Revolutionizing Vonoprazan Production: A Deep Dive into Green Synthesis and Commercial Scalability
Revolutionizing Vonoprazan Production: A Deep Dive into Green Synthesis and Commercial Scalability
The pharmaceutical landscape for gastric acid-related diseases (ARDs) has shifted dramatically with the advent of Potassium-Competitive Acid Blockers (P-CABs), among which Vonoprazan stands out as a premier therapeutic agent. Recent intellectual property developments, specifically patent CN107915720B, have unveiled a transformative preparation method that addresses long-standing inefficiencies in the manufacturing of this critical API intermediate. This technical insight report analyzes the breakthrough synthesis route, which leverages aqueous aminolysis and controlled borohydride reduction to achieve superior yield and purity profiles. For R&D Directors and Procurement Managers seeking a reliable Vonoprazan supplier, understanding these mechanistic advancements is crucial for securing a cost-effective and scalable supply chain. The new methodology not only simplifies the reaction sequence but also aligns with stringent global environmental regulations, offering a compelling value proposition for commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Vonoprazan has been plagued by convoluted reaction pathways that impose significant operational burdens and safety risks. Traditional routes, such as those described in earlier patent literature like CN101300229, often necessitate a minimum of six to eight discrete chemical steps to reach the final fumarate salt. A critical bottleneck in these legacy processes is the reliance on diisobutylaluminum hydride (DIBAL-H) for the reduction of ester groups, a reagent that is not only prohibitively expensive but also highly pyrophoric and sensitive to air and moisture. Furthermore, these reactions typically demand cryogenic conditions, often requiring temperatures as low as -78°C, which drastically increases energy consumption and necessitates specialized cooling infrastructure that is difficult to maintain in large-scale production environments. The subsequent oxidation steps to convert alcohols back to aldehydes frequently suffer from over-oxidation issues, leading to carboxylic acid impurities that are challenging to remove without resorting to column chromatography. Such purification techniques are fundamentally incompatible with Good Manufacturing Practice (GMP) standards for bulk pharmaceutical production due to solvent intensity and low throughput.
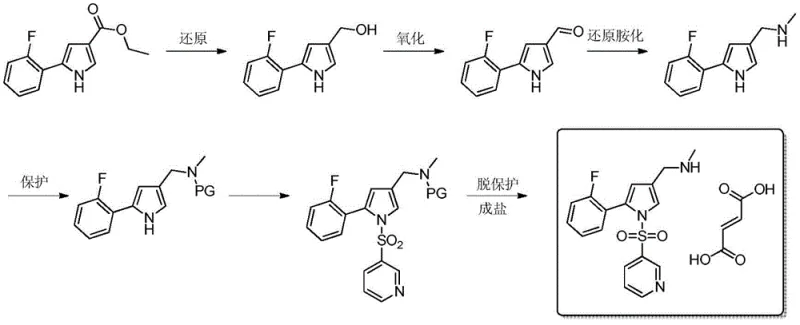
The Novel Approach
In stark contrast to the cumbersome legacy pathways, the innovative method disclosed in CN107915720B streamlines the synthesis into a highly efficient three-step sequence comprising aminolysis, reduction, and salt formation. This novel approach strategically bypasses the unstable aldehyde intermediate stage that characterizes older routes, thereby eliminating the need for repetitive oxidation-reduction cycles that degrade overall yield. By utilizing 5-(2-fluorophenyl)-1-(pyridine-3-sulfonyl)-1H-pyrrole-3-carboxylate as a commercially available starting material, the process initiates with a direct aminolysis reaction in an aqueous medium, which is a significant departure from the anhydrous organic solvents typically required. The subsequent reduction step employs a robust combination of metal borohydrides and Lewis acids, which operates effectively at moderate temperatures between 50°C and 80°C, removing the safety hazards associated with ultra-low temperature operations. This fundamental redesign of the synthetic logic ensures that the process is not only chemically superior but also inherently safer and more amenable to continuous manufacturing workflows, providing a robust foundation for cost reduction in API intermediate manufacturing.
