Scalable Synthesis of Fall Webworm Sex Pheromone Intermediates via Sharpless Asymmetric Dihydroxylation
The agricultural sector continuously demands more efficient and environmentally sustainable solutions for pest management, particularly for invasive species like the fall webworm (Hyphantria cunea). A pivotal advancement in this domain is detailed in patent CN112661725A, which discloses a robust synthetic method for producing the sex pheromone (3Z,6Z,9S,10R)-9,10-epoxy-3,6-heneicosadiene. This specific molecule serves as a potent attractant for monitoring and controlling fall webworm populations, offering a green alternative to broad-spectrum chemical insecticides. The patented methodology represents a significant departure from historical synthesis routes that often suffered from low yields, typically around 7%, and the instability of polyacetylene intermediates. By leveraging propiolic alcohol as a foundational building block and integrating a Sharpless asymmetric dihydroxylation as the stereo-defining step, this process achieves a total reaction yield of approximately 10 percent while maintaining exceptional enantiomeric excess. For R&D directors and procurement specialists in the agrochemical industry, this innovation signals a shift towards more reliable supply chains for high-value pheromone intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex lepidopteran sex pheromones has been plagued by significant chemical challenges that hindered commercial viability. Early approaches, such as those developed by K. Mori in the 1980s, relied on polyacetylene intermediates which were notoriously unstable and prone to isomerization or oxidation, leading to poor reproducibility and low overall yields. Furthermore, subsequent attempts, including those described in Chinese patent CN 101798293A, utilized glycerol as a starting material but introduced a critical bottleneck: the sulfoesterification of key epoxy-alcohol intermediates using trifluoromethyl sulfonic anhydride. This reagent is highly reactive and possesses strong oxidizing properties, making the reaction extremely difficult to control. The resulting trifluoromethyl sulfonates are so active that they frequently undergo unwanted elimination or substitution side reactions, often resulting in the complete failure to isolate the desired sulfoesterified product. These technical hurdles not only inflated production costs due to material loss but also created severe safety hazards associated with handling aggressive fluorinating agents on an industrial scale.
The Novel Approach
The methodology outlined in CN112661725A elegantly circumvents these historical pitfalls by redesigning the synthetic logic to avoid hazardous sulfoesterification entirely. Instead of relying on unstable polyacetylenes or aggressive sulfonating agents, the new route utilizes a convergent strategy centered on the Sharpless asymmetric dihydroxylation reaction. This key transformation allows for the precise installation of the (9S,10R) chiral centers under mild, controllable conditions using AD-mix-alpha. By eliminating the step involving trifluoromethyl sulfonic anhydride, the process drastically reduces the formation of elimination by-products and simplifies the purification workflow. The reaction sequence proceeds through stable intermediates, such as protected alkynes and epoxides, which can be handled with standard laboratory equipment. This strategic pivot not only enhances the safety profile of the manufacturing process but also ensures a more consistent output of the biologically active isomer, addressing the core pain points of yield and purity that have long困扰 ed pheromone synthesis.
Mechanistic Insights into Sharpless Asymmetric Dihydroxylation and Stereoselective Hydrogenation
The cornerstone of this synthetic achievement is the application of the Sharpless asymmetric dihydroxylation in step 5, which dictates the absolute stereochemistry of the final pheromone molecule. In this critical transformation, the alkene precursor (compound 5) is treated with AD-mix-alpha at 0°C, facilitating the syn-addition of two hydroxyl groups across the double bond. The chiral ligand within the AD-mix creates a specific steric environment that directs the osmium tetroxide catalyst to approach the olefin from a defined face, thereby establishing the (9S,10R) configuration with high fidelity. This step is crucial because the biological activity of the fall webworm sex pheromone is strictly dependent on this specific stereochemical arrangement; any deviation would render the attractant ineffective in the field. Following the dihydroxylation, the subsequent conversion to the epoxide (compound 7) and the nucleophilic opening with trimethylsilyl acetylene preserve this chirality, ensuring that the stereochemical integrity established early in the sequence is maintained throughout the ten-step synthesis.
