Advanced Nickel-Mediated Transmetallation for Scalable Metal-NHC Catalyst Production
The landscape of homogeneous catalysis is undergoing a significant transformation driven by the need for more sustainable and cost-efficient synthetic methodologies for organometallic complexes. Patent CN102351907A introduces a groundbreaking approach to synthesizing metal N-heterocyclic carbene (NHC) complexes, which are pivotal in modern organic synthesis, particularly for cross-coupling reactions and hydrosilylation processes utilized in the production of high-value pharmaceutical intermediates. This technology replaces the conventional and costly silver-mediated transmetallation routes with a novel nickel-mediated pathway. By leveraging nickel N-heterocyclic carbene complexes as carbene transfer reagents, the method achieves high yields under mild reaction conditions while drastically reducing raw material costs. The versatility of this approach is demonstrated through the successful synthesis of complexes involving palladium, platinum, cobalt, ruthenium, and gold, establishing a robust foundation for the industrialized application of transition metal N-heterocyclic carbene catalysts in fine chemical manufacturing.
The limitations of conventional methods for preparing metal-NHC complexes have long hindered their widespread adoption in large-scale industrial processes. Traditionally, the most common strategies involve either the coordination of free carbenes with metal salts or the transmetallation from silver-NHC complexes. The former requires stringent anhydrous and oxygen-free conditions along with specialized basic reagents, as free carbenes are notoriously unstable and prone to self-polymerization. The latter, while more direct, relies heavily on expensive silver oxide reagents to generate the silver-NHC intermediate. These silver complexes are often light-sensitive and can induce oxidative degradation of the carbene ligand, leading to inconsistent batch quality and significant purification challenges. Moreover, the removal of residual silver from the final product adds a costly and complex downstream processing step, which is a major bottleneck for procurement teams aiming for cost reduction in fine chemical manufacturing.
In stark contrast, the novel approach detailed in the patent utilizes nickel-NHC complexes as the carbene source, offering a superior alternative that addresses these historical pain points. This method capitalizes on the stability and lower cost of nickel precursors compared to their silver counterparts. The reaction proceeds via a metathesis mechanism where the nickel complex exchanges its metal center with the target metal precursor, such as palladium or platinum dichloride. A critical advantage of this system is the solubility profile of the byproducts; while the reactants are soluble in organic solvents like acetonitrile, the resulting nickel chloride byproduct is virtually insoluble. This precipitation effectively removes the byproduct from the equilibrium, driving the reaction forward to completion without the need for excessive reagents or harsh conditions. This simplicity in post-treatment and the high tolerance for various functional groups on the NHC ligand make this method exceptionally suitable for the commercial scale-up of complex catalysts.
Mechanistic Insights into Nickel-Mediated Transmetallation
The core chemical innovation lies in the thermodynamic driving force provided by the precipitation of nickel chloride during the metathesis reaction. When the nickel N-heterocyclic carbene complex reacts with a metal precursor of the general formula M(L)nClm in a solvent like acetonitrile, a ligand exchange occurs. The NHC ligand transfers from the nickel center to the target metal M (where M can be Pd, Pt, Ru, etc.), forming the desired metal-NHC complex. Simultaneously, the nickel ion binds with the chloride ions released from the metal precursor to form NiCl2. In solvents such as acetonitrile, NiCl2 has extremely low solubility, causing it to precipitate out of the solution as a solid. According to Le Chatelier's principle, this removal of a product species shifts the equilibrium position strongly towards the formation of the target metal-NHC complex, ensuring high conversion rates and minimizing the presence of unreacted starting materials.
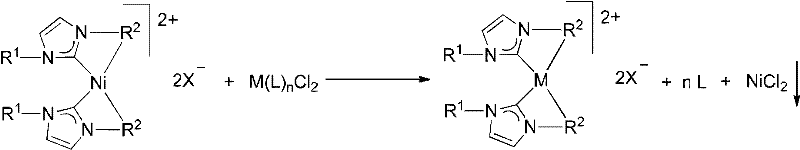
This mechanistic pathway also offers significant advantages regarding impurity control and product purity, which are critical concerns for R&D directors overseeing API intermediate synthesis. Because the reaction does not require strong bases or sensitive free carbenes, side reactions such as ligand decomposition or metal reduction are minimized. The functional group tolerance of the nickel-NHC precursor is excellent, allowing for the incorporation of diverse substituents on the N-heterocyclic ring, such as pyridyl, quinolinyl, or triazolyl groups, without compromising the integrity of the catalytic center. The resulting metal complexes exhibit stable catalytic centers, which is essential for maintaining consistent activity during subsequent organic transformations. The ability to tune the steric and electronic properties of the ligand while maintaining a robust synthesis protocol provides a powerful tool for optimizing catalyst performance for specific industrial applications.
