Advanced Base-Catalyzed Synthesis of Polysubstituted Pyrazole Intermediates for Commercial Scale-Up
Advanced Base-Catalyzed Synthesis of Polysubstituted Pyrazole Intermediates for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally benign routes to construct nitrogen-containing heterocycles, which serve as critical scaffolds in drug discovery. A groundbreaking development detailed in patent CN113968819A introduces a highly efficient synthesis method for polysubstituted pyrazole compounds that fundamentally shifts the paradigm from harsh, multi-step processes to a streamlined, one-pot transformation. This innovation leverages a simple base-catalyzed cyclization between hydrazones and alpha,beta-unsaturated aldehydes, operating effectively at room temperature under ambient air conditions. By eliminating the need for expensive transition metal catalysts and extreme thermal inputs, this technology offers a compelling value proposition for manufacturers aiming to optimize their supply chains for high-purity pharmaceutical intermediates. The ability to simultaneously generate both 1,3,4-trisubstituted and 1,3,4,5-tetrasubstituted pyrazole derivatives from a single reaction setup represents a significant leap in atom economy and process efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of polysubstituted pyrazole skeletons has relied heavily on the Knorr pyrazole synthesis or post-synthetic structural modifications via electrophilic or nucleophilic substitutions. The Knorr method, while classic, typically necessitates the use of concentrated acids and elevated reaction temperatures, creating substantial conflicts with modern green chemistry principles and production safety standards. Furthermore, attempting to introduce specific functional groups onto an existing pyrazole ring through substitution reactions is often plagued by the unique steric structure and electronic effects of the heterocycle, leading to poor regioselectivity and limited yields. These conventional pathways frequently suffer from long synthetic routes, low atom economy, and complex operational procedures that involve hazardous reagents and difficult purification steps. For large-scale manufacturing, these factors translate into higher operational expenditures, increased waste generation, and significant challenges in maintaining consistent product quality across batches.
The Novel Approach
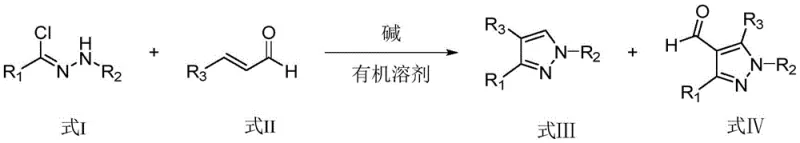
In stark contrast, the novel methodology disclosed in the patent utilizes readily available commodity chemicals—specifically hydrazones and olefinic aldehydes—to construct the pyrazole core under remarkably mild conditions. The reaction proceeds via a base-mediated mechanism where the hydrazone loses a proton to undergo Michael addition with the unsaturated aldehyde, followed by spontaneous intramolecular cyclization. This approach allows for the simultaneous construction of 1,3,4-trisubstituted and 1,3,4,5-tetrasubstituted pyrazole compounds in a single vessel, effectively doubling the output utility of the reaction batch. The process operates at room temperature (10-30°C) using environmentally friendly solvents like ethanol, removing the energy burden associated with heating or cooling systems. Moreover, the tetrasubstituted products feature a formyl group at the 4-position, which serves as a versatile handle for further structural modification, enabling the rapid introduction of diverse biological activities without requiring additional complex synthetic steps.