Another vital mechanistic feature is the final stereoselective hydrogenation performed in step 10, which secures the geometric configuration of the double bonds. The precursor compound 10 contains internal alkyne functionalities that must be reduced to cis-alkenes (Z-configuration) to match the natural pheromone structure. The patent specifies the use of either a P-2 nickel catalyst or a Lindlar catalyst under a hydrogen atmosphere. These catalysts are uniquely capable of halting the reduction at the alkene stage, preventing over-reduction to the saturated alkane. The mechanism involves the adsorption of the alkyne onto the catalyst surface, where hydrogen is delivered syn to the triple bond, inherently favoring the formation of the Z-isomer. This precision is paramount, as the (3Z,6Z) geometry is essential for receptor binding in the male fall webworm. The combination of high enantioselectivity from the dihydroxylation and high geometric selectivity from the hydrogenation results in a final product with 95% e.e., demonstrating the robustness of the catalytic systems employed.
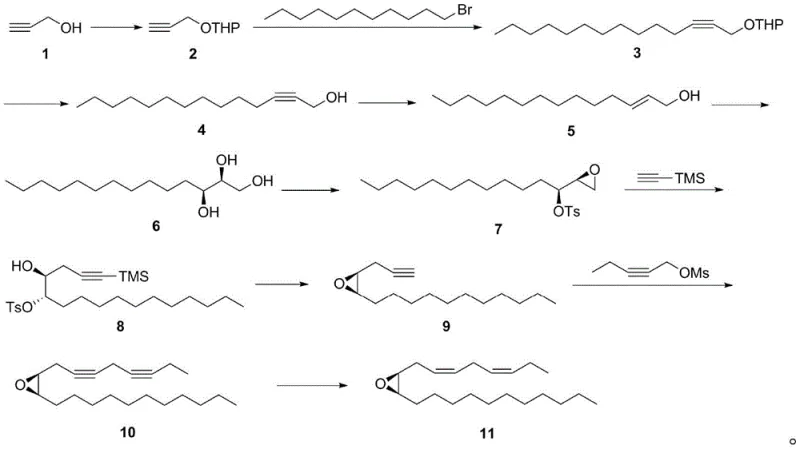
How to Synthesize (3Z,6Z,9S,10R)-9,10-epoxy-3,6-heneicosadiene Efficiently
The execution of this synthesis requires careful attention to reaction conditions, particularly during the coupling and reduction phases. The process begins with the protection of propiolic alcohol using 3,4-dihydropyran (DHP) in dichloromethane, followed by a low-temperature coupling with 1-bromoundecane using n-butyllithium to build the carbon backbone. Subsequent deprotection and reduction with lithium aluminum hydride yield the allylic alcohol substrate required for the asymmetric dihydroxylation. Once the chiral diol is formed, it is converted into an epoxide via tosylation and base-mediated cyclization. The chain is further extended through nucleophilic attack by trimethylsilyl acetylene, followed by deprotection and a second coupling with 2-pentyne-1-ol mesylate. The detailed standardized operating procedures for each of these ten steps, including specific molar ratios, solvent choices, and workup protocols, are provided in the technical guide below.
- Protection of propiolic alcohol with DHP followed by coupling with 1-bromoundecane to extend the carbon chain.
- Execution of Sharpless asymmetric dihydroxylation using AD-mix-alpha to establish the critical (9S,10R) chiral centers.
