Advanced Synthesis of High-Purity Letrozole: Overcoming Isomer Impurities for Commercial Scale-up
Advanced Synthesis of High-Purity Letrozole: Overcoming Isomer Impurities for Commercial Scale-up
The pharmaceutical industry constantly seeks robust manufacturing routes for critical oncology medications, and the synthesis of Letrozole stands as a prime example of where process innovation drives commercial viability. Patent CN100560573C introduces a groundbreaking preparation method for high-purity Letrozole that fundamentally addresses the persistent challenge of isomeric impurities which have plagued traditional production lines. This technology offers a sophisticated solution by leveraging selective salt formation to purify key intermediates, thereby bypassing the inefficient and costly chromatographic steps associated with legacy methods. As a next-generation aromatase inhibitor, Letrozole plays a pivotal role in reducing estrogen levels to inhibit tumor growth in advanced breast cancer treatment, making its reliable supply a matter of global health importance. The structural integrity and purity of the final active pharmaceutical ingredient are paramount, necessitating a synthesis route that guarantees the elimination of the troublesome 1,3,4-position isomer.
This technical insight report analyzes the proprietary methodology detailed in the patent, highlighting how the strategic manipulation of acid-base chemistry during the intermediate stage results in a significantly cleaner reaction profile. For R&D directors and procurement leaders, understanding this shift from physical separation techniques to chemical selectivity is crucial for evaluating long-term supply chain stability. The ability to produce high-purity Letrozole without resorting to complex purification trains not only enhances the economic feasibility of the drug but also ensures a consistent quality profile that meets rigorous regulatory standards. By adopting this refined approach, manufacturers can secure a competitive advantage in the market for reliable API intermediate suppliers, offering products with superior impurity profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
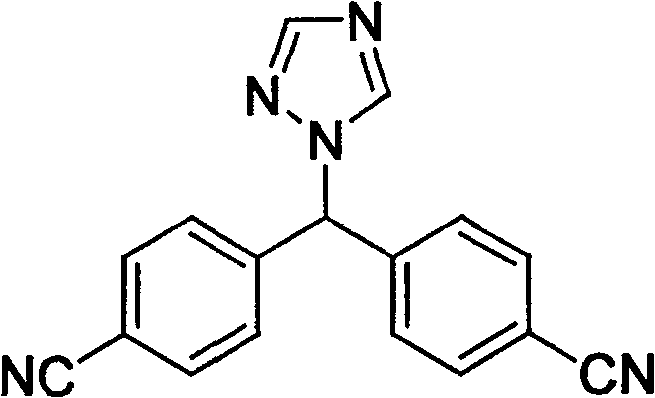
Historically, the preparation of Letrozole has been dominated by the methods described in patent EP0236940, which, while effective in generating the core scaffold, suffers from significant downstream processing drawbacks. The primary deficiency of this conventional route lies in the initial alkylation step, which inevitably produces a mixture of the desired target intermediate (Formula I) and a structurally similar 1,3,4-position isomer (Formula II). These two compounds possess remarkably similar polarity and solubility characteristics, rendering standard physical separation techniques such as simple crystallization or extraction largely ineffective. Consequently, manufacturers relying on this legacy technology are forced to employ column chromatography and multiple rounds of recrystallization to achieve acceptable purity levels. This reliance on labor-intensive purification not only drastically reduces the overall yield of the process but also introduces substantial operational complexity and cost burdens. Furthermore, if the isomer is not completely removed at the intermediate stage, it carries through to the final product, where it becomes extremely difficult to eliminate, thereby compromising the quality of the final API.
