Scalable Enzymatic Synthesis of High-Purity Esomeprazole Intermediates for Global API Production
Scalable Enzymatic Synthesis of High-Purity Esomeprazole Intermediates for Global API Production
The pharmaceutical industry is currently witnessing a paradigm shift towards biocatalytic processes for the synthesis of complex chiral intermediates, driven by the urgent need for greener chemistry and higher stereochemical purity. Patent CN108690836B represents a significant technological breakthrough in this domain, specifically addressing the synthesis of Esomeprazole, the S-isomer of Omeprazole, which is a critical Proton Pump Inhibitor (PPI) used globally for treating gastric disorders. This patent discloses a novel cyclohexanone monooxygenase obtained through precise site-specific mutagenesis, capable of catalyzing the asymmetric oxidation of omeprazole thioether intermediates with exceptional efficiency. Unlike traditional chemical methods that struggle with low yields and environmental hazards, this enzymatic approach leverages genetic engineering to create biocatalysts that operate under mild conditions while tolerating high substrate concentrations. For R&D directors and process chemists, this technology offers a compelling alternative to resolution-based methods, promising a streamlined pathway to high-purity active pharmaceutical ingredients (APIs) that meets stringent regulatory standards for impurity profiles and residual solvents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
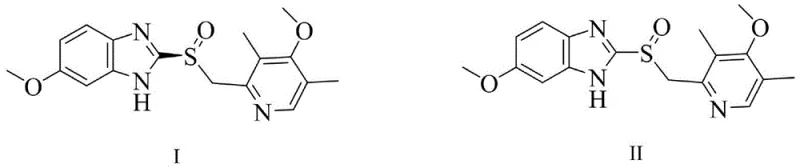
Historically, the industrial production of esomeprazole has relied heavily on chemical oxidation followed by chiral resolution or asymmetric synthesis using transition metal catalysts. These conventional routes are fraught with significant technical and economic drawbacks that hinder efficient manufacturing. Chemical oxidation often results in a racemic mixture, necessitating a resolution step that theoretically limits the maximum yield to 50%, thereby doubling the material cost and waste generation for the discarded R-isomer. Furthermore, the use of heavy metal oxidants introduces severe contamination risks, requiring complex and costly purification steps to remove trace metals to levels acceptable for pharmaceutical use. The harsh reaction conditions typically associated with these chemical processes, such as extreme pH levels or high temperatures, can also lead to the degradation of the sensitive benzimidazole and pyridine moieties, generating difficult-to-remove impurities that compromise the safety profile of the final drug product. Consequently, manufacturers face substantial challenges in achieving consistent quality while maintaining cost-effectiveness and environmental compliance.
The Novel Approach
In stark contrast, the technology disclosed in CN108690836B utilizes a genetically engineered cyclohexanone monooxygenase to achieve direct asymmetric sulfoxidation. This novel approach bypasses the need for chiral resolution by selectively producing the desired S-enantiomer directly from the prochiral sulfide precursor. The core innovation lies in the specific mutation of the enzyme's amino acid sequence, particularly the substitution of serine at position 435 with threonine, which significantly enhances the enzyme's catalytic activity and stability. This engineered biocatalyst demonstrates a remarkable ability to function effectively even at high substrate concentrations, a critical factor for industrial viability that previous enzymatic methods failed to address adequately. By operating in aqueous buffer systems with mild organic co-solvents, this method drastically reduces the reliance on hazardous organic solvents and eliminates the risk of heavy metal contamination. The result is a cleaner reaction profile with higher atom economy, positioning this biocatalytic route as a superior choice for modern cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Site-Directed Mutagenesis and Biocatalysis
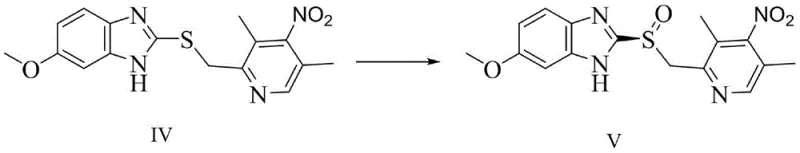
The success of this synthetic route hinges on the precise structural modifications made to the cyclohexanone monooxygenase enzyme through site-directed mutagenesis. The patent details a systematic engineering strategy where specific amino acid residues within the enzyme's active pocket are altered to optimize substrate binding and oxygen transfer efficiency. For instance, the mutation of Serine to Threonine at position 435 likely introduces a methyl group that alters the hydrophobicity and steric environment of the active site, facilitating better accommodation of the bulky omeprazole thioether molecule. This structural tuning enhances the enzyme's affinity for the substrate, allowing for higher turnover numbers and improved enantioselectivity towards the S-configuration. The catalytic mechanism involves the activation of molecular oxygen by the flavin cofactor within the enzyme, followed by the nucleophilic attack on the sulfur atom of the thioether. The chiral environment of the mutated protein pocket ensures that oxygen is delivered from a specific face of the sulfur atom, thereby enforcing the formation of the single enantiomer required for esomeprazole. This level of control is difficult to achieve with small-molecule catalysts and underscores the power of protein engineering in fine chemical synthesis.
Furthermore, the impurity control mechanism inherent in this enzymatic process is a major advantage for quality assurance teams. Because the enzyme is highly specific, side reactions such as over-oxidation to the sulfone or oxidation at other sites on the molecule are minimized. The patent data indicates that the resulting esomeprazole intermediate (V) possesses high optical purity (ee values exceeding 99% in optimized examples), which significantly simplifies the downstream purification process. In traditional chemical synthesis, removing closely related structural impurities often requires multiple recrystallizations or preparative chromatography, which drives up costs and reduces overall yield. With the enzymatic route, the primary impurity burden is shifted from chemical byproducts to biological residues like proteins, which are generally easier to remove via standard filtration and extraction techniques. This inherent selectivity not only ensures a high-purity pharmaceutical intermediate but also enhances the robustness of the manufacturing process against batch-to-batch variability.
