Scalable Synthesis of Fall Webworm Pheromone Intermediate via Nickel Catalysis
Introduction to Advanced Agrochemical Intermediate Synthesis
The global demand for environmentally friendly pest control solutions has intensified the search for efficient synthesis routes for insect sex pheromones. Patent CN113004223A discloses a groundbreaking method for synthesizing (3Z,6Z,9S,10R)-9,10-epoxy-3,6-heneicosadiene, the primary active ingredient of the fall webworm sex pheromone. This compound plays a pivotal role in integrated pest management strategies by disrupting the mating cycles of Hypophantria cunea, a devastating quarantine pest. The structural complexity of this molecule, featuring both specific Z-configured double bonds and a chiral epoxy center, presents significant synthetic challenges that this new methodology effectively addresses. 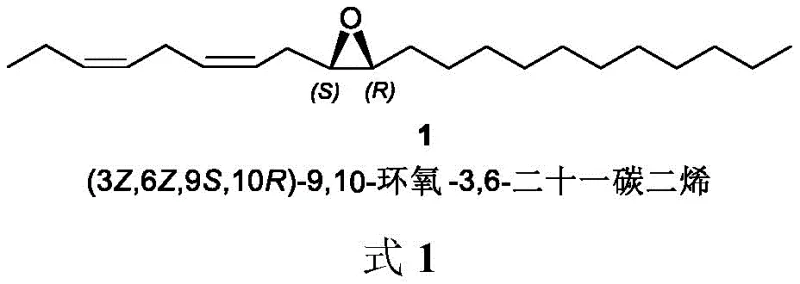 By leveraging a novel nickel-catalyzed hydrogenation strategy, the process achieves high stereoselectivity and operational simplicity, positioning it as a superior alternative for reliable agrochemical intermediate suppliers seeking to optimize their manufacturing portfolios.
By leveraging a novel nickel-catalyzed hydrogenation strategy, the process achieves high stereoselectivity and operational simplicity, positioning it as a superior alternative for reliable agrochemical intermediate suppliers seeking to optimize their manufacturing portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex epoxy-diene pheromones has relied on methodologies that suffer from significant drawbacks in terms of yield, selectivity, and environmental impact. Traditional approaches such as the alkynyl lithium method often require harsh reaction conditions and exhibit poor control over stereochemistry during the coupling steps. Similarly, the dialkyl copper lithium method, while effective for carbon chain extension, involves the use of moisture-sensitive organometallic reagents that complicate handling and scale-up procedures. Furthermore, the Wittig coupling method, frequently employed to construct Z-type double bonds, generates stoichiometric amounts of triphenylphosphine oxide waste, creating substantial downstream purification burdens and increasing the overall cost of goods. These legacy processes often struggle to maintain the rigorous purity specifications required for bioactive agrochemicals, leading to inconsistent batch quality and reduced field efficacy.
The Novel Approach
The innovative route described in the patent overcomes these hurdles by introducing a streamlined sequence centered around nickel acetate and sodium borohydride catalysis. This approach utilizes 1-bromoundecane as a cost-effective starting material, reacting it with lithium propargyl alcohol to initiate chain growth. A critical breakthrough is the application of nickel-catalyzed hydrogenation to convert alkynes into Z-alkenes, a transformation that traditionally requires expensive palladium-based catalysts like Lindlar's catalyst. 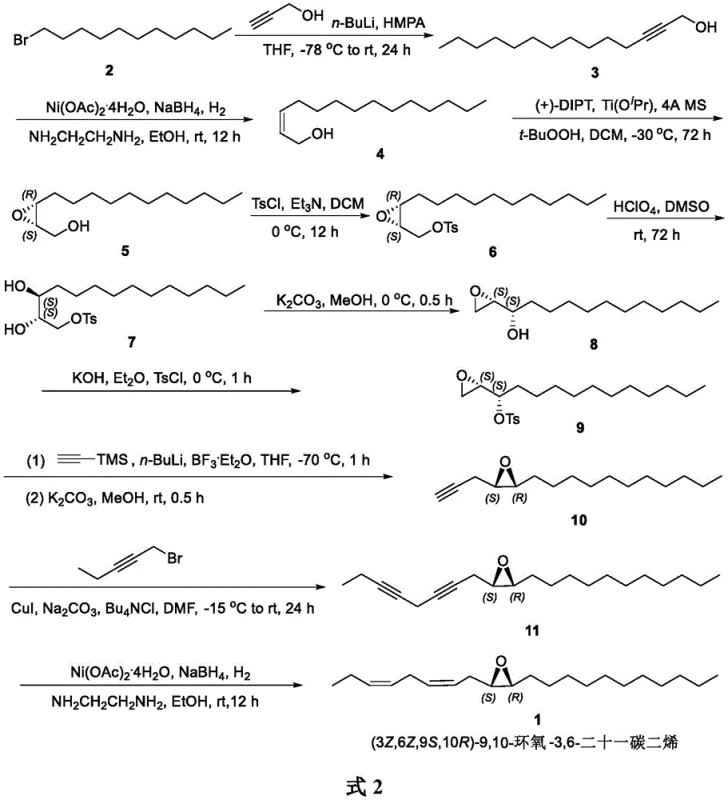 This novel catalytic system operates under mild conditions, significantly reducing energy consumption and safety risks associated with high-pressure hydrogenation. The integration of Sharpless asymmetric epoxidation ensures precise control over the chiral centers, delivering the target molecule with high optical purity. This holistic strategy not only simplifies the synthetic pathway but also enhances the overall atom economy, making it an ideal candidate for cost reduction in agrochemical intermediate manufacturing.
