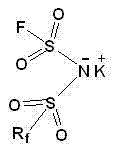
Advanced Synthesis and Commercial Scale-Up of Asymmetric Bisfluorosulfonimide Potassium for Next-Gen Batteries
The landscape of lithium-ion battery technology is undergoing a critical transformation, driven by the urgent need for electrolyte salts that offer superior thermal stability and electrochemical performance compared to the industry-standard LiPF6. Patent CN102180816A introduces a groundbreaking preparation method for asymmetric bisfluorosulfonimide potassium, a pivotal precursor for next-generation electrolytes that mitigate the hydrolysis and corrosion issues plaguing current technologies. This innovation represents a significant leap forward for any reliable battery electrolyte supplier seeking to enhance product longevity and safety profiles in energy storage systems. By leveraging a novel synthetic pathway that circumvents the use of extremely hazardous fluorinating agents, this technology enables the production of high-purity lithium battery additives with reduced environmental impact. The structural integrity of the resulting asymmetric imide ensures optimal ionic conductivity while maintaining compatibility with aluminum current collectors, addressing a major bottleneck in high-voltage battery applications. As the demand for electric vehicles and grid storage escalates, mastering this synthesis route becomes essential for maintaining competitiveness in the global supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of asymmetric imides and their alkali metal salts has been fraught with significant safety hazards and operational complexities that hinder efficient cost reduction in electronic chemical manufacturing. Traditional protocols frequently rely on the utilization of bis(fluorosulfonyl)imide or fluorosulfonic acid, substances characterized by extreme toxicity and corrosiveness that demand specialized containment infrastructure and rigorous waste management procedures. These aggressive reagents not only pose severe risks to personnel safety but also accelerate the degradation of standard reactor vessels, leading to frequent maintenance downtime and inflated capital expenditure. Furthermore, the handling of such volatile compounds often results in inconsistent reaction yields due to side reactions triggered by trace moisture or thermal instability during processing. The reliance on these dangerous precursors creates a substantial barrier to entry for manufacturers aiming to scale production without compromising on regulatory compliance or worker safety standards. Consequently, the industry has long sought an alternative pathway that balances chemical efficacy with operational practicality and economic viability.
The Novel Approach
The methodology disclosed in the patent data presents a transformative solution by substituting hazardous fluorinating agents with sulfonyl chloride, a reagent that offers a markedly improved safety profile while maintaining high reaction efficiency. This strategic substitution eliminates the need for handling free fluorosulfonic acid, thereby drastically simplifying the equipment requirements and allowing for the use of standard glass-lined or stainless-steel reactors commonly found in fine chemical facilities. The process facilitates the generation of gaseous trimethylsilyl chloride as a byproduct, which is easily removed from the reaction system, driving the equilibrium towards the desired product and enhancing overall conversion rates. This inherent advantage streamlines the purification workflow, reducing the number of downstream processing steps required to achieve pharmaceutical-grade purity levels. By mitigating the risks associated with toxic raw materials, this approach not only safeguards the workforce but also aligns with increasingly stringent global environmental regulations regarding hazardous chemical usage. Ultimately, this novel route provides a robust foundation for the commercial scale-up of complex fluorinated compounds needed for advanced energy storage solutions.
Mechanistic Insights into Silylation and Fluorination Chemistry
A deep understanding of the reaction mechanism is crucial for R&D teams aiming to optimize yield and minimize impurity profiles during the production of these specialized electrolyte salts. The process initiates with the formation of sodium sulfonamide through a nucleophilic substitution reaction, followed by a critical silylation step where hexamethyldisilazane reacts with the sodium salt under nitrogen protection to form a trimethylsilyl intermediate. This silylation is pivotal as it activates the nitrogen center, making it susceptible to subsequent electrophilic attack by sulfonyl chloride in a controlled manner. The reaction conditions, specifically the temperature range of 110°C and the use of anhydrous acetonitrile, are carefully calibrated to prevent premature decomposition of the sensitive silylated species. Following this, the introduction of sulfonyl chloride proceeds via a displacement mechanism that releases volatile byproducts, effectively pushing the reaction to completion without the need for excessive reagent loading. This mechanistic precision ensures that the resulting intermediate possesses the correct stoichiometry for the final fluorination step, which is essential for achieving the target asymmetric structure.
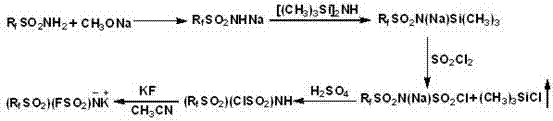
Impurity control is maintained through a series of meticulous workup procedures designed to isolate the target potassium salt from unreacted starting materials and inorganic salts. After the fluorination reaction with potassium fluoride, the mixture undergoes filtration to remove insoluble inorganic residues, followed by a concentration step that removes the bulk of the solvent under reduced pressure. The subsequent recrystallization using dichloromethane is a critical purification stage that exploits solubility differences to exclude organic impurities and residual halides that could detrimentally affect battery performance. This level of purification is vital because even trace amounts of water or acidic impurities can lead to the generation of hydrofluoric acid within the battery cell, causing catastrophic failure over time. The ability to consistently produce material with stringent purity specifications demonstrates the robustness of this synthetic design against common contamination vectors. For procurement managers, this implies a lower risk of batch rejection and higher reliability in the supply of high-purity electrolyte salts for sensitive electronic applications.
