Advanced Berberine Purification Technology: Scalable Acetone Adduct Crystallization for Global API Markets
Advanced Berberine Purification Technology: Scalable Acetone Adduct Crystallization for Global API Markets
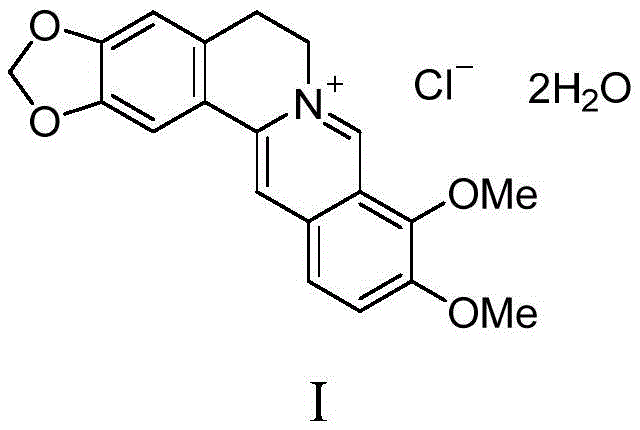
The pharmaceutical industry continuously seeks robust purification strategies to ensure the safety and efficacy of active pharmaceutical ingredients, particularly for widely used alkaloids like berberine. Patent CN111205285B introduces a groundbreaking purification methodology that addresses the longstanding challenges associated with obtaining ultra-high purity berberine and its salts. This technology leverages a unique salt-acetone adduct-salt conversion mechanism to effectively strip away complex impurity profiles that traditional methods fail to remove. By transforming the crude material into a distinct crystalline acetone adduct intermediate, manufacturers can achieve a level of purity where total impurities are maintained below 0.2% and single impurities are suppressed to less than 0.1%. This technical breakthrough is critical for meeting stringent global regulatory standards and ensuring the therapeutic reliability of berberine-based medications used in treating gastrointestinal disorders and bacterial infections.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of berberine hydrochloride and related salts has relied heavily on iterative solvent recrystallization techniques, which are fraught with significant operational and economic inefficiencies. These conventional processes often suffer from low separation efficiency, necessitating multiple recrystallization cycles that drastically reduce overall yield and increase production time. Furthermore, the reproducibility of these methods is notoriously poor, as the outcome is strongly correlated with the variable source and initial purity of the starting botanical extracts or synthetic crude materials. In many instances, crude products containing diverse structural analogs and degradation byproducts simply cannot reach the required pharmacopoeial standards through simple recrystallization, leading to high batch failure rates and substantial material waste. The inability to consistently reduce single impurity levels below the critical 0.1% threshold poses a major risk for drug safety evaluations and toxicological studies.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a transformative chemical strategy involving the formation of a berberine acetone adduct. This method fundamentally alters the physicochemical properties of the target molecule during the purification phase, allowing for a highly selective crystallization process. As illustrated in the reaction scheme below, the process involves converting the raw berberine salt into a specific acetone adduct, which precipitates out of the solution while leaving the vast majority of impurities dissolved in the mother liquor. This intermediate adduct acts as a purification gatekeeper; once isolated, it is converted back into the desired berberine salt, resulting in a product of exceptional purity. This salt-adduct-salt cycle effectively bypasses the thermodynamic limitations of direct recrystallization, offering a reliable pathway to high-quality API production.
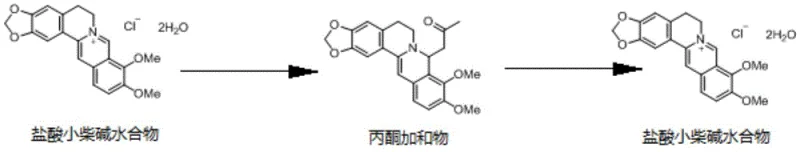
Mechanistic Insights into Acetone Adduct Crystallization
The core mechanism driving this purification success lies in the specific molecular recognition and lattice energy differences between the berberine acetone adduct and potential impurities. When acetone is introduced to the reaction mixture containing free berberine base, it forms a stable solvate or adduct crystal structure that is thermodynamically favored under the specified conditions (0-30°C). The structural integrity of this adduct, as depicted in the molecular diagram, creates a rigid crystalline environment that sterically hinders the incorporation of foreign molecules or structurally similar alkaloids. Most impurities present in the crude starting material lack the specific functional group arrangement or electronic properties required to co-crystallize with the berberine-acetone complex, causing them to remain in the liquid phase. This phenomenon effectively decouples the target molecule from the impurity matrix, enabling a level of separation that is unattainable through standard solubility-based recrystallization alone.
