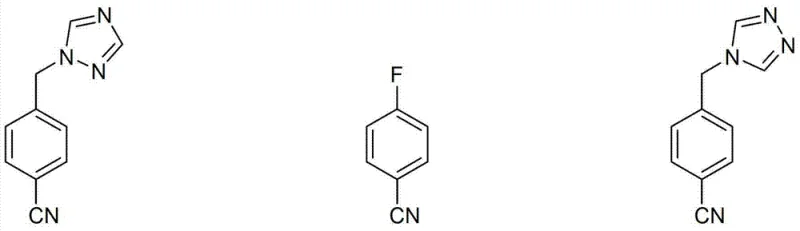
Scalable Regioselective Synthesis of High-Purity Letrozole Intermediates for Commercial API Production
The pharmaceutical industry continuously seeks robust manufacturing pathways for oncology therapeutics, particularly for aromatase inhibitors like Letrozole. Patent CN103298795A introduces a transformative approach to synthesizing the critical intermediate 4-[(1-(1,2,4-triazolyl)methyl]benzonitrile and the final active pharmaceutical ingredient with exceptional regiochemical control. This technology addresses the longstanding challenge of regioisomeric impurity formation, specifically the unwanted 1,3,4-triazolyl derivative, which has historically plagued production yields and purity profiles. By leveraging a novel organic acid salt crystallization strategy, specifically utilizing oxalic acid, the process achieves chemical purities exceeding 99.8% without resorting to industrially impractical purification techniques like column chromatography. This represents a significant leap forward for reliable pharmaceutical intermediate supplier networks aiming to deliver high-quality raw materials for breast cancer treatments.
For R&D teams evaluating process viability, the ability to suppress regioisomer formation at the source is paramount. The disclosed methodology not only optimizes the alkylation step through specific solvent engineering but also introduces a purification checkpoint via salt formation that acts as a highly effective filter for structural impurities. This dual-strategy ensures that the downstream synthesis of Letrozole proceeds with minimal risk of carrying over difficult-to-remove contaminants, thereby streamlining the entire value chain from intermediate to final drug substance.
![Synthetic scheme for 4-[(1-(1,2,4-triazolyl)methyl]benzonitrile showing the alkylation and salt formation steps](/insights/img/letrozole-intermediate-regioselective-synthesis-supplier-20260309075600-01.webp)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Letrozole intermediates has been fraught with significant technical hurdles related to regioselectivity and purification scalability. Traditional methods, such as those described in earlier patents, often result in the formation of substantial quantities of the 1,3,4-triazolyl regioisomer, sometimes reaching levels of 20% to 25% in the crude reaction mixture. This high level of impurity necessitates rigorous purification steps that are ill-suited for large-scale manufacturing. For instance, reliance on column chromatography to separate these isomers results in massive material loss and excessive solvent consumption, rendering the process economically unviable for commercial API production. Furthermore, alternative purification strategies involving hydrochloride salt formation have proven inconsistent, often failing to completely exclude the unwanted isomer or leading to significant yield losses during the isolation of the free base. These inefficiencies create bottlenecks in cost reduction in pharmaceutical intermediates manufacturing and compromise the supply continuity required by global health markets.
The Novel Approach
The innovative process detailed in CN103298795A circumvents these legacy issues by integrating a mild alkylation protocol with a highly selective crystallization technique. Instead of forcing the reaction under harsh conditions that promote isomer scrambling, the new method utilizes a mixed solvent system of acetone and methanol with catalytic potassium iodide to drive the reaction at ambient temperatures. More critically, the purification strategy shifts from non-selective acid salts to specific organic acid salts, with oxalate being the preferred embodiment. This specific salt form exhibits distinct solubility characteristics that allow the desired 1,2,4-triazolyl intermediate to crystallize while leaving the 1,3,4-isomer in the mother liquor. This fundamental shift in purification logic eliminates the need for chromatography entirely, offering a pathway to high-purity intermediates that is both economically efficient and technically robust for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Regioselective Alkylation and Salt Crystallization
The success of this synthetic route lies in the precise manipulation of reaction kinetics and thermodynamic solubility profiles. In the alkylation step, the use of potassium iodide serves as a crucial catalyst, likely facilitating a Finkelstein-type halogen exchange that enhances the electrophilicity of the benzyl bromide species in the polar aprotic-protic solvent mixture. This activation allows the nucleophilic attack by the 1,2,4-triazole to proceed efficiently at lower temperatures, typically between 20°C and 35°C, minimizing the thermal energy available for the formation of the thermodynamically stable but undesired 1,3,4-regioisomer. The solvent system, comprising a specific ratio of ketone and alcohol, further modulates the solvation shell around the transition state, favoring the kinetic product. Following the reaction, the mechanism of purification relies on the differential lattice energy and solvation of the organic acid salts. The oxalate salt of the desired intermediate forms a stable crystal lattice that energetically excludes the structurally similar isomer, a phenomenon known as lattice rejection. This ensures that even if trace amounts of the isomer are formed during the reaction, they are effectively purged during the crystallization of the salt, resulting in a free base with regioisomeric purity that meets stringent regulatory standards.
Furthermore, the control of impurity propagation is maintained throughout the subsequent conversion to Letrozole. By ensuring the intermediate is substantially free of the 1,3,4-isomer before the coupling with p-fluorobenzonitrile, the process prevents the formation of the corresponding Letrozole regioisomer, which is notoriously difficult to separate from the final API due to their similar physicochemical properties. The final crystallization of Letrozole itself, using solvent systems like ethyl acetate and methyl tert-butyl ether, provides an additional layer of purification. This multi-stage control strategy ensures that the final impurity profile is dominated by process-related impurities that are easily managed, rather than intractable structural isomers. Such mechanistic rigor is essential for maintaining the integrity of the supply chain for high-purity pharmaceutical intermediates and ensuring patient safety through consistent drug quality.
