Revolutionizing Meropenem Intermediate Production: A Technical Analysis of Patent CN101410399A
The pharmaceutical industry constantly seeks more efficient pathways for producing critical antibiotic intermediates, and patent CN101410399A presents a significant advancement in the preparation of meropenem precursors. This intellectual property details a robust process for producing (4R,5S,6S)-3-[[(3S,5S)-1-(p-nitrobenzyloxycarbonyl)-5-(dimethylaminocarbonyl)-3-pyrrolidinyl]thio]-6-[(1R)-1-hydroxyethyl]-4-methyl-7-oxo-1-azabicyclo[3.2.0]hept-2-ene-2-carboxylate p-nitrobenzyl ester, a pivotal synthetic intermediate for the broad-spectrum carbapenem antibiotic meropenem. The core innovation lies not just in the chemical synthesis but specifically in the isolation technique, which overcomes historical bottlenecks related to purification time and physical form. By optimizing the crystallization conditions through a specific solvent mixing protocol, the inventors have achieved a method that allows for rapid separation of the target compound in a solid form that is ideally suited for downstream processing. This technical breakthrough addresses the urgent need for reliable meropenem intermediate suppliers who can deliver high-purity materials with consistent quality attributes essential for GMP manufacturing environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods for isolating this complex beta-lactam intermediate have historically suffered from significant inefficiencies that hinder large-scale production. As noted in the background of the patent, previous techniques often relied on crystallization from single solvents like alkyl alkanoates, which frequently resulted in the compound separating as an intractable oily substance rather than a filterable solid. Even when solids were obtained, the process required excessively long stirring times, often exceeding 72 hours at ambient temperatures, to achieve acceptable recovery rates. This prolonged processing time not only ties up reactor capacity and increases energy consumption but also exposes the sensitive beta-lactam ring to potential degradation over extended periods. Furthermore, the formation of oils necessitates additional, costly purification steps such as silica gel chromatography to remove impurities, which is economically unviable and operationally cumbersome on an industrial scale. These limitations create a bottleneck in the supply chain, making it difficult to ensure the continuous availability of high-purity pharmaceutical intermediates required for global antibiotic production.
The Novel Approach
The methodology disclosed in CN101410399A fundamentally re-engineers the isolation step by utilizing a controlled mixture of a good solvent and a poor solvent to induce rapid precipitation. Instead of relying on slow evaporation or cooling from a single solvent, the process involves mixing a solution of the carboxylic ester in a good solvent (such as ethyl acetate) with a specific amount of a poor solvent (such as n-hexane) under strictly controlled temperature conditions. Crucially, the amount of poor solvent is calibrated so that the compound does not precipitate as an oil but rather forms a solid with a crystallinity of less than 99%. This specific physical form, often containing amorphous regions, is surprisingly advantageous because it exhibits excellent solubility and dispersibility in the solvents used for the subsequent conversion to meropenem. 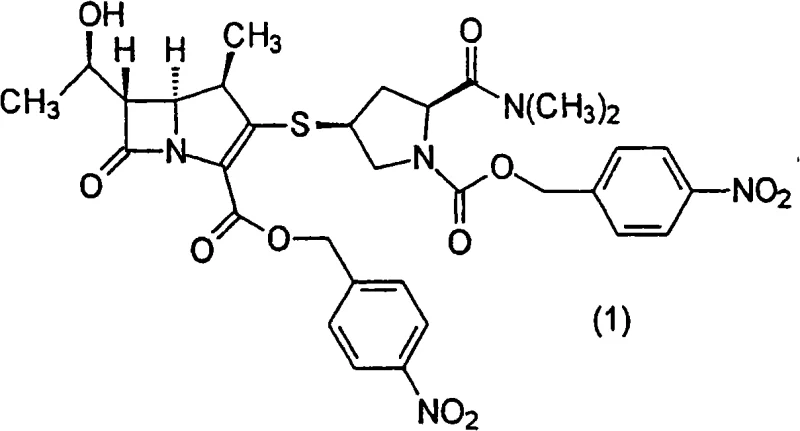
Mechanistic Insights into Thioether Coupling and Crystallization Control
The synthesis of the target molecule involves a nucleophilic substitution reaction where a thiol group on a pyrrolidine ring attacks a leaving group on the beta-lactam core. Specifically, the process reacts a phosphonate ester derivative (Compound 2) with a thiol-containing pyrrolidine derivative (Compound 3) in the presence of a base. The reaction mechanism proceeds via the deprotonation of the thiol to form a thiolate anion, which then performs a nucleophilic attack on the carbon bearing the phosphonate leaving group. This S-alkylation step is critical for establishing the C-S bond that links the two major fragments of the meropenem molecule. The choice of base, typically an organic amine like triethylamine or DIPEA, and the solvent system, often acetonitrile or DMF, are optimized to maximize the reaction rate while minimizing side reactions such as beta-lactam ring opening. The reaction is typically conducted at low temperatures, ranging from -40°C to 20°C, to preserve the stereochemical integrity of the chiral centers at the 4, 5, and 6 positions of the bicyclic system.
