Revolutionizing Meropenem Production: A Safe and Scalable Zinc Powder Deprotection Strategy
Introduction to Patent CN102348710A
The pharmaceutical industry continuously seeks robust manufacturing routes for critical antibiotics, and patent CN102348710A presents a transformative approach to synthesizing meropenem trihydrate, a premier carbapenem antibiotic. This intellectual property details an improved preparation method that fundamentally shifts the deprotection paradigm from traditional catalytic hydrogenation to a chemically driven reduction using zinc powder. By replacing hazardous high-pressure hydrogenation with a mild, atmospheric zinc-mediated process, this technology addresses long-standing safety and scalability bottlenecks inherent in carbapenem production. For global supply chain leaders and R&D directors, this patent represents a viable pathway to enhance production continuity while mitigating the risks associated with explosive gases and expensive noble metal catalysts. The innovation lies not just in the reagent swap, but in the holistic optimization of reaction conditions, including precise pH control via phosphate buffers to preserve the fragile beta-lactam ring integrity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of meropenem has relied heavily on catalytic hydrogenation using palladium on carbon (Pd/C) to remove the p-nitrobenzyl protecting group, a step that introduces significant operational complexity and risk. As illustrated in the prior art reaction schemes, this conventional route necessitates the use of high-pressure hydrogen gas, which poses severe explosion hazards and requires specialized, costly pressure-rated reactor vessels that are difficult to scale safely. Furthermore, the reliance on palladium, a precious metal with volatile market pricing, creates unpredictable cost structures for procurement managers, while the removal of trace metal residues demands additional purification steps that can erode overall yield. The conventional process often involves complex workups, including column chromatography and freeze-drying, which are time-consuming and energy-intensive, ultimately limiting the throughput capacity for high-purity pharmaceutical intermediates required by the global market.
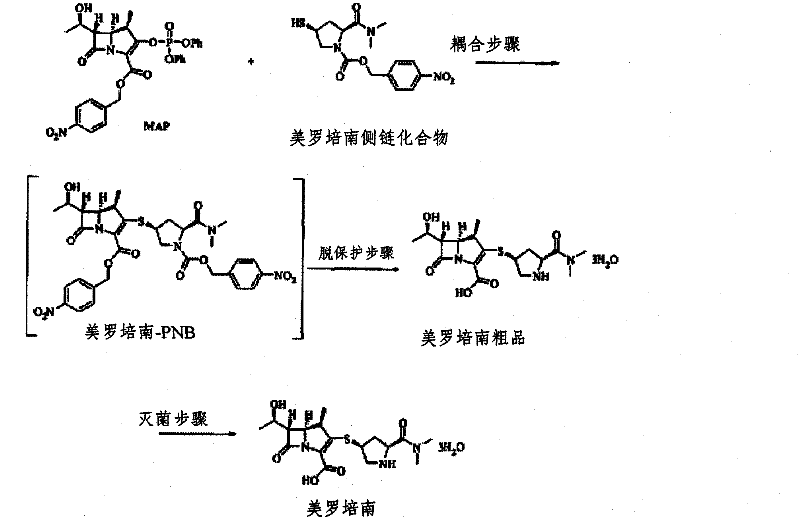
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent utilizes zinc powder as a reducing agent under mild conditions, effectively circumventing the dangers of high-pressure hydrogenation while drastically simplifying the equipment requirements. This zinc-mediated deprotection operates efficiently at ambient pressure and moderate temperatures ranging from 20°C to 50°C, allowing for the use of standard glass-lined or stainless steel reactors that are readily available in most fine chemical facilities. The substitution of expensive palladium catalysts with economical zinc powder results in a direct material cost reduction, while the absence of heavy metal contamination simplifies the downstream purification workflow. By enabling the reaction to proceed in a mixed solvent system of organic solvents and aqueous phosphate buffers, this approach ensures excellent solubility of the intermediate while maintaining the chemical stability of the sensitive carbapenem core, leading to a cleaner reaction profile and higher purity crude product.
Mechanistic Insights into Zinc-Mediated Reductive Deprotection
The core chemical transformation involves the reductive cleavage of the p-nitrobenzyl ester moiety from the meropenem-PNB intermediate, a process driven by the electron transfer capabilities of activated zinc powder in a buffered environment. The presence of a phosphate buffer, specifically potassium dihydrogen phosphate (KH2PO4), is critical not merely as a solvent component but as a pH regulator that prevents the acid- or base-catalyzed hydrolysis of the beta-lactam ring, which is highly susceptible to degradation under extreme pH conditions. During the reaction, zinc acts as the electron donor, reducing the nitro group and facilitating the fragmentation of the protecting group without generating harsh acidic byproducts that could compromise the stereochemical integrity of the molecule. This mechanistic precision ensures that the chiral centers at positions 4, 5, and 6 of the carbapenem nucleus remain intact, preserving the biological activity of the final antibiotic substance.