- Final stereoselective hydrogenation using Lindlar or P-2 Nickel catalyst to secure the (3Z,6Z) double bond geometry.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible benefits regarding cost structure and operational reliability. The primary advantage lies in the elimination of trifluoromethyl sulfonic anhydride, a reagent that is not only expensive but also requires specialized handling and disposal protocols due to its corrosive nature. By removing this hazardous step, the manufacturing process becomes significantly safer and more compliant with increasingly stringent environmental regulations. Furthermore, the avoidance of complex side reactions means that less raw material is wasted on by-products, leading to a more efficient utilization of starting materials like propiolic alcohol and 1-bromoundecane. This efficiency translates directly into a more predictable cost of goods sold (COGS), allowing for better budget forecasting and pricing stability for downstream agrochemical formulators.
- Cost Reduction in Manufacturing: The streamlined nature of this synthesis eliminates the need for expensive and difficult-to-handle fluorinating agents, which substantially lowers the cost of reagents and waste treatment. Additionally, the higher overall yield compared to legacy methods means that less feedstock is required to produce the same amount of active pheromone, driving down the unit cost. The use of common organic solvents such as methanol, THF, and dichloromethane further ensures that solvent recovery and recycling can be implemented easily, contributing to long-term operational savings without compromising on quality.
- Enhanced Supply Chain Reliability: By utilizing readily available starting materials like propiolic alcohol and avoiding reagents that are subject to strict regulatory controls or supply shortages, this method ensures a more resilient supply chain. The robustness of the Sharpless dihydroxylation step means that production batches are less likely to fail due to sensitivity issues, guaranteeing consistent delivery schedules. This reliability is critical for agrochemical companies that need to stockpile pheromone dispensers ahead of the pest breeding season, as it mitigates the risk of production delays that could impact market availability.
- Scalability and Environmental Compliance: The mild reaction conditions, often ranging from ice bath temperatures to ambient heat, make this process highly amenable to scale-up from pilot plant to commercial tonnage. Unlike processes requiring cryogenic conditions or high-pressure reactors, this route can be executed in standard glass-lined or stainless steel reactors, reducing capital expenditure requirements. Moreover, the reduction in hazardous waste generation aligns with global sustainability goals, making the final product more attractive to environmentally conscious buyers and regulatory bodies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this fall webworm sex pheromone. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on the process capabilities and product specifications. Understanding these details is essential for partners looking to integrate this intermediate into their broader pest management portfolios.
Q: How does this synthetic route improve upon previous methods for fall webworm pheromone production?
A: Unlike prior art which utilized unstable polyacetylene intermediates or harsh trifluoromethyl sulfonic anhydride reactions prone to side products, this method employs a mild Sharpless asymmetric dihydroxylation. This eliminates difficult-to-control sulfoesterification steps, significantly reducing impurities and improving overall process safety and controllability.
Q: What is the optical purity achievable with this manufacturing process?
A: The process utilizes AD-mix-alpha in the key dihydroxylation step, ensuring high enantioselectivity. The final product, (3Z,6Z,9S,10R)-9,10-epoxy-3,6-heneicosadiene, consistently achieves an optical purity of approximately 95% e.e., which is critical for effective biological activity in pest control applications.
Q: Is this synthesis suitable for large-scale commercial production?
A: Yes, the route avoids hazardous reagents like triflic anhydride and uses standard organic solvents such as dichloromethane, THF, and methanol. The reaction conditions are mild (often 0°C to room temperature), making the process highly adaptable for scale-up from laboratory to multi-ton commercial manufacturing without significant safety barriers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (3Z,6Z,9S,10R)-9,10-epoxy-3,6-heneicosadiene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity pheromones play in modern integrated pest management strategies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the fluctuating demands of the global agrochemical market. We are committed to delivering (3Z,6Z,9S,10R)-9,10-epoxy-3,6-heneicosadiene with stringent purity specifications, leveraging our rigorous QC labs to verify optical purity and geometric isomer ratios before shipment. Our facility is equipped to handle the specific solvent systems and catalytic requirements of this synthesis, guaranteeing a supply of material that performs consistently in field applications.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthetic route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can evaluate the economic benefits of switching to this safer, more efficient manufacturing method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume requirements, ensuring a seamless integration of this high-value intermediate into your production schedule.
