Advanced Synthesis of Asymmetric Bisfluorosulfonimide Potassium for Next-Generation Lithium Battery Electrolytes
The rapid evolution of lithium-ion battery technology demands electrolyte salts that offer superior thermal stability and electrochemical performance compared to traditional LiPF6. Patent CN102180816B introduces a groundbreaking preparation method for asymmetric bisfluorosulfonimide potassium, a critical precursor for advanced electrolyte systems. This innovation addresses the longstanding industry challenges associated with the thermal decomposition and hydrolysis of conventional salts, which often generate corrosive HF and degrade battery lifespan. By leveraging a novel synthetic pathway that minimizes the use of highly toxic reagents, this technology paves the way for safer and more efficient energy storage solutions. For global procurement teams seeking a reliable battery & energy storage materials supplier, understanding the technical nuances of this patent is essential for securing a competitive edge in the supply chain. The method not only ensures high product purity but also simplifies the separation process, making it an attractive candidate for industrial adoption.
The transition to next-generation energy storage requires materials that can withstand rigorous operating conditions without compromising safety. The asymmetric structure of the bisfluorosulfonimide anion provides a unique balance of conductivity and stability, mitigating the corrosion issues often seen with aluminum current collectors in high-voltage applications. This patent outlines a robust chemical framework that allows for the precise tuning of the perfluoroalkyl chain length, enabling manufacturers to optimize viscosity and conductivity profiles for specific battery chemistries. As the demand for electric vehicles and grid storage escalates, the ability to source high-purity electrolyte salt intermediates becomes a strategic imperative. This report analyzes the technical merits and commercial viability of this synthesis route, providing actionable insights for R&D directors and supply chain leaders aiming to integrate superior electrolyte components into their product lines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of asymmetric imide salts has relied heavily on hazardous reagents such as bis(fluorosulfonyl) oxide or fluorosulfonic acid, which pose significant safety risks and require specialized corrosion-resistant equipment. These traditional pathways often involve complex purification steps to remove toxic byproducts, leading to increased production costs and environmental burdens. Furthermore, the use of phosphorus pentachloride in the presence of fluorosulfonic acid generates substantial waste streams that complicate regulatory compliance and waste management protocols. The thermal instability of precursors in these older methods can also lead to inconsistent batch quality, affecting the reliability of the final electrolyte formulation. For procurement managers focused on cost reduction in electronic chemical manufacturing, these inefficiencies represent a major bottleneck. The reliance on剧毒 (highly toxic) materials not only elevates operational risks but also restricts the scalability of production facilities, limiting the ability to meet growing market demand without significant capital investment in safety infrastructure.
The Novel Approach
In contrast, the method disclosed in CN102180816B utilizes sulfonyl chloride and hexamethyldisilazane (HMDS) to construct the asymmetric imide framework, drastically reducing the toxicity profile of the raw materials. This approach eliminates the need for handling extremely corrosive fluorosulfonic acid, thereby lowering the requirements for reactor material specifications and extending equipment lifespan. The reaction conditions are moderated, with reflux temperatures typically around 110°C, which are easily manageable in standard stainless steel reactors commonly found in fine chemical plants. The byproduct of the silylation step, trimethylsilyl chloride, is gaseous and easily removed from the reaction system, driving the equilibrium forward and simplifying the workup procedure. This streamlined process enhances overall yield consistency and reduces the burden on downstream purification units. By shifting to this safer chemistry, manufacturers can achieve substantial cost savings through reduced waste treatment expenses and lower maintenance costs, aligning perfectly with the goals of a reliable battery & energy storage materials supplier committed to sustainable operations.
Mechanistic Insights into Silylation and Fluorination Chemistry
The core of this synthesis lies in the strategic use of silylation to activate the sulfonamide nitrogen, facilitating the subsequent introduction of the sulfonyl group. Initially, the sulfonamide is converted to its sodium salt using sodium methoxide, which serves as a nucleophile in the reaction with HMDS. This step forms a trimethylsilyl-protected intermediate, effectively shielding the nitrogen atom and increasing its reactivity towards electrophilic attack by sulfonyl chloride. The molar ratio of HMDS to sodium sulfonamide is carefully controlled between 5:1 and 15:1 to ensure complete conversion while minimizing excess reagent waste. The reaction is conducted under nitrogen protection to prevent moisture ingress, which could hydrolyze the sensitive silyl groups. This meticulous control over reaction parameters is crucial for maintaining the integrity of the asymmetric structure, ensuring that the final product exhibits the desired electrochemical properties. Understanding this mechanism allows R&D teams to optimize process parameters for maximum efficiency and purity.
Following the formation of the silylated intermediate, the introduction of the fluorine atom is achieved through a nucleophilic substitution reaction with potassium fluoride (KF). The intermediate chlorosulfonyl perfluoroalkylsulfonimide is reacted with KF in anhydrous acetonitrile under reflux conditions for 12 to 48 hours. This extended reaction time ensures the complete displacement of the chlorine atom, which is critical for achieving high purity levels required for battery-grade materials. The use of KF is advantageous due to its low cost and availability compared to other fluorinating agents. After the reaction, the product is isolated through filtration and recrystallization using dichloromethane, which effectively removes inorganic salts and unreacted starting materials. 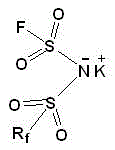 This structural confirmation is vital for quality control, as the presence of symmetric impurities can detrimentally affect battery performance. The final drying step under vacuum ensures the removal of residual solvents, yielding an anhydrous salt suitable for electrolyte formulation.
