Electrochemical Synthesis of Polysubstituted Pyrazoles for Commercial Pharmaceutical and Agrochemical Applications
Electrochemical Synthesis of Polysubstituted Pyrazoles for Commercial Pharmaceutical and Agrochemical Applications
The landscape of heterocyclic chemistry is undergoing a significant transformation driven by the urgent need for sustainable and efficient manufacturing processes. Patent CN114411178B introduces a groundbreaking electrochemical synthesis method for producing polysubstituted pyrazole compounds, a structural motif ubiquitous in high-value active pharmaceutical ingredients (APIs) and agrochemical agents. This innovation leverages constant current electrolysis in an undivided cell to facilitate the cyclization of β-enaminones and sulfonyl hydrazides, effectively replacing traditional stoichiometric oxidants and transition metal catalysts with electricity. By utilizing electrons as the primary oxidant, this technology addresses critical environmental and economic pain points associated with conventional pyrazole synthesis, offering a robust platform for the reliable pharmaceutical intermediate supplier seeking to modernize their production capabilities.
The significance of pyrazole derivatives cannot be overstated, as they serve as the core scaffold for numerous blockbuster drugs and crop protection agents, ranging from anti-inflammatory medications like Celecoxib to potent insecticides like Fipronil. 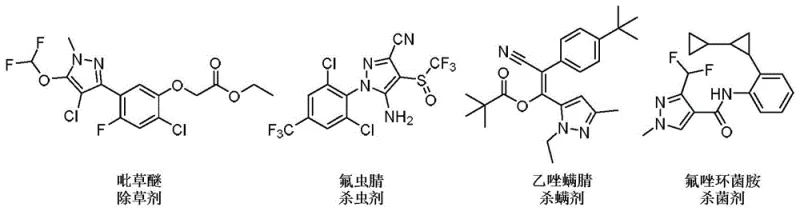 . The ability to construct these complex nitrogen-containing heterocycles under mild conditions with high atom economy represents a substantial leap forward in process chemistry. This patent details a versatile protocol that not only streamlines the synthetic route but also enhances the safety profile of the manufacturing process by avoiding hazardous reagents, thereby positioning it as a key technology for cost reduction in fine chemical manufacturing.
. The ability to construct these complex nitrogen-containing heterocycles under mild conditions with high atom economy represents a substantial leap forward in process chemistry. This patent details a versatile protocol that not only streamlines the synthetic route but also enhances the safety profile of the manufacturing process by avoiding hazardous reagents, thereby positioning it as a key technology for cost reduction in fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the pyrazole ring has relied heavily on the condensation of 1,3-dicarbonyl compounds with hydrazines or the cyclization of hydrazones. While these classical strategies are well-established, they are fraught with significant limitations that hinder large-scale industrial adoption. Most notably, these reactions frequently necessitate the use of strong chemical oxidants, expensive transition metal catalysts, and various additives to drive the reaction to completion and control regioselectivity. The reliance on heavy metal catalysts introduces severe challenges in downstream processing, particularly regarding the removal of trace metal impurities to meet stringent regulatory standards for pharmaceutical products. Furthermore, the generation of stoichiometric amounts of chemical waste from oxidants and catalysts creates a substantial environmental burden, increasing disposal costs and complicating compliance with increasingly rigorous global environmental regulations.
The Novel Approach
In stark contrast to these traditional methodologies, the electrochemical synthesis described in patent CN114411178B offers a paradigm shift by utilizing electric current to drive the oxidative cyclization. This novel approach operates in an undivided cell using readily available electrodes such as graphite, platinum, or nickel, eliminating the need for external chemical oxidants entirely. The reaction proceeds under mild conditions, typically between 20°C and 80°C, and demonstrates exceptional functional group tolerance, accommodating a wide array of substituents including halogens, alkoxy groups, and heteroaromatic rings. By removing the dependency on precious metal catalysts and hazardous oxidizing agents, this method drastically simplifies the workup procedure, often requiring only standard extraction and chromatography. This simplicity translates directly into operational efficiency and reduced production costs, making it an attractive option for the commercial scale-up of complex heterocyclic intermediates.