How to Synthesize Metal N-Heterocyclic Carbene Complexes Efficiently
The synthesis protocol outlined in the patent provides a standardized and reproducible method for generating a wide array of metal-NHC complexes suitable for immediate use in catalytic cycles. The process begins by selecting the appropriate nickel-NHC precursor and the desired metal halide precursor, ensuring that the molar ratio is optimized between 1:0.5 and 1:2 depending on the stoichiometry of the target complex. The reaction is typically conducted in acetonitrile, acetone, or nitromethane at moderate temperatures ranging from 35°C to 70°C. Following the reaction period, which can vary from 1 to 24 hours, the mixture is filtered to remove the insoluble nickel chloride byproduct. The filtrate is then concentrated, and the product is crystallized, often using diethyl ether as an anti-solvent, yielding high-purity crystals ready for characterization and application. Detailed standardized synthesis steps are provided in the guide below.
- Prepare the reaction mixture by adding the nickel N-heterocyclic carbene complex and the target metal precursor (e.g., Pd, Pt, Ru chlorides) into an organic solvent such as acetonitrile or acetone.
- Maintain the molar ratio of the nickel complex to the metal precursor between 1: 0.5 and 1:2 to ensure complete ligand exchange.
- Heat the reaction mixture to a temperature range of 35-70°C and stir for 1 to 24 hours, allowing the insoluble NiCl2 byproduct to precipitate and drive the equilibrium forward.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this nickel-mediated synthesis route presents a compelling value proposition centered on cost efficiency and supply reliability. The primary economic driver is the substitution of expensive silver reagents with significantly cheaper nickel salts and precursors. Silver is a precious metal with volatile pricing and complex recovery logistics, whereas nickel is abundant and cost-stable. By eliminating the need for silver-NHC intermediates, manufacturers can achieve substantial cost savings in raw material procurement. Furthermore, the simplified workup procedure, which relies on simple filtration and crystallization rather than complex chromatographic separations or silver scavenging steps, reduces labor costs and solvent consumption. This streamlined process directly contributes to cost reduction in fine chemical manufacturing by lowering the overall cost of goods sold (COGS) for high-value catalysts.
Enhanced supply chain reliability is another critical benefit derived from this technological shift. The nickel-NHC precursors used in this method are chemically stable and do not suffer from the light sensitivity issues that plague silver analogues. This stability allows for longer shelf lives and easier storage and transportation conditions, reducing the risk of material degradation before use. Additionally, the broad applicability of the method to various metal precursors (Pd, Pt, Ru, Fe, Co, etc.) means that a single synthetic platform can be used to produce a diverse portfolio of catalysts. This flexibility mitigates supply risks associated with sourcing specialized reagents for different metal centers. The mild reaction conditions also imply that the process can be easily scaled from laboratory benchtop to multi-ton production without requiring exotic high-pressure or cryogenic equipment, thereby ensuring consistent supply continuity for downstream customers.
Scalability and environmental compliance are further strengthened by the green chemistry attributes of this process. The precipitation of nickel chloride as a solid byproduct simplifies waste management, as the solid can be easily separated and potentially recycled or disposed of according to standard heavy metal waste protocols, avoiding the contamination of liquid waste streams with soluble silver ions. The use of common organic solvents like acetonitrile and acetone, which are readily available and easily recovered through distillation, aligns with modern sustainability goals. The high yields reported in the patent examples, often exceeding 80-90% for many complexes, indicate an atom-economical process that minimizes waste generation. These factors collectively support the commercial scale-up of complex catalysts while adhering to increasingly stringent environmental regulations governing chemical manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this nickel-mediated transmetallation technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy for R&D and technical procurement teams evaluating this synthesis route for their specific applications.
Q: Why is nickel used instead of silver for NHC ligand transfer?
A: Nickel N-heterocyclic carbene complexes are significantly more cost-effective than traditional silver-NHC complexes. Furthermore, the reaction byproduct, nickel chloride, is often insoluble in the reaction solvent (like acetonitrile), which thermodynamically drives the metathesis reaction to completion without requiring expensive silver recovery processes.
Q: What types of metal centers can be synthesized using this method?
A: This transmetallation protocol is highly versatile and supports a wide range of transition metals including Palladium (II), Platinum (II), Cobalt (II/I), Rhodium (I), Iridium (I), Iron (II), Ruthenium (II), Gold (I), and Nickel (II), making it suitable for diverse catalytic applications.
Q: What are the optimal reaction conditions for this synthesis?
A: The process operates under mild conditions, typically requiring temperatures between 35°C and 70°C and reaction times ranging from 1 to 24 hours. Common solvents include acetonitrile, acetone, or nitromethane, which facilitate the precipitation of the nickel byproduct.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Metal N-Heterocyclic Carbene Complex Supplier
As the demand for efficient and sustainable catalytic solutions grows, partnering with an experienced CDMO is essential for translating laboratory innovations into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from pilot studies to full-scale manufacturing is seamless. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of metal-NHC complex meets the highest industry standards, providing the reliability needed for critical pharmaceutical and agrochemical synthesis. We understand the nuances of handling air- and moisture-sensitive organometallic compounds and have the infrastructure to manage them safely and effectively.
We invite you to collaborate with us to optimize your catalyst supply chain and reduce your overall production costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how this advanced nickel-mediated synthesis can enhance your operational efficiency. Let us help you secure a reliable supply of high-performance catalysts that drive your innovation forward.