This novel catalytic system operates under mild conditions, significantly reducing energy consumption and safety risks associated with high-pressure hydrogenation. The integration of Sharpless asymmetric epoxidation ensures precise control over the chiral centers, delivering the target molecule with high optical purity. This holistic strategy not only simplifies the synthetic pathway but also enhances the overall atom economy, making it an ideal candidate for cost reduction in agrochemical intermediate manufacturing.
Mechanistic Insights into Nickel-Catalyzed Z-Selective Hydrogenation
The core mechanistic advantage of this synthesis lies in the unique behavior of the nickel boride catalyst generated in situ from nickel acetate and sodium borohydride. Unlike heterogeneous palladium catalysts that may lead to over-reduction or isomerization, this homogeneous-like system facilitates the syn-addition of hydrogen across the triple bond with exceptional Z-selectivity. The presence of ethylenediamine as a ligand further modulates the catalyst's activity, preventing the reduction of the sensitive epoxy group while ensuring complete conversion of the alkyne functionality. This chemoselectivity is paramount, as the preservation of the epoxide ring is essential for the biological activity of the final pheromone. The mechanism likely involves the formation of a nickel-hydride species that coordinates with the alkyne substrate, followed by sequential hydride transfer to generate the cis-alkene geometry without compromising the adjacent stereocenters.
Furthermore, the establishment of chirality via Sharpless asymmetric epoxidation is a masterstroke in impurity control. By utilizing L-diisopropyl tartrate and titanium tetraisopropoxide, the process dictates the absolute configuration of the epoxy alcohol intermediate with high enantiomeric excess, reported up to 90% ee in the examples. This early introduction of chirality allows for the subsequent chemical transformations, such as tosylation and nucleophilic substitution, to proceed with retention or predictable inversion of configuration. The rigorous control over stereochemistry minimizes the formation of diastereomeric impurities that could otherwise act as antagonists in the field, thereby ensuring the high potency of the final product. This level of precision is critical for meeting the stringent quality standards demanded by regulatory bodies for pheromone-based pest control agents.
How to Synthesize (3Z,6Z,9S,10R)-9,10-epoxy-3,6-heneicosadiene Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing this high-value intermediate. The process begins with the alkylation of propargyl alcohol followed by selective hydrogenation to set the first Z-double bond. Subsequent steps involve the installation of the chiral epoxide and the extension of the carbon chain through iterative alkynylation reactions. The final step employs the same nickel-catalyzed hydrogenation system to install the second Z-double bond, demonstrating the versatility of the catalytic method. For detailed operational parameters, reagent ratios, and workup procedures, please refer to the standardized synthesis guide below which encapsulates the critical process controls identified in the patent documentation.
- React 1-bromoundecane with lithium propargyl alcohol followed by nickel-catalyzed hydrogenation to form (Z)-2-tetradecen-1-ol.