The Novel Approach
In stark contrast to the cumbersome physical separation methods of the past, the novel approach disclosed in CN100560573C utilizes a clever chemical differentiation strategy to isolate the desired intermediate. The inventors discovered that despite the similar physical properties of the Formula I target and the Formula II isomer, they exhibit distinct differences in their reactivity when interacting with acids to form salts. By dissolving the crude reaction mixture in a suitable alcoholic solvent, such as ethanol, and carefully controlling the addition rate and quantity of an inorganic acid like hydrochloric acid, the process selectively precipitates the hydrochloride salt of the target Formula I compound. This selective precipitation allows the isomer to remain in the solution phase, enabling a simple filtration step to recover the highly purified intermediate salt. This chemical purification step effectively breaks the bottleneck of isomer removal, streamlining the entire synthesis workflow and setting the stage for a high-yield final coupling reaction that delivers Letrozole with exceptional purity.
Mechanistic Insights into Selective Salt Formation Purification
The core innovation of this synthesis route lies in the exploitation of subtle electronic and steric differences between the regioisomers during the salt formation process. When the mixture of 4,4'-(1H-1,2,4-triazol-1-ylmethylene)dibenzonitrile isomers is exposed to acidic conditions in a protic solvent, the nitrogen atoms on the triazole ring compete for protonation. However, the specific substitution pattern of the target Formula I compound creates a microenvironment that favors the formation of a stable, insoluble hydrochloride salt under controlled conditions. The kinetics of this salt formation are critical; by adding the acid slowly and maintaining specific concentration gradients, the system is driven towards the nucleation and growth of the target salt crystals while the isomer, having a different pKa or solubility product for its corresponding salt, remains dissolved. This mechanism transforms a difficult separation problem into a straightforward unit operation, leveraging fundamental thermodynamic principles to achieve high selectivity. The result is an intermediate stream that is virtually free of the 1,3,4-isomer, which is essential for preventing impurity carryover into the final drug substance.
Following the isolation of the purified intermediate salt, the process proceeds to the final coupling reaction, which requires precise temperature control to maintain product integrity. The free base of the purified intermediate is reacted with p-fluorobenzonitrile in the presence of a strong base, typically potassium tert-butoxide, within a polar aprotic solvent like N,N-dimethylformamide. The reaction is conducted at low temperatures, ranging from -10°C to 0°C, to suppress side reactions and ensure high regioselectivity during the nucleophilic aromatic substitution. This low-temperature protocol is vital for minimizing the formation of new byproducts and preserving the high purity established in the previous purification step. The combination of the selective salt purification and the controlled low-temperature coupling creates a synergistic effect, resulting in a final product with an HPLC purity exceeding 99.6%, demonstrating the robustness of this mechanistic approach for commercial API intermediate manufacturing.
How to Synthesize High-Purity Letrozole Efficiently
Implementing this advanced synthesis route requires a disciplined approach to reaction conditions and stoichiometry to fully realize the benefits of the selective purification mechanism. The process begins with the standard alkylation of 1,2,4-triazole with p-cyanobenzyl bromide to generate the crude isomeric mixture, followed immediately by the critical acid-treatment step in ethanol. Operators must exercise precise control over the acid addition rate to ensure the selective precipitation of the target salt, as rushing this step could lead to co-precipitation of the impurity. Once the purified intermediate is recovered and converted back to the free base, the final coupling with p-fluorobenzonitrile must be maintained strictly within the -10°C to 0°C window to guarantee optimal yield and purity. Detailed standardized operating procedures for each of these stages are essential for technology transfer and scale-up.
- Synthesize the crude intermediate mixture by reacting p-cyanobenzyl bromide with 1,2,4-triazole in chloroform under reflux conditions.
- Purify the intermediate by dissolving the crude mixture in ethanol and slowly adding hydrochloric acid to selectively precipitate the target Formula I hydrochloride salt, filtering out the isomer.