How to Synthesize 4-[(1-(1,2,4-triazolyl)methyl]benzonitrile Efficiently
The implementation of this advanced synthesis protocol requires careful attention to solvent ratios and temperature controls to maximize the benefits of the regioselective pathway. The process begins with the preparation of a reaction mixture containing p-cyanobenzyl bromide, potassium carbonate, and catalytic potassium iodide in a blend of acetone and methanol. The 1,2,4-triazole is then introduced slowly to maintain thermal equilibrium, ensuring the reaction proceeds smoothly without exothermic spikes that could degrade selectivity. Once the alkylation is complete, the workup involves a strategic conversion to the oxalate salt, which serves as the primary purification vehicle. Detailed standard operating procedures for stoichiometry, addition rates, and crystallization parameters are critical for reproducing the high purity levels reported in the patent data. The following guide outlines the standardized synthesis steps derived from the proprietary methodology.
- React p-cyanobenzyl bromide with 1,2,4-triazole in an acetone and methanol solvent mixture using potassium carbonate and catalytic potassium iodide at ambient temperature.
- Purify the crude intermediate by forming an organic acid salt, specifically the oxalate salt, through crystallization to remove regioisomeric impurities.
- Isolate the pure free base from the purified salt using aqueous base treatment followed by crystallization from non-polar solvents like n-heptane.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this patented process offers substantial strategic advantages over conventional manufacturing routes. The elimination of column chromatography is perhaps the most significant driver of value, as it removes a major bottleneck associated with solvent intensity, batch cycle time, and yield loss. By replacing chromatographic purification with crystallization, the process drastically reduces the volume of organic solvents required, leading to significant cost reduction in pharmaceutical intermediates manufacturing through lower waste disposal fees and reduced raw material procurement costs. Additionally, the ability to run the key alkylation step at ambient temperature reduces energy consumption associated with heating and cooling cycles, further enhancing the economic profile of the production line. These efficiencies translate directly into more competitive pricing structures for buyers seeking long-term supply agreements for oncology API precursors.
- Cost Reduction in Manufacturing: The transition from chromatographic purification to selective salt crystallization fundamentally alters the cost structure of producing Letrozole intermediates. Chromatography is inherently inefficient at scale, consuming vast amounts of silica gel and solvents while recovering only a fraction of the input material. By utilizing the oxalate salt crystallization method, manufacturers can achieve high recovery rates and minimize solvent usage, resulting in substantial cost savings. The avoidance of expensive purification media and the reduction in processing time allow for a leaner manufacturing operation that can pass savings on to procurement partners, ensuring a more sustainable economic model for high-volume API production.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by complex purification steps that are prone to variability and failure. The robust nature of the crystallization-based purification described in this patent ensures consistent batch-to-batch quality, reducing the risk of production delays caused by out-of-specification results. Furthermore, the reagents used, such as potassium carbonate and oxalic acid, are commodity chemicals with stable global supply chains, unlike specialized catalysts or resins that might face availability constraints. This reliance on widely available raw materials enhances the resilience of the supply chain, guaranteeing reducing lead time for high-purity pharmaceutical intermediates and providing buyers with greater confidence in meeting their own production schedules.
- Scalability and Environmental Compliance: As regulatory pressure mounts regarding solvent emissions and waste generation, processes that minimize environmental impact are increasingly valuable. The new method's reliance on crystallization rather than chromatography significantly lowers the E-factor (mass of waste per mass of product), aligning with green chemistry principles. The use of common solvents like acetone and methanol, which are easier to recover and recycle compared to complex solvent mixtures used in chromatography, simplifies waste management protocols. This environmental efficiency not only reduces compliance costs but also future-proofs the manufacturing process against tightening environmental regulations, making it a preferred choice for sustainable commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis pathway is crucial for stakeholders evaluating its integration into their supply chains. The following questions address common inquiries regarding the purity capabilities, solvent systems, and scalability of the process described in CN103298795A. These answers are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for technical decision-making.
Q: How does the oxalate salt method improve purity compared to hydrochloride salts?
A: The patent demonstrates that forming the oxalate salt allows for superior selectivity in crystallization, effectively excluding the unwanted 1,3,4-triazolyl regioisomer which often co-crystallizes or remains in solution with hydrochloride salts, achieving purity levels above 99.8%.
Q: What solvent system is optimal for the alkylation reaction?
A: The preferred solvent system is a mixture of a dialkyl ketone, such as acetone, and a fatty alcohol, such as methanol, typically in a volume ratio ranging from 95:5 to 50:50, which facilitates the reaction at ambient temperatures without requiring reflux.
Q: Can this process eliminate the need for column chromatography?
A: Yes, the described method relies on selective salt formation and crystallization rather than chromatographic separation, making it highly suitable for industrial scale-up where column chromatography is economically and technically unfeasible.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-[(1-(1,2,4-triazolyl)methyl]benzonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the production of life-saving oncology medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated crystallization techniques required for this Letrozole intermediate are executed with precision. We are committed to delivering products that meet stringent purity specifications, leveraging our rigorous QC labs to verify that every batch is substantially free of regioisomeric impurities. Our infrastructure is designed to support the complex solvent management and temperature controls necessary for this advanced synthesis, guaranteeing a supply of material that adheres to the highest international standards for pharmaceutical manufacturing.
We invite procurement leaders and technical directors to engage with us to explore how this optimized process can benefit your specific production needs. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this regioselective route. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your volume requirements. Let us collaborate to secure a reliable, cost-effective, and high-quality supply chain for your Letrozole API production.