- Perform Sharpless asymmetric epoxidation to establish chirality, followed by tosylation and perchloric acid ring opening.
- Extend the carbon chain via alkynylation with lithium trimethylsilylacetylene and 1-bromo-1-pentyne, concluding with final nickel-catalyzed hydrogenation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers tangible strategic benefits beyond mere technical feasibility. The shift towards a nickel-based catalytic system represents a significant opportunity for cost optimization, as nickel salts are substantially more abundant and less expensive than precious metals like palladium or platinum. This substitution directly impacts the raw material cost structure, allowing for more competitive pricing in the volatile agrochemical market. Additionally, the mild reaction conditions reduce the need for specialized high-pressure equipment and extreme temperature controls, lowering capital expenditure requirements for manufacturing facilities. The simplified purification processes, resulting from higher selectivity and fewer byproducts, further contribute to operational efficiency and reduced waste disposal costs.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts and the reduction of stoichiometric waste streams significantly lower the variable costs associated with production. By avoiding the generation of heavy metal residues, the downstream purification burden is drastically simplified, removing the need for costly scavenging resins or complex filtration steps. This streamlined workflow translates into substantial cost savings per kilogram of finished product, enhancing the overall margin profile for manufacturers. Furthermore, the use of readily available starting materials like 1-bromoundecane ensures stable input pricing, shielding the supply chain from the volatility often seen with specialized organometallic reagents.
- Enhanced Supply Chain Reliability: The robustness of the nickel-catalyzed hydrogenation process ensures consistent batch-to-batch quality, which is essential for maintaining long-term contracts with agrochemical formulators. The method's tolerance to functional groups and its operation under ambient pressure conditions minimize the risk of production delays caused by equipment failure or safety incidents. This reliability is crucial for just-in-time delivery models, ensuring that critical pest control seasons are supported by uninterrupted material flow. The scalability of the process means that supply can be rapidly ramped up to meet seasonal demand spikes without compromising on the stringent purity specifications required for bioactive efficacy.
- Scalability and Environmental Compliance: From an environmental perspective, the replacement of toxic reagents and the reduction of hazardous waste align perfectly with modern green chemistry principles. The process generates fewer organic solvents and heavy metal wastes, simplifying compliance with increasingly strict environmental regulations. This eco-friendly profile not only reduces the liability associated with waste treatment but also enhances the brand value of the final agrochemical product as a sustainable solution. The ease of amplification from laboratory to pilot and commercial scale ensures that the technology can be deployed globally, supporting a resilient and distributed supply network for essential crop protection ingredients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of this pheromone intermediate. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear understanding of the technology's capabilities. Understanding these details is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing infrastructure.
Q: What is the key advantage of the nickel-catalyzed hydrogenation in this process?
A: The use of nickel acetate/sodium borohydride allows for the construction of Z-type carbon-carbon double bonds under mild conditions, avoiding the high costs and toxicity associated with traditional Lindlar catalysts while maintaining high stereoselectivity.
Q: How is the chiral center established in the synthesis of the pheromone intermediate?
A: The chiral epoxy center is introduced early in the synthesis via Sharpless asymmetric epoxidation using L-diisopropyl tartrate and titanium tetraisopropoxide, achieving high enantiomeric excess (up to 90% ee) which is critical for biological activity.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the patent highlights that the method features mild reaction conditions, ease of operation, and scalability. The avoidance of extremely sensitive reagents and the use of robust catalytic systems make it highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (3Z,6Z,9S,10R)-9,10-epoxy-3,6-heneicosadiene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the development of next-generation green pesticides. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex synthetic routes like the nickel-catalyzed pheromone synthesis can be successfully translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee the stereochemical integrity and chemical purity of every batch. Our commitment to quality assurance means that clients can rely on us for consistent supply of this vital agrochemical building block, supporting their R&D and commercialization efforts without interruption.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific volume requirements. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that evaluates the economic benefits of switching to this nickel-catalyzed process within your supply chain. We encourage you to request specific COA data and route feasibility assessments to verify the compatibility of our manufacturing capabilities with your project timelines. Let us help you secure a sustainable and cost-effective supply of high-performance pheromone intermediates for the global market.