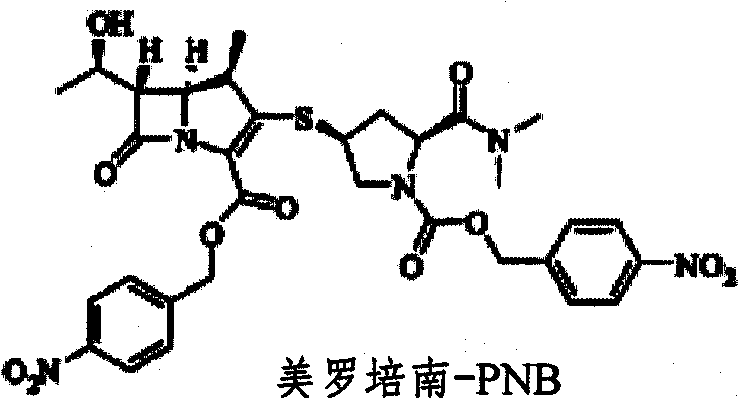
Impurity control is another pivotal aspect of this mechanism, particularly regarding the management of phosphate salts generated during the reaction and workup phases. The patent describes a sophisticated purification strategy where phosphate removal is achieved through a combination of crystallization and ion-exchange chromatography, ensuring that the final API meets stringent regulatory limits for inorganic residues. By carefully controlling the temperature during methanol addition, bulk phosphate salts can be precipitated and filtered off before the solution undergoes secondary polishing through cationic resins like BCMB50. This multi-stage purification logic effectively separates the highly polar inorganic impurities from the organic meropenem molecule, resulting in a product with a superior impurity profile compared to methods that rely solely on extraction or single-step crystallization.
How to Synthesize Meropenem Trihydrate Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for transitioning from the protected intermediate to the final active pharmaceutical ingredient with high efficiency and reproducibility. The process begins with the coupling of the MAP precursor with the specific pyrrolidine side chain to generate the meropenem-PNB intermediate, which serves as the substrate for the subsequent zinc-mediated reduction. Following the coupling, the reaction mixture is subjected to the deprotection conditions using zinc powder and phosphate buffer, followed by a rigorous purification sequence involving solvent exchange, resin treatment, and controlled crystallization. For detailed operational parameters, stoichiometry, and specific temperature ramps required to replicate this high-yield process, please refer to the standardized synthesis guide below.
- Perform coupling reaction between MAP and the side chain compound to form Meropenem-PNB intermediate.
- Execute reductive deprotection using zinc powder in a phosphate buffer solution at mild temperatures (20-50°C).
- Purify the crude product through phosphate removal via crystallization or ion-exchange resin, followed by final crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this zinc-based synthesis route offers compelling strategic advantages that extend beyond simple chemical efficiency to impact the bottom line and operational resilience. The elimination of high-pressure hydrogenation equipment reduces capital expenditure requirements and lowers the barrier for contract manufacturing organizations to produce this key antibiotic intermediate at scale. Furthermore, the shift away from noble metal catalysts insulates the production cost from the volatility of the palladium market, providing more predictable budgeting for long-term supply agreements. The inherent safety of operating at atmospheric pressure also reduces insurance premiums and regulatory compliance burdens associated with handling explosive gases, thereby smoothing the path for facility audits and approvals.
- Cost Reduction in Manufacturing: The replacement of expensive palladium on carbon catalysts with commodity-grade zinc powder delivers a substantial reduction in raw material costs, directly improving the gross margin of the manufacturing process. Additionally, the ability to use standard reaction vessels instead of specialized high-pressure autoclaves lowers depreciation costs and maintenance expenses, while the simplified workup procedure reduces solvent consumption and waste disposal fees. These cumulative savings allow for a more competitive pricing structure in the global market for carbapenem intermediates without compromising on quality standards.
- Enhanced Supply Chain Reliability: By removing the dependency on high-pressure hydrogen gas, the manufacturing process becomes less vulnerable to utility disruptions and safety shutdowns, ensuring a more consistent supply of critical antibiotic ingredients. The use of widely available zinc powder and common phosphate salts mitigates the risk of raw material shortages that can occur with specialized catalysts, enhancing the overall robustness of the supply chain. This reliability is crucial for meeting the demanding delivery schedules of multinational pharmaceutical companies that require uninterrupted access to high-quality API intermediates.
- Scalability and Environmental Compliance: The mild reaction conditions and aqueous workup components of this process facilitate easier scale-up from pilot plant to commercial production volumes, minimizing the technical risks typically associated with process transfer. The reduced generation of heavy metal waste aligns with increasingly stringent environmental regulations, simplifying the permitting process and reducing the ecological footprint of the manufacturing site. This environmental compatibility supports sustainable manufacturing goals and enhances the corporate social responsibility profile of the supply chain partners involved.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this zinc-mediated deprotection technology in industrial settings. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a factual basis for evaluating the feasibility of this route. Understanding these nuances is essential for technical teams assessing the integration of this method into existing production lines.
Q: Why is zinc powder preferred over palladium carbon for meropenem deprotection?
A: Zinc powder eliminates the need for high-pressure hydrogen gas and expensive noble metal catalysts, significantly reducing safety risks and operational costs while maintaining high yield.
Q: How does the new method handle phosphate impurities?
A: The process utilizes a dual-removal strategy involving initial crystallization to precipitate bulk phosphates, followed by secondary purification using cationic or adsorption resins to ensure ultra-low impurity levels.
Q: Is the quality of zinc-reduced meropenem comparable to USP standards?
A: Yes, NMR spectral analysis confirms that the target compound synthesized via this zinc-mediated route is structurally identical to the United States Pharmacopoeia (USP) reference standard.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Meropenem Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies like the zinc powder deprotection route to maintain competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex chemical transformations are executed with precision and consistency. We are committed to delivering high-purity meropenem intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging our expertise in process optimization, we can help you navigate the transition to safer and more cost-effective manufacturing routes.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis method can be tailored to your specific supply chain needs. Contact us today to request a Customized Cost-Saving Analysis and obtain specific COA data and route feasibility assessments for your next project. Together, we can build a resilient and efficient supply chain for next-generation antibiotics.