This structural confirmation is vital for quality control, as the presence of symmetric impurities can detrimentally affect battery performance. The final drying step under vacuum ensures the removal of residual solvents, yielding an anhydrous salt suitable for electrolyte formulation.
How to Synthesize Asymmetric Bisfluorosulfonimide Potassium Efficiently
Implementing this synthesis route requires precise adherence to the stoichiometric ratios and temperature profiles outlined in the patent to ensure reproducibility and safety. The process begins with the preparation of sodium sulfonamide, followed by the silylation step which sets the stage for the asymmetric coupling. Operators must maintain strict anhydrous conditions throughout the silylation and fluorination stages to prevent hydrolysis of the reactive intermediates. The final purification via recrystallization is a critical control point for achieving the stringent purity specifications demanded by the battery industry. Detailed standard operating procedures (SOPs) should be developed based on the experimental examples provided, scaling from laboratory glassware to pilot plant reactors. 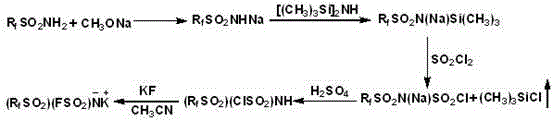
- Preparation of Sodium Sulfonamide via reaction of sulfonamide with sodium methoxide in methanol and ether.
- Silylation using hexamethyldisilazane (HMDS) to form trimethylsilane perfluoroalkylsulfonimide sodium.
- Reaction with sulfonyl chloride followed by fluorination with KF to yield the final potassium salt.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders, the adoption of this synthesis method offers significant strategic benefits beyond mere technical performance. The reduction in raw material toxicity translates directly into lower insurance premiums and reduced regulatory compliance burdens, creating a more resilient supply chain. By avoiding the use of specialized corrosion-resistant alloys required for handling fluorosulfonic acid, capital expenditure for new production lines can be significantly optimized. The simplicity of the workup procedure, particularly the easy removal of gaseous byproducts, reduces the load on wastewater treatment facilities, contributing to a smaller environmental footprint. These factors collectively enhance the commercial scale-up of complex fluorinated compounds, making it feasible to meet large-volume orders without compromising on safety or quality. Procurement managers can leverage these efficiencies to negotiate better terms with suppliers who utilize this advanced technology.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents like bis(fluorosulfonyl) oxide leads to a direct decrease in raw material costs. Furthermore, the mild reaction conditions reduce energy consumption associated with heating and cooling cycles, contributing to overall operational efficiency. The ability to use standard solvents like acetonitrile and ether, which are readily available in the global chemical market, mitigates supply risk and price volatility. This economic advantage allows manufacturers to offer competitive pricing for high-purity electrolyte salt precursors, strengthening their position in the market. The simplified purification process also reduces labor costs and solvent usage, further driving down the cost of goods sold.
- Enhanced Supply Chain Reliability: By utilizing widely available reagents such as sulfonyl chloride and KF, the dependency on niche chemical suppliers is minimized, reducing the risk of supply disruptions. The robustness of the reaction against minor variations in conditions ensures consistent batch-to-batch quality, which is critical for maintaining long-term contracts with battery manufacturers. The lower equipment corrosion rates extend the maintenance intervals for reactors and piping, ensuring higher uptime and production availability. This reliability is essential for reducing lead time for high-purity battery materials, allowing customers to plan their production schedules with greater confidence. A stable supply of key intermediates is a cornerstone of a resilient battery supply chain.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the use of standard unit operations like reflux, distillation, and crystallization. The reduced generation of hazardous waste simplifies the permitting process for new facilities and lowers the cost of waste disposal. Compliance with increasingly strict environmental regulations is easier to achieve when the process avoids the generation of persistent organic pollutants or heavy metal contaminants. This environmental stewardship enhances the brand reputation of the manufacturer and aligns with the sustainability goals of major automotive and electronics clients. Scalability ensures that production can be ramped up quickly to meet surges in demand without compromising safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of asymmetric bisfluorosulfonimide potassium. These answers are derived directly from the technical disclosures in patent CN102180816B and general industry knowledge regarding electrolyte salt manufacturing. Understanding these details helps stakeholders make informed decisions about integrating this material into their supply chains. The focus is on practical implementation, safety considerations, and quality assurance measures that define the commercial viability of this technology.
Q: What are the safety advantages of this synthesis method compared to traditional routes?
A: This method avoids highly toxic reagents like fluorosulfonic acid or bis(fluorosulfonyl) oxide, utilizing sulfonyl chloride instead, which significantly reduces equipment corrosion and operational hazards.
Q: How does the purity of the product impact battery performance?
A: High purity is critical for electrolyte salts to prevent side reactions at the electrode interface. The recrystallization steps described ensure minimal impurity levels, enhancing cycle life and thermal stability.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the method uses standard solvents like acetonitrile and ether and operates at moderate temperatures, making it adaptable for commercial scale-up without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Asymmetric Bisfluorosulfonimide Potassium Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of electrolyte salt precursor meets the highest industry standards. We understand the critical nature of battery materials and have implemented robust quality management systems to track every step of the manufacturing process. Our team of expert chemists is dedicated to optimizing synthesis routes to maximize yield and minimize environmental impact, aligning with our commitment to sustainable chemistry. Partnering with us means gaining access to a supply chain that prioritizes safety, quality, and continuity.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific production requirements. Our experts are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your project goals. Whether you are developing next-generation lithium batteries or exploring new energy storage technologies, we are committed to being your trusted partner in chemical innovation. Reach out today to discuss how we can collaborate to bring high-performance electrolyte solutions to the market efficiently and safely.
