Advanced Manufacturing of Elagolix Sodium: A Cost-Effective Route for Global API Supply Chains
The pharmaceutical landscape for endometriosis treatment has been significantly shaped by the approval of Elagolix sodium, commercially known as Orilissa. As the first oral GnRH antagonist approved by the FDA in over a decade, its market potential is immense, with sales projections indicating a robust demand for high-quality active pharmaceutical ingredients. However, the complexity of its molecular architecture has historically posed challenges for manufacturers seeking efficient production methods. Patent CN112679442B introduces a groundbreaking preparation method that addresses these bottlenecks by optimizing the synthetic pathway. This innovation focuses on simplifying the deprotection steps and utilizing greener solvents, thereby enhancing both yield and purity while drastically reducing operational complexity. For industry stakeholders, this represents a pivotal shift towards more sustainable and cost-effective manufacturing paradigms.
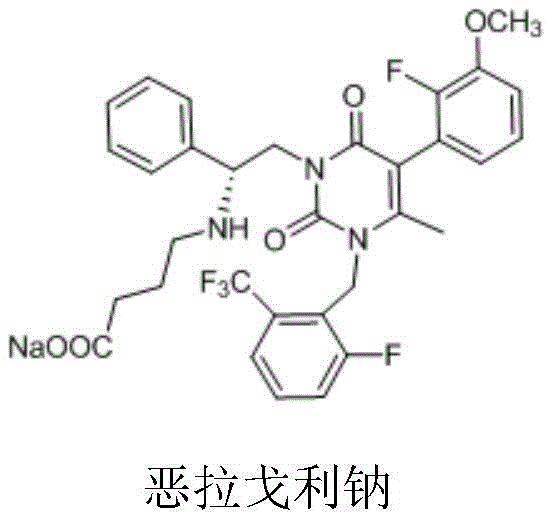
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Elagolix sodium has relied on routes that are fraught with inefficiencies and high operational costs. The original synthetic pathways, such as those described in early patents like US7056927, typically employ expensive and hazardous reagents like trifluoroacetic acid or methanesulfonic acid for the removal of Boc protecting groups. These acidic conditions often lead to the formation of mesylate-based toxic impurities, complicating the purification process and posing significant quality control risks. Furthermore, the de-protected intermediates in these traditional routes frequently manifest as oils or foams with low purity, necessitating cumbersome downstream processing. The introduction of the final ester-butyl fragment often requires organic solvents like acetonitrile or DMF and organic bases, generating substantial amounts of difficult-to-treat wastewater. The final isolation usually involves complex pH adjustments and ion exchange procedures, which are not only labor-intensive but also ill-suited for large-scale industrial application due to high salt waste generation.
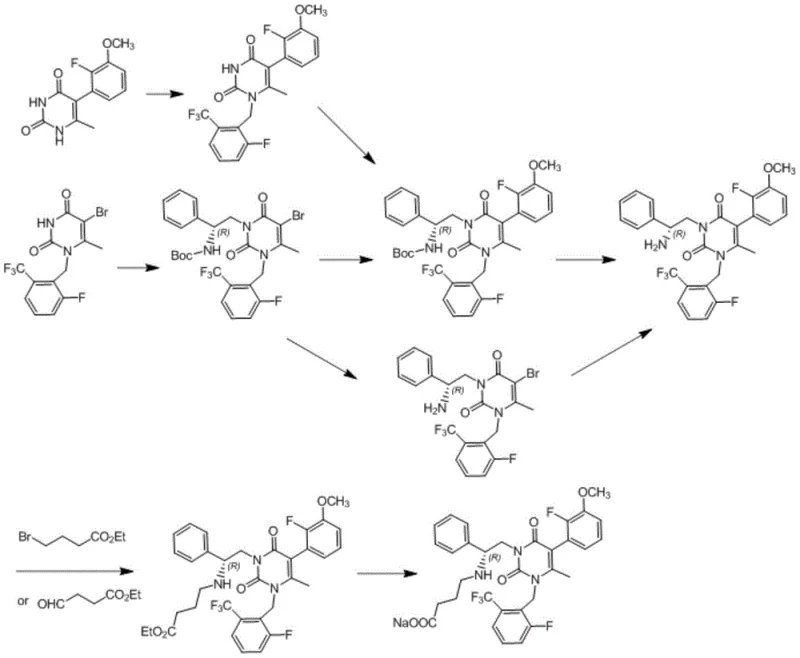
The Novel Approach
In stark contrast, the methodology disclosed in patent CN112679442B offers a streamlined alternative that fundamentally reimagines the synthesis workflow. The core innovation lies in the strategic use of concentrated hydrochloric acid for Boc deprotection, which facilitates the direct precipitation of the intermediate as a stable hydrochloride salt. This simple yet effective modification eliminates the formation of oily byproducts and significantly boosts both yield and purity without the need for complex chromatographic separations. Additionally, the introduction of the critical fragment D is achieved using water as the reaction solvent and inorganic bases as acid-binding agents. This shift from organic to aqueous media not only moderates reaction conditions but also drastically reduces the volume of ammonia nitrogen wastewater. The final product is isolated through a straightforward isopropanol crystallization process, bypassing the need for intricate ion exchange steps. This holistic optimization results in a route that is operationally simple, environmentally benign, and economically superior for commercial manufacturing.