How to Synthesize Esomeprazole Intermediate Efficiently
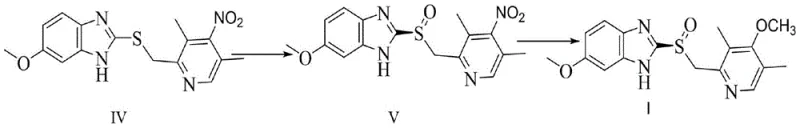
The implementation of this biocatalytic process involves a well-defined sequence of steps starting from strain construction to the final catalytic reaction. The patent outlines a comprehensive protocol for creating the recombinant E. coli strains, including PCR amplification of the mutated gene, ligation into expression vectors like pET28a, and transformation into host cells. Once the strain is established, the focus shifts to fermentation optimization, where parameters such as pH, dissolved oxygen, and induction timing are tightly controlled to maximize enzyme expression. The actual conversion step is performed in a biphasic or co-solvent system to ensure substrate solubility while maintaining enzyme activity. Detailed standardized synthesis steps see the guide below.
- Construct recombinant E. coli strains carrying the mutated cyclohexanone monooxygenase gene (e.g., SEQ ID NO: 3) via site-directed mutagenesis and plasmid transformation.
- Perform high-density fermentation of the engineered strain in LB or defined medium, inducing expression with IPTG at optimal OD600 values to maximize enzyme yield.
- Conduct the biocatalytic oxidation of omeprazole thioether (IV) in a phosphate buffer system with organic co-solvents, maintaining pH 6.5-8.5 and temperatures between 10-35°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this enzymatic technology translates into tangible strategic benefits beyond mere technical superiority. The shift from chemical resolution to direct enzymatic synthesis fundamentally alters the cost structure of esomeprazole production by eliminating the 50% yield loss associated with discarding the unwanted enantiomer. This theoretical doubling of yield potential from the same amount of starting material represents a massive opportunity for cost reduction in API manufacturing. Additionally, the mild reaction conditions reduce energy consumption and the need for specialized corrosion-resistant equipment, further lowering capital and operational expenditures. The ability to run reactions at higher substrate concentrations means that reactor volumes can be utilized more efficiently, increasing throughput without proportional increases in facility footprint. These factors combined create a more resilient and cost-effective supply chain capable of meeting global demand fluctuations.
- Cost Reduction in Manufacturing: The elimination of chiral resolving agents and the reduction in solvent usage significantly lower the raw material costs per kilogram of product. Since the enzyme catalyzes the formation of the desired S-enantiomer directly, there is no need to purchase or synthesize chiral auxiliaries, which are often expensive and supply-constrained. Furthermore, the simplified purification train reduces the consumption of chromatography media and crystallization solvents, leading to substantial savings in waste disposal and utility costs. The overall process efficiency is enhanced by the high turnover rate of the engineered enzyme, allowing for shorter cycle times and better asset utilization.
- Enhanced Supply Chain Reliability: Relying on fermentation-based production mitigates the risks associated with the supply of rare earth metals or specialized chemical catalysts that are subject to geopolitical volatility. The raw materials for fermentation, such as glucose and yeast extract, are commodity chemicals with stable and abundant global supplies. This ensures a consistent and reliable source of the biocatalyst, reducing the risk of production stoppages due to raw material shortages. Moreover, the robustness of the engineered strains allows for flexible manufacturing schedules, enabling producers to scale up or down rapidly in response to market demand without compromising product quality or delivery timelines.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, having been validated in both shake flask and fermenter environments with controlled parameters. This facilitates a smooth transition from pilot scale to commercial production, ensuring that the commercial scale-up of complex pharmaceutical intermediates is predictable and manageable. From an environmental perspective, the aqueous nature of the reaction and the absence of heavy metals align perfectly with increasingly strict global environmental regulations. This reduces the regulatory burden on manufacturers and minimizes the risk of fines or shutdowns due to non-compliance, making it a sustainable choice for long-term production strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enzymatic synthesis route. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these details is crucial for evaluating the feasibility of integrating this technology into existing manufacturing workflows.
Q: How does the mutated monooxygenase improve substrate tolerance compared to wild-type enzymes?
A: The specific amino acid mutations, such as Ser435Thr, alter the active site geometry to accommodate higher concentrations of the bulky omeprazole thioether substrate without significant loss of catalytic activity or enantioselectivity.
Q: What are the critical purification challenges addressed by this enzymatic route?
A: Traditional chemical oxidation often leaves heavy metal residues and racemic byproducts. This enzymatic method produces the S-enantiomer directly with high ee values, simplifying downstream crystallization and reducing the need for extensive chromatographic purification.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the patent demonstrates successful fermentation in both shake flasks and fermenters with controlled dissolved oxygen and pH, indicating robust scalability from laboratory pilot runs to multi-ton commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Esomeprazole Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this enzymatic technology for the global supply of proton pump inhibitor intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate such innovative patent methodologies into robust commercial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are equipped with state-of-the-art fermentation facilities and rigorous QC labs capable of meeting stringent purity specifications required by top-tier pharmaceutical companies. Our commitment to quality ensures that every batch of esomeprazole intermediate produced adheres to the highest international standards, providing our partners with peace of mind and supply security.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this advanced biocatalytic route for their supply chains. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you optimize your production costs and secure a sustainable supply of high-quality esomeprazole intermediates for your future growth.