Mechanistic Insights into Borohydride-Mediated Amide Reduction
The core chemical innovation of this patented process lies in the selective reduction of the amide intermediate to the corresponding amine without affecting other sensitive functional groups within the molecular scaffold. The mechanism involves the activation of the amide carbonyl by a Lewis acid, such as boron trifluoride etherate, which enhances the electrophilicity of the carbonyl carbon and facilitates hydride transfer from the borohydride species. This synergistic catalytic system allows for the direct conversion of the amide to the amine in a single pot, avoiding the formation of the aldehyde intermediate that is prone to side reactions in traditional routes. The use of sodium borohydride or potassium borohydride as the reducing agent provides a controlled release of hydride ions, ensuring that the reduction stops at the amine stage without over-reducing the pyrrole ring or the sulfonyl group. This high degree of chemoselectivity is paramount for maintaining the structural integrity of the Vonoprazan molecule and minimizing the generation of structurally related impurities that could complicate downstream purification. The reaction kinetics are optimized by conducting the process in tetrahydrofuran or methyltetrahydrofuran, which solubilizes the intermediates effectively while maintaining a stable reaction environment for the Lewis acid complex.
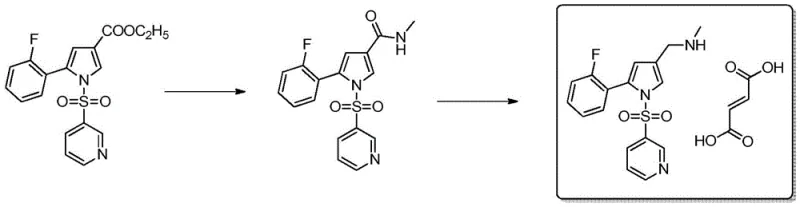
Impurity control is another critical aspect where this new mechanism offers substantial advantages over conventional methods. In older synthesis routes, the oxidation of alcohol intermediates often leads to the formation of carboxylic acid byproducts, which are difficult to separate from the target aldehyde due to similar polarity. The new aminolysis-reduction pathway completely circumvents this oxidative step, thereby eliminating the root cause of these specific acidic impurities. Furthermore, the aqueous workup conditions employed in the aminolysis step allow for the efficient removal of water-soluble byproducts and excess methylamine, resulting in a cleaner crude intermediate before the reduction step even begins. The final salt formation with fumaric acid is conducted under mild conditions that promote the crystallization of the target polymorph while leaving residual organic impurities in the mother liquor. This inherent ability to purge impurities through crystallization rather than chromatography is a key factor in achieving the high purity specifications required for pharmaceutical-grade Vonoprazan, ensuring that the final product meets stringent regulatory standards for impurity profiles.
How to Synthesize Vonoprazan Efficiently
Implementing this advanced synthesis route requires precise control over reaction parameters to maximize yield and ensure reproducibility at scale. The process begins with the aminolysis of the ester starting material using methylamine in water, followed by the critical borohydride reduction step, and concludes with fumarate salt formation. Detailed operational parameters, including specific molar equivalents, temperature ramps, and quenching protocols, are essential for successful technology transfer. The following guide outlines the standardized synthesis steps derived from the patent data to assist process engineers in evaluating feasibility.
- Perform aminolysis of the ester intermediate with methylamine in water at 50-80°C to form the amide.
- Reduce the amide intermediate using sodium borohydride and a Lewis acid in THF at 50-80°C.