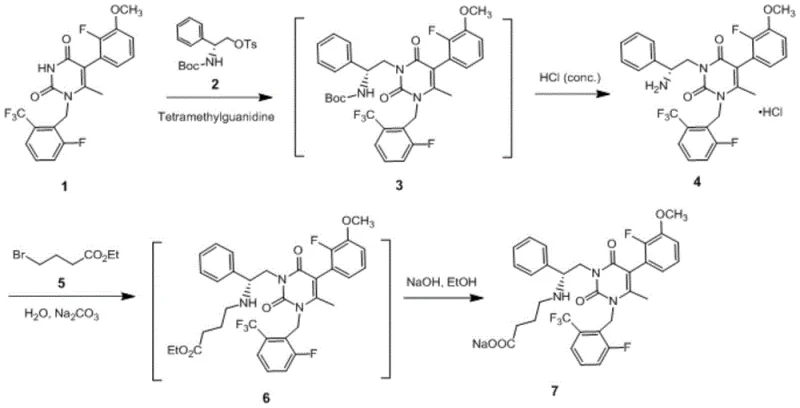
Mechanistic Insights into Alkylation and Deprotection Strategies
The success of this novel synthesis hinges on a deep understanding of the reactivity of the pyrimidine-2,4-dione core and the precise control of protecting group chemistry. The initial alkylation step involves the reaction of the parent nucleus with a protected aminophenylethyl fragment. By utilizing strong non-nucleophilic bases like tetramethylguanidine in polar aprotic solvents, the reaction achieves high conversion rates while minimizing side reactions. The subsequent deprotection mechanism is particularly noteworthy; the use of concentrated hydrochloric acid promotes the cleavage of the tert-butoxycarbonyl group while simultaneously protonating the free amine. This dual action drives the equilibrium towards the formation of the hydrochloride salt, which possesses low solubility in the reaction medium, thus precipitating out of the solution. This phenomenon effectively purifies the intermediate in situ, preventing the accumulation of degradation products that typically plague acid-mediated deprotections in organic phases.
Furthermore, the mechanism for introducing the butyl ester fragment demonstrates a sophisticated application of phase-transfer principles in an aqueous environment. The use of inorganic carbonates in water creates a heterogeneous system where the organic substrate reacts efficiently at the interface or within micellar structures formed by the surfactant-like properties of the intermediate. This approach mitigates the risk of hydrolysis of the ester group during the alkylation phase, a common pitfall in aqueous alkylations. The controlled hydrolysis in the final step, using sodium hydroxide in an alcohol-water mixture, ensures complete saponification of the ethyl ester to the carboxylic acid salt without affecting the sensitive fluorinated aromatic rings. This precise orchestration of reaction conditions ensures a clean impurity profile, which is critical for meeting the stringent regulatory standards required for API production.
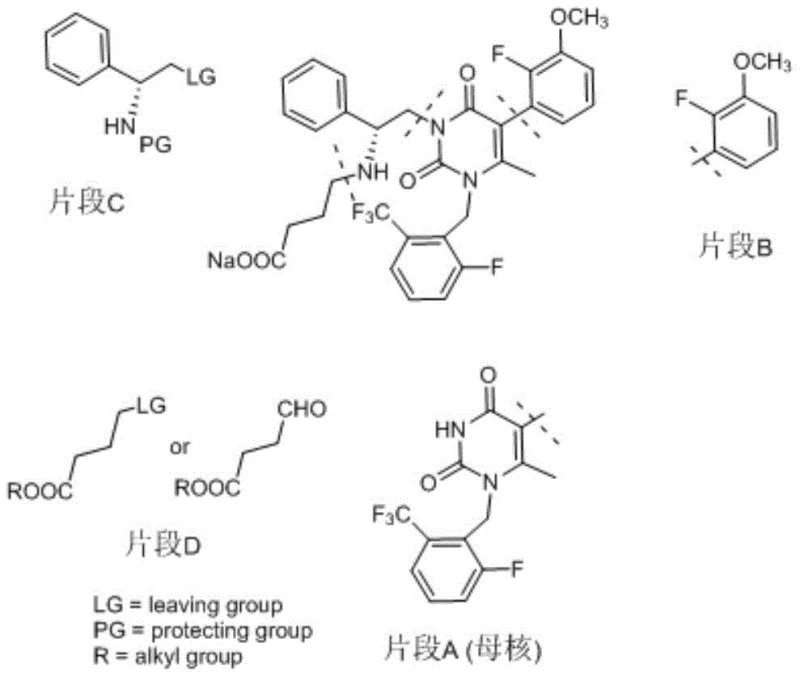
How to Synthesize Elagolix Sodium Efficiently
The optimized protocol outlined in the patent provides a clear roadmap for executing this synthesis with high fidelity. It begins with the coupling of the pyrimidine core and the chiral amine fragment, followed by the critical acid-mediated deprotection that yields a solid intermediate. The process concludes with the aqueous alkylation and final hydrolysis steps. This sequence is designed to maximize throughput while minimizing the need for specialized equipment or hazardous waste disposal. For technical teams looking to implement this process, the detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety.