Mechanistic Insights into Electrochemical Oxidative Cyclization
The core of this innovative synthesis lies in the anodic oxidation of the reactants within the electrochemical cell. The process initiates with the oxidation of the sulfonyl hydrazide or the β-enaminone at the anode surface, generating reactive radical or cationic intermediates that facilitate the subsequent C-N bond formation. 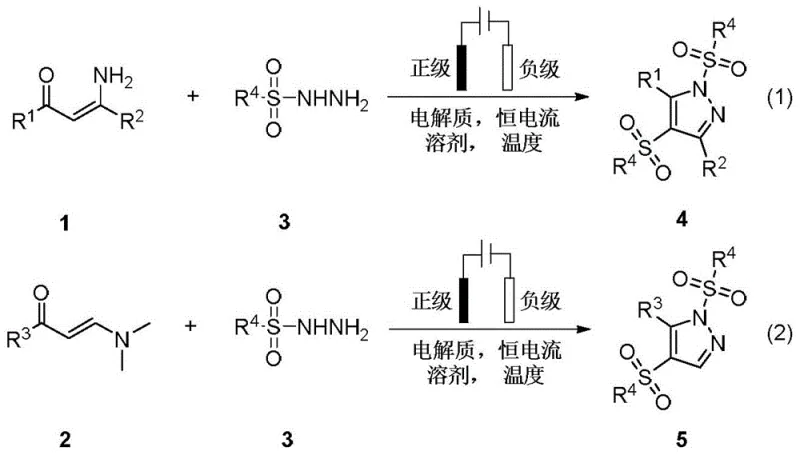 . Unlike thermal reactions that rely on high energy input to overcome activation barriers, electrochemical activation allows for precise control over the oxidation potential, minimizing side reactions and decomposition of sensitive functional groups. The supporting electrolyte, such as tetrabutylammonium salts or lithium perchlorate, plays a crucial role in maintaining conductivity and stabilizing the charged intermediates throughout the reaction cycle. This controlled environment ensures high chemical selectivity, favoring the formation of the desired pyrazole ring over potential byproducts.
. Unlike thermal reactions that rely on high energy input to overcome activation barriers, electrochemical activation allows for precise control over the oxidation potential, minimizing side reactions and decomposition of sensitive functional groups. The supporting electrolyte, such as tetrabutylammonium salts or lithium perchlorate, plays a crucial role in maintaining conductivity and stabilizing the charged intermediates throughout the reaction cycle. This controlled environment ensures high chemical selectivity, favoring the formation of the desired pyrazole ring over potential byproducts.
From an impurity control perspective, the absence of transition metals is a decisive advantage for R&D teams focused on purity profiles. Traditional metal-catalyzed routes often leave behind trace residues that require specialized scavenging resins or additional recrystallization steps, which inevitably reduce overall yield. In this electrochemical protocol, the primary byproducts are typically hydrogen gas at the cathode and protonated species, which are easily managed during the aqueous workup. The high selectivity observed across various substrates, including those with electron-withdrawing groups like trifluoromethyl or halogens, suggests a robust mechanism that is less susceptible to electronic perturbations than acid or base-catalyzed alternatives. This reliability is essential for ensuring batch-to-batch consistency, a critical factor for any reliable agrochemical intermediate supplier aiming to secure long-term contracts with multinational corporations.
How to Synthesize Polysubstituted Pyrazoles Efficiently
Implementing this electrochemical protocol requires careful attention to reaction parameters to maximize yield and efficiency. The general procedure involves dissolving the β-enaminone and sulfonyl hydrazide substrates in a suitable organic solvent such as dichloromethane, acetonitrile, or ethanol, along with a supporting electrolyte. The choice of electrode material can influence the reaction rate, with graphite and platinum showing excellent performance in the reported examples. Once the cell is assembled, a constant current is applied, typically ranging from 1 mA to 40 mA depending on the scale and specific substrate reactivity. The reaction is allowed to proceed for a defined period, usually between 1 to 48 hours, under controlled temperature conditions. Upon completion, the mixture is quenched and processed using standard extraction techniques to isolate the high-purity pyrazole product. For detailed standardized synthesis steps see the guide below.
- Combine beta-enaminone and sulfonyl hydrazide substrates with a supporting electrolyte such as lithium perchlorate or tetrabutylammonium salts in an organic solvent.
- Insert appropriate electrodes (e.g., graphite, platinum, or nickel) into an undivided cell and apply a constant current between 1 mA and 40 mA.