- React the purified free-base intermediate with p-fluorobenzonitrile and potassium tert-butoxide in DMF at low temperatures (-10°C to 0°C) to yield high-purity Letrozole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis method represents a significant opportunity to optimize cost structures and enhance supply reliability for oncology intermediates. By eliminating the need for column chromatography and multiple recrystallizations, the process drastically simplifies the manufacturing workflow, reducing both the time and resources required to produce each batch of Letrozole. This simplification translates directly into lower operational expenditures, as the expensive consumables and specialized equipment associated with chromatographic purification are no longer necessary. Furthermore, the improved yield resulting from the efficient removal of isomers means that less raw material is wasted, contributing to substantial cost savings in the overall production of high-purity API intermediates. The use of common, inexpensive solvents like ethanol and hydrochloric acid for the purification step further enhances the economic attractiveness of this route compared to legacy methods.
- Cost Reduction in Manufacturing: The elimination of chromatographic purification steps removes a major cost driver from the production budget, allowing for more competitive pricing strategies in the global market. Without the need for silica gel, large columns, or extensive solvent recycling systems for chromatography, the capital and operational expenditure for the facility is significantly lowered. Additionally, the higher recovery rate of the desired intermediate means that the effective cost per kilogram of the final product is reduced, providing a clear financial advantage. This efficiency gain allows manufacturers to absorb fluctuations in raw material prices more effectively, ensuring stable pricing for long-term contracts.
- Enhanced Supply Chain Reliability: The simplified process flow reduces the number of potential failure points in the manufacturing line, leading to more consistent batch cycles and reliable delivery schedules. Since the purification relies on standard filtration and crystallization equipment rather than specialized chromatography setups, the process is easier to scale and less prone to technical bottlenecks. This robustness ensures that supply chain disruptions are minimized, providing pharmaceutical partners with a dependable source of critical cancer treatment intermediates. The use of widely available reagents like ethanol and hydrochloric acid also mitigates the risk of raw material shortages that can plague more exotic chemical processes.
- Scalability and Environmental Compliance: The transition from chromatography to selective crystallization greatly improves the environmental footprint of the manufacturing process by reducing solvent waste and hazardous solid waste generation. Crystallization is inherently more scalable than chromatography, allowing for seamless expansion from pilot plant quantities to multi-ton commercial production without losing efficiency. This scalability supports the growing global demand for Letrozole while adhering to increasingly stringent environmental regulations regarding solvent discharge and waste management. The process aligns with green chemistry principles by maximizing atom economy and minimizing the use of auxiliary substances for separation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this high-purity Letrozole synthesis method. These insights are derived directly from the patent data and are intended to clarify the operational benefits and chemical mechanisms for potential partners. Understanding these details is key for R&D teams evaluating the feasibility of technology transfer and for procurement teams assessing the quality assurance capabilities of the supplier.
Q: How does this method remove the difficult 1,3,4-position isomer impurity?
A: The method exploits the significant difference in reactivity between the target intermediate (Formula I) and its isomer (Formula II) when forming salts with acid. By controlling the addition of hydrochloric acid in ethanol, the target compound precipitates as a salt while the isomer remains in solution, allowing for simple filtration.
Q: Does this process require column chromatography for purification?
A: No, this novel approach completely eliminates the need for column chromatography and multiple recrystallizations, which are traditionally required to separate the isomers in older methods like EP0236940.
Q: What purity levels can be achieved with this synthesis route?
A: Experimental data from the patent indicates that this method consistently achieves HPLC purity levels exceeding 99.6%, effectively meeting stringent pharmaceutical standards for API intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Letrozole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of delivering high-quality oncology intermediates that meet the rigorous demands of the global pharmaceutical industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative processes like the selective salt formation method for Letrozole can be successfully translated from the laboratory to full-scale manufacturing. We are committed to maintaining stringent purity specifications and utilizing our rigorous QC labs to verify that every batch of Letrozole intermediate exceeds the 99.6% purity benchmark established by the latest patent technologies. Our infrastructure is designed to support the complex requirements of API synthesis, providing a secure and compliant foundation for your supply chain.
We invite you to collaborate with us to leverage these advanced manufacturing capabilities for your next project. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized synthesis of Letrozole can drive value and efficiency for your organization.
