volatile pricing swings.
- Cost Reduction in Manufacturing: The implementation of this novel crystallization method drives cost reduction in steroid manufacturing through multiple mechanisms that do not rely on speculative percentage savings but rather on tangible process improvements. The elimination of extended drying cycles reduces energy consumption significantly, as vacuum ovens or fluid bed dryers can operate for shorter durations to achieve the same residual solvent specifications. Furthermore, the improved filtration characteristics of the larger crystals reduce the loss of product during the separation phase, effectively increasing the overall process yield and reducing the cost of goods sold per kilogram of API. The ability to recycle the ester solvent efficiently also adds to the economic viability, as solvent recovery rates are typically high, minimizing waste disposal costs and the need for fresh solvent purchases. These cumulative effects create a leaner manufacturing process that is inherently more cost-effective than the traditional acetone-based route.
- Enhanced Supply Chain Reliability: Supply chain reliability is profoundly enhanced by the robustness of this new method, which mitigates the risks associated with processing difficult-to-handle materials. The production of a free-flowing, non-hygroscopic powder simplifies packaging and logistics, reducing the likelihood of product clumping or degradation during transit and storage. This physical stability ensures that the API arrives at the formulation site in optimal condition, reducing the risk of batch rejection due to out-of-specification moisture content or particle size issues. Moreover, the process uses readily available commodity chemicals for solvents, reducing the dependency on niche reagents that might face supply shortages. This accessibility ensures continuity of supply even in disrupted market conditions, providing procurement teams with greater confidence in securing long-term contracts for this critical hormone medicine.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, the process aligns well with modern green chemistry principles and regulatory expectations for pharmaceutical manufacturing. The solvents used, such as ethyl acetate and methanol, are Class 3 solvents with low toxic potential, making them preferable from a regulatory compliance perspective compared to more hazardous alternatives. The ease of solvent recovery reduces the volume of hazardous waste generated, lowering the environmental footprint of the manufacturing site. Scalability is inherently supported by the simplicity of the unit operations involved—dissolution, distillation, and pulping—which are easily transferred from pilot scale to multi-ton commercial production without complex engineering modifications. This facilitates the commercial scale-up of complex corticosteroids, allowing manufacturers to rapidly ramp up production capacity to meet global health demands without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Betamethasone Sodium Phosphate using this advanced methodology. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and quality implications for potential partners and technical stakeholders. Understanding these details is crucial for evaluating the feasibility of integrating this supply source into your existing pharmaceutical development pipelines.
Q: Why is ethyl acetate preferred over acetone for the crystallization of Betamethasone Sodium Phosphate?
A: Ethyl acetate promotes the formation of larger crystal particles with lower water content compared to acetone, which typically yields fine, hygroscopic powders that are difficult to dry and handle.
Q: What are the optimal temperature conditions for the salt formation reaction?
A: The reaction between betamethasone phosphate and sodium hydroxide is best conducted at low temperatures, specifically between 0°C and 10°C, to ensure controlled salt formation and minimize degradation.
Q: How does this process improve the stability of the final API?
A: By utilizing ester solvents for pulping, the resulting product exhibits significantly reduced moisture absorption and improved physical stability, leading to a longer shelf life and easier storage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Betamethasone Sodium Phosphate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates and APIs in the global healthcare ecosystem, and we are uniquely positioned to support your needs with our advanced manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the clinical trial phase or full-scale commercial launch. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Betamethasone Sodium Phosphate meets the highest international standards for safety and efficacy. Our commitment to technical excellence means we can replicate the optimized ester-based crystallization process described in recent patents to deliver a superior product that simplifies your downstream formulation work.
We invite you to engage with our technical procurement team to discuss how our manufacturing solutions can optimize your supply chain and reduce your overall project costs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to our refined production method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your specific project requirements, ensuring a seamless partnership that drives value for your organization.