Mechanistic Insights into Base-Catalyzed Cyclization
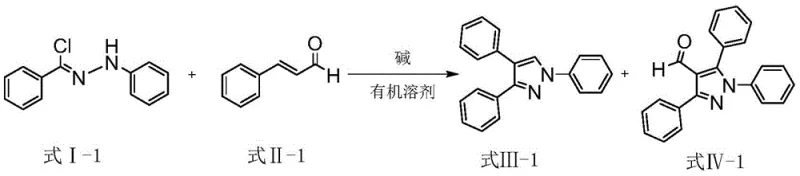
The core of this technological breakthrough lies in the elegant simplicity of the base-catalyzed cyclization mechanism, which bypasses the need for transition metal activation. The reaction initiates when a mild base, such as potassium carbonate (K2CO3), deprotonates the hydrazone species (Formula I), generating a nucleophilic intermediate capable of attacking the beta-carbon of the alpha,beta-unsaturated aldehyde (Formula II). This Michael addition step is crucial as it establishes the carbon-carbon bond necessary for ring closure. Following the addition, the intermediate undergoes an intramolecular condensation where the nitrogen atom attacks the carbonyl carbon, followed by dehydration to aromatize the pyrazole ring. This pathway is highly favorable thermodynamically and kinetically at room temperature, avoiding the high-energy barriers that typically require thermal activation in acid-catalyzed variants. The use of a simple inorganic base ensures that the reaction environment remains neutral to slightly basic, preventing the degradation of acid-sensitive functional groups that might be present on the aromatic rings.
From an impurity control perspective, this mechanism offers distinct advantages over traditional methods. Because the reaction occurs under mild conditions without strong acids or oxidants, the formation of polymeric byproducts or decomposition species is significantly minimized. The selectivity of the base catalysis ensures that the cyclization proceeds cleanly to form the desired pyrazole skeleton, with the primary variation being the degree of substitution at the 5-position (hydrogen vs. formyl group). This inherent selectivity simplifies the downstream purification process, often allowing for effective isolation of the target compounds via standard column chromatography or recrystallization. For R&D directors focused on purity profiles, this means a cleaner crude product with fewer unknown impurities, reducing the analytical burden and accelerating the timeline for regulatory filing. The robustness of the mechanism across various substituents (halogens, methoxy, naphthyl) further confirms its reliability for generating diverse chemical libraries.
How to Synthesize Polysubstituted Pyrazoles Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting is straightforward and requires minimal specialized equipment, making it highly accessible for process development teams. The standard protocol involves drying the reaction vessel to remove moisture, although the reaction tolerates ambient air, ensuring operational ease. The key reagents, N-phenylbenzohydrazone acid chloride and the chosen cinnamaldehyde derivative, are mixed with a stoichiometric amount of base (preferably K2CO3) in a solvent like ethanol. The mixture is then stirred at room temperature, with reaction progress monitored by thin-layer chromatography (TLC). Upon completion, the solvent is removed under reduced pressure, and the residue is purified using silica gel column chromatography with a petroleum ether and ethyl acetate gradient. This simplicity allows for rapid iteration and optimization, facilitating the quick generation of analogues for structure-activity relationship (SAR) studies. Detailed standardized synthesis steps follow below for technical reference.
- Prepare the reaction mixture by combining N-phenylbenzohydrazone acid chloride (Formula I) and potassium carbonate (K2CO3) in an organic solvent such as ethanol.
- Add the alpha,beta-unsaturated aldehyde (Formula II), such as cinnamaldehyde, to the reaction vessel under ambient air conditions without inert gas protection.
- Stir the mixture at room temperature until completion, then concentrate the solvent and purify the crude product via column chromatography to isolate both trisubstituted and tetrasubstituted pyrazoles.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this base-catalyzed synthesis route presents a multitude of strategic benefits that directly impact the bottom line and operational resilience. The primary advantage stems from the drastic simplification of the raw material portfolio; the process relies on cheap, easily obtained commodity chemicals rather than specialized, high-cost organometallic reagents. This shift not only lowers the direct material costs but also mitigates supply risk, as the precursors are widely available from multiple global vendors, ensuring continuity of supply even during market fluctuations. Furthermore, the elimination of transition metal catalysts removes the necessity for expensive metal scavenging resins and the associated validation testing required to prove residual metal levels are within ppm limits for pharmaceutical grades. This reduction in downstream processing steps translates to significant time savings and lower utility consumption.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound due to the replacement of expensive catalytic systems with inexpensive inorganic bases like potassium carbonate. Traditional methods often rely on precious metals or complex ligands that drive up the cost of goods sold (COGS); by contrast, this method utilizes reagents that are orders of magnitude cheaper. Additionally, the ability to run the reaction at room temperature eliminates the energy costs associated with heating reactors to high temperatures or maintaining cryogenic conditions for sensitive intermediates. The solvent system, favoring ethanol or other common organic solvents, is also cost-effective and easier to recover and recycle compared to exotic aprotic solvents. These factors combine to create a manufacturing process with a significantly leaner cost structure, allowing for better margin protection in competitive generic drug markets.