Following the coupling reaction, the isolation mechanism becomes the defining feature of this technology. The patent describes a phenomenon where the physical state of the precipitate is governed by the degree of supersaturation and the rate of nucleation. By adding the poor solvent (anti-solvent) gradually to the good solvent solution at temperatures between -20°C and 0°C, the system is driven into a metastable zone where nucleation occurs rapidly without the growth of large, highly ordered crystals that might trap impurities or form oils. The resulting solid has a crystallinity of less than 99%, which is intentionally targeted. Highly crystalline materials can sometimes be too stable and dissolve too slowly in the next reaction step, whereas this semi-amorphous solid dissolves readily, facilitating the rapid removal of the p-nitrobenzyl protecting groups in the final stages of meropenem synthesis. 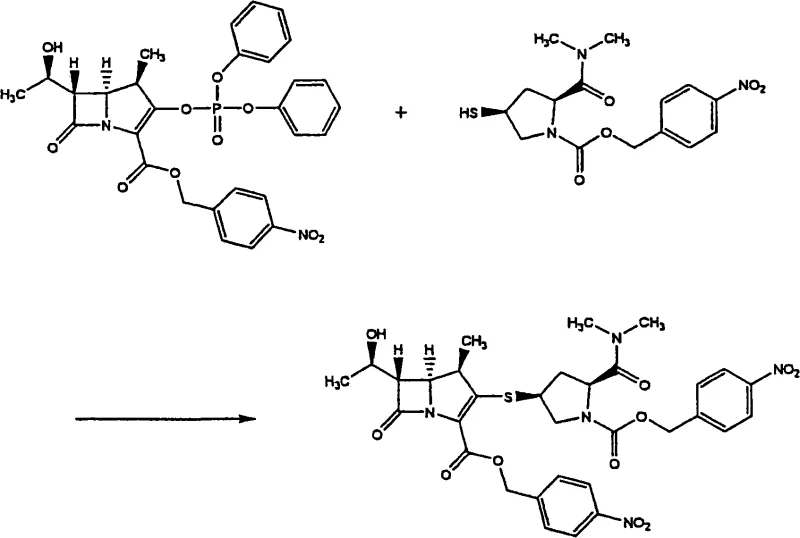
How to Synthesize Meropenem Intermediate Efficiently
The practical implementation of this synthesis route requires careful attention to solvent ratios and temperature control to replicate the benefits seen in the patent examples. The process begins with the preparation of a water-saturated good solvent solution of the crude product, typically at a concentration of 10 to 25 wt%. This solution is then cooled to sub-zero temperatures, and the poor solvent is added dropwise or in portions while maintaining vigorous stirring to ensure homogeneity and prevent local over-concentration that could lead to oiling out. The detailed standardized synthesis steps, including specific molar ratios and workup procedures, are outlined in the guide below for technical teams looking to adopt this methodology.
- React the phosphonate ester precursor with the thiol pyrrolidine derivative in an organic solvent like acetonitrile under basic conditions at low temperature.
- Prepare a water-saturated good solvent solution (e.g., ethyl acetate) containing the crude product at a concentration of 10-25 wt%.