How to Synthesize Asymmetric Bisfluorosulfonimide Potassium Efficiently
Implementing this synthesis route requires strict adherence to anhydrous conditions and precise stoichiometric control to ensure reproducible results across different production batches. The process begins with the preparation of the sulfonamide sodium salt, which serves as the foundational building block for the entire molecular architecture. Operators must ensure that all solvents, particularly methanol and ether, are thoroughly dried to prevent hydrolysis of the reactive intermediates formed in subsequent stages. The silylation reaction demands a nitrogen atmosphere to exclude oxygen and moisture, which could otherwise oxidize the sensitive silicon-nitrogen bonds. Following the formation of the silylated intermediate, the addition of sulfonyl chloride must be performed slowly to manage the exothermic nature of the reaction and prevent localized overheating. Finally, the fluorination step utilizes potassium fluoride in acetonitrile under reflux, a condition that maximizes the nucleophilic attack on the sulfur center to install the final fluorine atom. Detailed standardized synthesis steps are provided below to guide technical teams through this intricate process.
- Preparation of Sodium Sulfonamide via refluxing sulfonamide with sodium methoxide in methanol and ether.
- Silylation reaction using hexamethyldisilazane to form trimethylsilane perfluoroalkylsulfonimide sodium.
- Final fluorination using potassium fluoride in acetonitrile followed by recrystallization to isolate the potassium salt.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patented method offers compelling benefits that directly address the core concerns of supply chain heads regarding continuity and cost efficiency. By eliminating the dependency on scarce and heavily regulated toxic reagents, manufacturers can secure a more stable supply of raw materials that are less subject to geopolitical restrictions or sudden price volatility. The simplified equipment requirements mean that production can be distributed across a wider network of qualified chemical plants, reducing the risk of single-point failures in the supply chain. Furthermore, the reduced corrosivity of the process chemicals extends the lifespan of production assets, lowering the total cost of ownership for manufacturing facilities. These factors combine to create a resilient supply model capable of meeting the surging demand for battery materials without compromising on delivery schedules or quality assurance. For organizations focused on reducing lead time for high-purity electrolyte salts, this technology provides a clear pathway to accelerated market entry.
- Cost Reduction in Manufacturing: The substitution of highly expensive and dangerous fluorinating agents with more common sulfonyl chlorides results in substantial cost savings on raw material procurement and hazardous waste disposal. The process avoids the need for specialized alloy reactors resistant to extreme corrosion, allowing for the use of standard industrial equipment which significantly lowers capital investment barriers. Additionally, the efficient removal of volatile byproducts reduces the energy consumption associated with extensive purification and drying processes. These cumulative efficiencies translate into a more competitive pricing structure for the final electrolyte salt without sacrificing performance metrics. Such economic advantages are critical for maintaining margins in the highly price-sensitive battery materials market.
- Enhanced Supply Chain Reliability: Utilizing readily available reagents like potassium fluoride and hexamethyldisilazane ensures that production is not bottlenecked by the availability of exotic chemicals that often have long lead times. The robustness of the reaction conditions allows for flexible scheduling and easier scaling of production volumes to match fluctuating market demands. This flexibility is paramount for maintaining consistent inventory levels and fulfilling large-scale orders from automotive and electronics clients. Moreover, the lower hazard profile simplifies logistics and transportation, reducing regulatory burdens and shipping costs associated with dangerous goods. This reliability fosters stronger partnerships between chemical suppliers and battery manufacturers.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of persistent organic pollutants make this process highly amenable to green chemistry principles and environmental compliance standards. Scaling up from laboratory to commercial production involves minimal re-optimization, as the chemistry remains consistent regardless of batch size. The reduced generation of hazardous waste simplifies effluent treatment and aligns with corporate sustainability goals increasingly demanded by end-users. This environmental stewardship enhances the brand reputation of manufacturers and facilitates smoother regulatory approvals in key markets. It ensures long-term viability of the production line amidst tightening global environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this synthesis technology for potential partners and stakeholders. These insights are derived directly from the patent specifications and practical considerations for industrial application. Understanding these details is essential for evaluating the feasibility of integrating this material into existing battery supply chains. The answers reflect a commitment to transparency and technical excellence in the production of advanced chemical intermediates.
Q: What are the safety advantages of this synthesis method compared to traditional routes?
A: This method avoids highly toxic reagents like fluorosulfonic acid or bis(fluorosulfonyl)imide, utilizing sulfonyl chloride instead which significantly reduces corrosion risks and equipment requirements.
Q: How does this process impact the purity of the final electrolyte salt?
A: The process includes specific washing and recrystallization steps using dichloromethane that effectively remove byproducts, ensuring high-purity specifications suitable for sensitive lithium battery applications.
Q: Is this method scalable for industrial production of battery materials?
A: Yes, the use of moderate temperatures and readily available solvents like acetonitrile and ether facilitates commercial scale-up of complex fluorinated compounds without requiring exotic high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Asymmetric Bisfluorosulfonimide Potassium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance electrolyte salts play in the advancement of energy storage technologies and are committed to delivering superior chemical solutions. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric needs of global battery manufacturers with consistency. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of Asymmetric Bisfluorosulfonimide Potassium meets the exacting standards required for lithium-ion applications. Our infrastructure is designed to handle complex fluorinated chemistry safely and efficiently, providing a secure source of supply for your most demanding projects. We understand that reliability is just as important as quality in this sector, and our track record reflects our dedication to uninterrupted service.
We invite you to collaborate with us to explore how this advanced synthesis route can optimize your product formulation and reduce overall manufacturing costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate our capability to support your development timeline. Let us be your partner in driving the next generation of battery innovation through superior chemical engineering and supply chain excellence.