- Enhanced Supply Chain Reliability: Supply chain stability is bolstered by the use of robust, non-hazardous reagents that do not require special handling permits or cold-chain logistics. The starting materials, such as hydrazones and cinnamaldehydes, are stable solids or liquids with long shelf lives, reducing the risk of spoilage during storage and transport. Because the reaction does not require an inert atmosphere (nitrogen or argon protection), the operational complexity is reduced, minimizing the potential for human error or equipment failure related to gas lines and seals. This robustness ensures that production schedules can be met consistently, reducing lead times for high-purity pharmaceutical intermediates. The scalability of the process from gram to kilogram scale without re-optimization further guarantees that supply can be ramped up quickly to meet sudden increases in demand from downstream API manufacturers.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method aligns perfectly with modern sustainability goals. The absence of heavy metals means there is no toxic metal waste to treat or dispose of, significantly lowering waste management costs and environmental liability. The use of ethanol as a preferred solvent reduces the emission of volatile organic compounds (VOCs) compared to chlorinated solvents often used in traditional heterocycle synthesis. The high atom economy of the reaction, where most of the mass of the starting materials ends up in the product or benign byproducts, minimizes the E-factor (mass of waste per mass of product). This green profile facilitates easier regulatory approval and enhances the corporate sustainability rating of the manufacturing entity, which is increasingly important for partnerships with major multinational pharmaceutical companies committed to green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this polysubstituted pyrazole synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent literature, providing a clear understanding of the method's capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing pipelines. The responses cover aspects ranging from substrate compatibility to purification strategies, ensuring a comprehensive overview for decision-makers.
Q: How does this base-catalyzed method compare to traditional Knorr pyrazole synthesis?
A: Unlike the traditional Knorr synthesis which requires concentrated acid and high temperatures, this novel method operates at room temperature using cheap alkali catalysts like K2CO3. It eliminates the need for harsh acidic conditions, significantly improving production safety and environmental friendliness while allowing for the simultaneous construction of multiple pyrazole derivatives.
Q: What is the substrate scope for R1, R2, and R3 groups in this synthesis?
A: The method demonstrates excellent tolerance for various substituents. R1, R2, and R3 can be selected from substituted or unsubstituted phenyl, naphthyl, furyl, thienyl, and indolyl groups. Specific embodiments successfully utilize halogens (chlorine, fluorine, bromine) and methoxy groups, proving the route's versatility for generating diverse libraries of bioactive molecules.
Q: Does this process require expensive transition metal catalysts?
A: No, this process is entirely metal-free regarding transition metals. It utilizes inexpensive inorganic bases such as potassium carbonate (K2CO3) or organic bases like triethylamine. This absence of heavy metals simplifies the purification process, removes the need for costly metal scavenging steps, and ensures the final product meets stringent purity specifications for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Pyrazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this base-catalyzed synthesis route for the production of high-value heterocyclic intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific requirements of this chemistry, including rigorous QC labs capable of verifying stringent purity specifications and complete impurity profiling. We are committed to delivering high-purity polysubstituted pyrazoles that meet the exacting standards of the global pharmaceutical industry, leveraging our expertise in process optimization to maximize yield and minimize cost.
We invite you to collaborate with us to explore how this innovative synthesis method can enhance your drug development pipeline. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating exactly how switching to this greener, more efficient route can improve your margins. Please contact us today to request specific COA data for our available pyrazole intermediates and to discuss route feasibility assessments for your target molecules. Let us help you secure a reliable supply of critical building blocks while advancing your commitment to sustainable and cost-effective chemical manufacturing.