- Form the fumarate salt in water or non-aqueous solvent to obtain the final API.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis route represents a strategic opportunity to optimize costs and mitigate supply risks associated with Vonoprazan production. The elimination of hazardous reagents like DIBAL-H and the removal of cryogenic requirements significantly lower the barrier to entry for manufacturing partners, expanding the pool of qualified suppliers capable of producing this intermediate. By simplifying the process from six or more steps down to just three, the overall production cycle time is drastically reduced, which enhances the responsiveness of the supply chain to market demand fluctuations. The use of water as a solvent in the initial step not only reduces the cost of raw materials but also minimizes the environmental footprint associated with solvent disposal and recovery, aligning with corporate sustainability goals. These operational efficiencies translate into a more resilient supply chain that is less susceptible to disruptions caused by reagent shortages or regulatory changes regarding hazardous chemical handling.
- Cost Reduction in Manufacturing: The replacement of expensive and sensitive reagents with commodity chemicals like sodium borohydride and aqueous methylamine drives down the direct material costs significantly. Eliminating the need for column chromatography removes a major cost center associated with silica gel, solvents, and labor-intensive purification processes. The higher overall yield achieved through this streamlined route means that less starting material is required to produce the same amount of final API, further enhancing the economic viability of the process. Additionally, the moderate reaction temperatures reduce energy consumption for heating and cooling, contributing to lower utility costs per kilogram of product. These cumulative savings allow for a more competitive pricing structure without compromising on the quality or purity of the final pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable reagents ensures that production is not held hostage by the supply constraints of specialized catalysts or cryogenic gases. The robustness of the aqueous aminolysis step makes the process less sensitive to moisture variations, reducing the risk of batch failures due to environmental factors. This stability allows for more accurate production planning and inventory management, ensuring consistent availability of high-purity Vonoprazan intermediates for downstream formulation. Furthermore, the simplified process flow reduces the number of intermediate isolation steps, minimizing the potential for material loss or contamination during transfer between processing units. This continuity is vital for maintaining a steady flow of materials to meet the rigorous delivery schedules of global pharmaceutical clients.
- Scalability and Environmental Compliance: The design of this synthesis route is inherently scalable, as it avoids unit operations that are difficult to enlarge, such as low-temperature batch reactions or complex chromatographic separations. The use of water and common organic solvents like THF simplifies waste treatment protocols, making it easier for manufacturing sites to comply with increasingly strict environmental regulations. The reduction in hazardous waste generation lowers the costs associated with waste disposal and environmental monitoring. By adopting this green chemistry approach, manufacturers can future-proof their operations against regulatory tightening while demonstrating a commitment to sustainable manufacturing practices. This alignment with environmental standards is increasingly becoming a key criterion for supplier selection by major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this new Vonoprazan synthesis technology. These answers are derived directly from the patent specifications and are intended to clarify the operational benefits and chemical rationale behind the process. Understanding these details is essential for stakeholders evaluating the feasibility of adopting this route for commercial production.
Q: How does the new synthesis route improve safety compared to conventional methods?
A: The new route eliminates the use of highly flammable DIBAL-H and ultra-low temperature conditions (-78°C), replacing them with safer borohydride reagents and moderate temperatures.
Q: What are the environmental benefits of using water as a solvent in the first step?
A: Using water avoids toxic organic solvents and methylamine gas, significantly reducing hazardous waste and aligning with green chemistry principles for large-scale manufacturing.
Q: Does this process avoid column chromatography for purification?
A: Yes, the process relies on crystallization and extraction, eliminating the need for column chromatography which is a major bottleneck for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vonoprazan Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthesis technologies to maintain competitiveness in the global pharmaceutical market. Our technical team has extensively analyzed the pathway described in CN107915720B and possesses the expertise to scale this diverse pathway from 100 kgs to 100 MT/annual commercial production with precision. We are committed to delivering stringent purity specifications through our rigorous QC labs, ensuring that every batch of Vonoprazan intermediate meets the highest industry standards. Our facility is equipped to handle the specific requirements of this green synthesis route, including aqueous processing and controlled reduction steps, guaranteeing a consistent and high-quality supply for your drug development needs.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic advantages of switching to this method. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your volume and timeline. Let us partner with you to engineer a supply chain that is not only cost-effective but also robust and sustainable for the long term.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