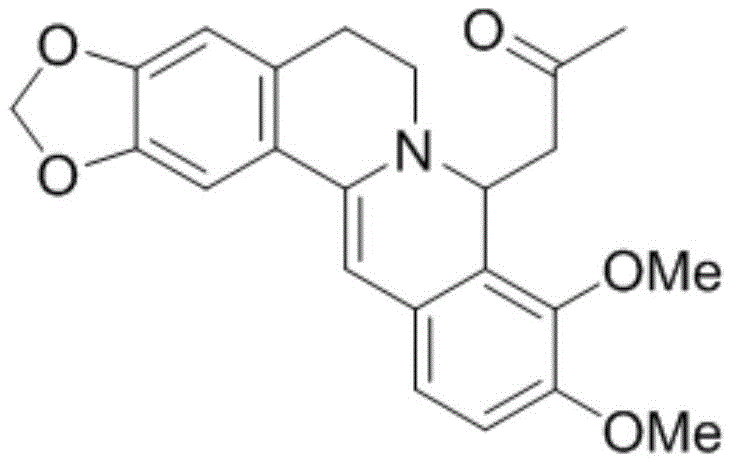
Furthermore, the control of impurity levels is enhanced by the reversibility of the adduct formation. Once the pure adduct crystals are isolated via solid-liquid separation, they are subjected to an acidification step that regenerates the berberine salt. Because the impurities were physically excluded during the adduct crystallization phase, the final regeneration step yields a salt with a dramatically cleaner impurity profile. The patent data indicates that this mechanism consistently delivers products with single impurity contents below 0.1%, a specification that is critical for modern pharmaceutical compliance. The use of mild reaction conditions, such as room temperature neutralization and low-temperature crystallization, ensures that no new degradation products are formed during the purification process, preserving the chemical integrity of the sensitive isoquinoline backbone.
How to Synthesize High-Purity Berberine Efficiently
The synthesis of high-purity berberine via this patented route involves a streamlined three-step sequence that is amenable to both laboratory optimization and industrial scale-up. The process begins with the dissociation of the crude berberine salt using a base in a polar solvent, followed by the critical addition of acetone to precipitate the purified adduct. Finally, the adduct is treated with the appropriate acid to regenerate the final salt form. This methodology eliminates the need for complex chromatographic separations or hazardous reagents, relying instead on fundamental crystallization principles driven by solvent polarity and molecular geometry. For detailed operational parameters, including specific molar ratios, temperature ramps, and stirring rates, please refer to the standardized synthesis guide provided below.
- Dissolve crude berberine salt (e.g., sulfate or hydrochloride) in a solvent such as water or methanol and treat with a base like sodium hydroxide to liberate free berberine.
- Introduce acetone to the reaction mixture under controlled temperatures (0-30°C) to induce crystallization of the specific berberine acetone adduct, leaving impurities in the mother liquor.
- Isolate the purified acetone adduct via filtration, suspend in water, and react with the corresponding acid (e.g., hydrochloric acid) to regenerate the final high-purity berberine salt.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this purification technology offers profound advantages for procurement managers and supply chain directors focused on cost efficiency and reliability. By replacing inefficient multi-cycle recrystallization with a single, high-yield adduct formation step, the process significantly reduces solvent consumption and energy usage associated with repeated heating and cooling cycles. The reliance on commodity chemicals such as acetone, water, and methanol ensures that raw material costs remain low and supply is secure, mitigating the risks associated with sourcing exotic or expensive reagents. Additionally, the dramatic improvement in yield and purity reduces the volume of waste generated per kilogram of product, aligning with increasingly strict environmental regulations and lowering disposal costs. This operational efficiency translates directly into a more competitive cost structure for the final API, allowing manufacturers to offer high-quality berberine derivatives at sustainable price points.
- Cost Reduction in Manufacturing: The elimination of multiple recrystallization steps and the use of inexpensive, readily available solvents like acetone and water drastically simplify the production workflow. This simplification reduces labor hours, equipment occupancy time, and utility consumption, leading to substantial overall cost savings in API manufacturing without compromising on quality standards.
- Enhanced Supply Chain Reliability: Because the process does not depend on scarce catalysts or specialized reagents, the supply chain is inherently more robust and less susceptible to market fluctuations. The ability to process various crude starting materials, including sulfates and hydrochlorides of varying initial purities, provides flexibility in sourcing raw materials, ensuring continuous production even when specific feedstock qualities vary.
- Scalability and Environmental Compliance: The mild operating conditions and aqueous workup procedures make this process highly scalable from pilot plant to commercial tonnage. The reduction in organic solvent waste and the high recovery rate of the target molecule contribute to a greener manufacturing profile, facilitating easier regulatory approval and reducing the environmental footprint of the production facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this berberine purification technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the method's capabilities and limitations. Understanding these details is essential for R&D teams evaluating process transfer and procurement teams assessing supplier capabilities.
Q: What is the primary advantage of the acetone adduct method over traditional recrystallization?
A: The acetone adduct method fundamentally changes the crystal lattice structure during purification, allowing for the exclusion of structurally similar impurities that co-crystallize in traditional solvent recrystallization, thereby achieving single impurity levels below 0.1%.
Q: Can this purification method be scaled for industrial production?
A: Yes, the process utilizes commodity solvents like acetone, water, and methanol, and operates at mild temperatures (0-30°C), making it highly suitable for large-scale commercial manufacturing without requiring exotic reagents or extreme conditions.
Q: What represents the critical quality attribute (CQA) improvement in this patent?
A: The critical improvement is the reduction of total impurities to less than 0.2% and single impurities to less than 0.1%, which significantly exceeds the current pharmacopoeia requirement of 4% total impurities, ensuring higher safety profiles for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Berberine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of purity and consistency in the production of active pharmaceutical ingredients like berberine hydrochloride. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated acetone adduct purification method can be seamlessly transferred to large-scale manufacturing environments. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify that every batch meets the <0.1% single impurity benchmark required for modern pharmaceutical applications. Our commitment to technical excellence ensures that our clients receive materials that are not only chemically pure but also consistent in crystal form and bioavailability.
We invite global partners to collaborate with us to leverage this advanced purification technology for their berberine supply needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific rigor and commercial reliability.