- Maintain the reaction temperature between 20°C and 80°C under an inert or air atmosphere for 1 to 48 hours until conversion is complete.
- Quench the reaction mixture with saturated brine, extract with dichloromethane, dry over anhydrous sodium sulfate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this electrochemical technology offers tangible strategic benefits beyond mere technical novelty. The elimination of expensive transition metal catalysts and stoichiometric oxidants directly impacts the bill of materials, leading to substantial cost savings in raw material procurement. Furthermore, the simplified purification process reduces the consumption of solvents and auxiliary materials required for metal scavenging, contributing to a leaner and more cost-effective manufacturing operation. The mild reaction conditions also imply lower energy consumption for heating or cooling compared to harsh thermal processes, further enhancing the economic viability of the route. These factors collectively contribute to significant cost reduction in fine chemical manufacturing, allowing companies to maintain competitive pricing while improving margins.
- Cost Reduction in Manufacturing: The removal of precious metal catalysts such as palladium or copper from the synthetic route eliminates a major cost driver and removes the need for expensive metal removal technologies. Additionally, the avoidance of hazardous chemical oxidants reduces the costs associated with safety handling, storage, and waste disposal. The overall process efficiency is improved by reducing the number of unit operations required for purification, which lowers labor and utility costs. This streamlined approach ensures that the production of high-purity polysubstituted pyrazoles remains economically sustainable even at large volumes.
- Enhanced Supply Chain Reliability: The starting materials, β-enaminones and sulfonyl hydrazides, are commercially available and chemically stable, reducing the risk of supply disruptions associated with specialized reagents. The robustness of the electrochemical method against variations in substrate electronics means that a single platform can be used to produce a diverse library of analogs without re-optimizing the entire process for each new compound. This flexibility allows for rapid response to market demands and accelerates the timeline for new product introductions. Consequently, this technology aids in reducing lead time for high-purity agrochemical intermediates, ensuring a steady flow of materials to downstream formulation plants.
- Scalability and Environmental Compliance: The patent explicitly demonstrates the feasibility of gram-scale synthesis, indicating a clear path towards kilogram and ton-scale production using flow electrochemical reactors or larger batch cells. The green nature of the process, characterized by the absence of toxic heavy metals and reduced chemical waste, aligns perfectly with corporate sustainability goals and regulatory requirements. This environmental compliance minimizes the risk of regulatory penalties and enhances the brand reputation of the manufacturer as a responsible producer. The ability to scale this green chemistry solution ensures long-term supply continuity and meets the growing demand for eco-friendly pharmaceutical and agrochemical ingredients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrochemical synthesis of pyrazoles, based on the detailed data provided in the patent literature. Understanding these aspects is crucial for technical teams evaluating the feasibility of integrating this method into their existing production workflows. The answers reflect the specific advantages and operational parameters outlined in the intellectual property, providing a clear picture of the technology's capabilities.
Q: What are the primary advantages of this electrochemical method over traditional pyrazole synthesis?
A: Unlike conventional methods that require harsh oxidants, transition metal catalysts, and additives, this electrochemical approach uses electrons as clean reagents. This eliminates heavy metal contamination risks, simplifies downstream purification, and significantly reduces waste generation, aligning with green chemistry principles.
Q: Does this synthesis method support a wide range of functional groups?
A: Yes, the patent demonstrates excellent functional group tolerance. The method successfully accommodates various substituents including alkyl, aryl, naphthyl, furan, and thiophene rings, as well as electron-withdrawing groups like fluorine, chlorine, and trifluoromethyl, making it versatile for diverse drug discovery campaigns.
Q: Is this electrochemical process scalable for industrial production?
A: The patent explicitly states that the reaction can achieve gram-scale synthesis with high efficiency. The use of simple undivided cells and readily available starting materials suggests strong potential for commercial scale-up, offering a viable pathway for reducing lead time in high-purity intermediate manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Pyrazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in delivering high-quality chemical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN114411178B can be successfully translated into robust industrial processes. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of polysubstituted pyrazole meets the exacting standards required by the global pharmaceutical and agrochemical industries. Our infrastructure is designed to handle complex heterocyclic chemistry with precision, safety, and efficiency.
We invite you to collaborate with us to leverage this advanced technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our electrochemical capabilities can optimize your supply chain and enhance your product portfolio. Let us be your partner in driving innovation and efficiency in fine chemical manufacturing.