- Alkylation of the pyrimidine dione core with a protected phenylethyl amine fragment using tetramethylguanidine.
- Deprotection of the Boc group using concentrated hydrochloric acid to form a stable hydrochloride salt intermediate.
- Final alkylation with ethyl 4-bromobutyrate in water using inorganic base, followed by hydrolysis and crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route translates into tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the substantial reduction of raw material costs associated with replacing expensive specialty acids like trifluoroacetic acid with commodity chemicals like concentrated hydrochloric acid. Moreover, the elimination of complex ion exchange resins and the reduction in organic solvent usage directly lower the variable costs of production. From a supply chain reliability perspective, the simplified workflow reduces the number of unit operations, thereby decreasing the potential for batch failures and shortening the overall manufacturing cycle time. This enhanced robustness ensures a more consistent supply of high-purity intermediates, mitigating the risk of stockouts for downstream API manufacturers.
- Cost Reduction in Manufacturing: The replacement of costly deprotecting agents and organic bases with inexpensive inorganic alternatives significantly lowers the bill of materials. Additionally, the ability to isolate intermediates via simple filtration rather than complex extraction or chromatography reduces labor and energy consumption. The reduction in solvent volume and the switch to water for key steps further diminishes waste disposal costs, contributing to a leaner and more profitable manufacturing model.
- Enhanced Supply Chain Reliability: The use of readily available reagents such as sodium carbonate and hydrochloric acid ensures that the supply chain is not vulnerable to shortages of exotic chemicals. The simplified process flow, characterized by fewer steps and easier workups, enhances the overall throughput capacity of the facility. This operational efficiency allows for faster response times to market demand fluctuations, ensuring that customers receive their orders without delay.
- Scalability and Environmental Compliance: The green chemistry principles embedded in this route, particularly the use of water as a solvent and the generation of less hazardous waste, facilitate easier regulatory compliance. The process is inherently scalable, as the exothermic nature of the reactions is easily managed in larger reactors, and the crystallization steps are robust enough for ton-scale production. This alignment with environmental, social, and governance (ESG) goals makes the supply chain more resilient against tightening environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. They are derived from the specific technical disclosures and beneficial effects highlighted in the patent documentation, providing clarity on how this route compares to existing technologies.
Q: How does the new method improve intermediate purity compared to traditional routes?
A: By using concentrated hydrochloric acid for deprotection, the intermediate precipitates directly as a solid hydrochloride salt, avoiding the formation of oily or foamy low-purity products common with trifluoroacetic acid methods.
Q: What are the environmental benefits of this synthesis route?
A: The process utilizes water as a solvent for the introduction of the ester-butyl fragment and employs inorganic bases, significantly reducing ammonia nitrogen wastewater and organic solvent consumption compared to DMF or acetonitrile-based methods.
Q: Is this route suitable for large-scale commercial production?
A: Yes, the method simplifies operations by eliminating complex ion exchange steps and uses cost-effective reagents like concentrated HCl and sodium carbonate, making it highly scalable and economically viable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Elagolix Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists has thoroughly analyzed the route disclosed in CN112679442B and is fully prepared to translate this laboratory-scale innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of Elagolix sodium intermediate we produce adheres to the highest international standards.
We invite potential partners to engage with our technical procurement team to discuss how this optimized route can benefit your specific supply chain needs. By leveraging our expertise, you can access a Customized Cost-Saving Analysis tailored to your production volumes. We encourage you to request specific COA data and route feasibility assessments to verify the superior quality and economic advantages of our manufacturing process. Let us collaborate to drive efficiency and innovation in the production of this vital therapeutic agent.