- Add a poor solvent (e.g., n-hexane) gradually at -20 to 0°C to induce rapid precipitation of the solid intermediate without oil formation, followed by filtration.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this crystallization technology translates directly into tangible operational improvements and cost efficiencies. The primary advantage is the drastic simplification of the purification workflow, which eliminates the need for expensive and time-consuming column chromatography on silica gel. In traditional fine chemical manufacturing, chromatography is a major cost driver due to the high price of stationary phases, the large volumes of solvents required for elution, and the significant labor involved in packing and running columns. By replacing this with a simple filtration and drying step, the manufacturing cost per kilogram is significantly reduced, allowing for more competitive pricing of the final API. Furthermore, the reduction in processing time from days to hours means that reactor turnover is much faster, effectively increasing the production capacity of existing facilities without the need for capital investment in new hardware.
- Cost Reduction in Manufacturing: The elimination of chromatographic purification steps results in substantial savings on consumables and solvent usage. Since the process relies on simple solvent mixing and filtration, the consumption of high-purity silica gel and the associated waste disposal costs are completely removed from the cost structure. Additionally, the shorter cycle time reduces utility costs related to heating, cooling, and agitation over extended periods. This lean manufacturing approach ensures that cost reduction in carbapenem antibiotic manufacturing is achieved through process intensification rather than compromising on raw material quality.
- Enhanced Supply Chain Reliability: The robustness of the isolation method significantly mitigates the risk of batch failures due to oil formation, a common issue in beta-lactam chemistry. When a batch turns into an oil, it often requires reprocessing or scrapping, leading to unpredictable delays in delivery. By guaranteeing the formation of a filterable solid, this method ensures consistent batch-to-batch performance and reliable lead times for high-purity pharmaceutical intermediates. This predictability is crucial for pharmaceutical companies managing tight inventory schedules and regulatory filing timelines, as it secures the continuity of supply for critical antibiotic production lines.
- Scalability and Environmental Compliance: The process is inherently scalable because it avoids unit operations that are difficult to enlarge, such as flash chromatography. Filtration and solvent exchange are standard unit operations that translate seamlessly from pilot plant to multi-ton commercial production. Moreover, the use of common solvents like ethyl acetate and n-hexane, which are easily recovered and recycled, aligns with modern green chemistry principles. The reduction in solvent waste volume and the avoidance of silica waste contribute to a lower environmental footprint, helping manufacturers meet increasingly stringent environmental regulations and sustainability goals without sacrificing throughput.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this meropenem intermediate. These answers are derived directly from the technical specifications and experimental data provided in the patent literature, ensuring accuracy for R&D and quality assurance teams evaluating this supply source.
Q: Why is low crystallinity (<99%) preferred for this meropenem intermediate?
A: According to patent CN101410399A, a solid with crystallinity below 99% (often containing amorphous regions) exhibits superior solubility and dispersibility in solvents compared to highly crystalline forms. This physical property facilitates the subsequent deprotection and conversion reactions required to synthesize the final meropenem API, ensuring faster reaction kinetics and higher overall yield.
Q: How does this method improve upon traditional isolation techniques?
A: Traditional methods often require prolonged stirring times exceeding 72 hours and risk the formation of intractable oily substances that are difficult to filter and purify. The novel approach described utilizes a specific good solvent/poor solvent mixing strategy that induces rapid solid precipitation within hours, eliminating the need for time-consuming chromatography and significantly simplifying the industrial workflow.
Q: What are the critical solvent parameters for successful isolation?
A: The process relies on a precise balance between a 'good solvent' (such as ethyl acetate or dichloromethane) where the compound is soluble, and a 'poor solvent' (such as n-hexane or n-heptane) where it is not. The key is adding the poor solvent in an amount that creates supersaturation without causing the compound to separate as an oil, typically maintaining the mixture temperature between -20°C and 0°C during the addition phase.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Meropenem Intermediate Supplier
At NINGBO INNO PHARMCHEM, we understand the critical role that high-quality intermediates play in the global supply of life-saving antibiotics. Our technical team has extensively analyzed the process described in CN101410399A and possesses the expertise to implement this advanced crystallization technology at scale. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to industrial manufacturing is seamless. Our facility is equipped with rigorous QC labs and stringent purity specifications to guarantee that every batch of meropenem intermediate meets the exacting standards required for GMP API synthesis, providing our partners with total confidence in material quality.
We invite procurement leaders and technical directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized process capabilities, we can help you reduce the total cost of ownership for your carbapenem supply chain. Please contact our technical procurement team to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation can support your strategic sourcing goals.
