Advanced Six-Step Synthesis of Berberine Hydrochloride for Scalable Pharmaceutical Manufacturing
Advanced Six-Step Synthesis of Berberine Hydrochloride for Scalable Pharmaceutical Manufacturing
The global demand for broad-spectrum antibiotics continues to drive innovation in the synthesis of critical active pharmaceutical ingredients (APIs) and their intermediates. A pivotal development in this sector is detailed in patent CN101245064A, which outlines a robust and economically viable preparation method for berberine hydrochloride. This specific chemical entity is renowned for its potent inhibitory effects against a wide array of Gram-positive and Gram-negative bacteria, including Shigella, Staphylococcus aureus, and Vibrio cholerae. While traditional extraction from plant sources like Coptis chinensis is limited by agricultural constraints, chemical synthesis offers a scalable alternative. However, historical synthetic routes have often been plagued by excessive step counts and prohibitive costs. The methodology presented in this patent addresses these critical bottlenecks by introducing a streamlined six-step sequence that prioritizes high yield, operational simplicity, and the use of readily available starting materials, positioning it as a superior choice for reliable pharmaceutical intermediate supplier networks seeking efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the total synthesis of berberine has been challenged by inefficient reaction pathways that hinder commercial viability. As early as 1912, Pictet and Gams reported a foundational route utilizing piperonyl ethylamine and 3,4-dimethoxyphenylacetic acid. While scientifically significant, this classical approach involves a cumbersome series of transformations including acid chloride preparation, condensation, dehydration ring closure, reduction, and final oxidation. 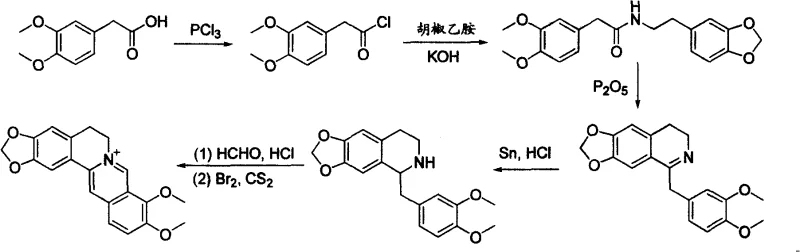 The inherent flaw in this legacy technology lies in the cyclization process, which requires extended reaction sequences and suffers from poor selectivity during the second ring-closing event. Consequently, the cumulative yield across these numerous steps is significantly diminished, driving up the cost of goods sold (COGS) and generating substantial chemical waste, which is untenable for modern green chemistry standards in high-purity API intermediate manufacturing.
The inherent flaw in this legacy technology lies in the cyclization process, which requires extended reaction sequences and suffers from poor selectivity during the second ring-closing event. Consequently, the cumulative yield across these numerous steps is significantly diminished, driving up the cost of goods sold (COGS) and generating substantial chemical waste, which is untenable for modern green chemistry standards in high-purity API intermediate manufacturing.
Furthermore, subsequent improvements reported in the 1980s, such as those found in the National Compendium of Raw Materials Processes, introduced new complications despite attempting to streamline the workflow. 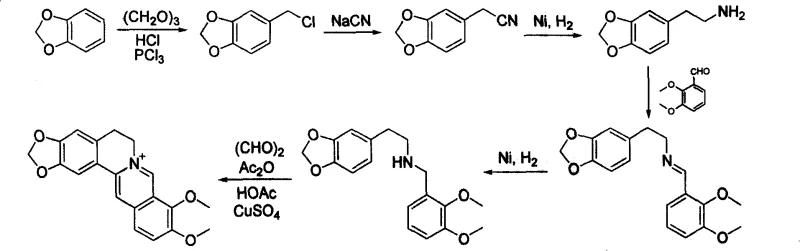 A critical drawback of these intermediate-generation processes was the reliance on copper catalysts during the crucial ring-formation stage. The presence of transition metals necessitates rigorous post-reaction purification protocols to meet stringent pharmacopeial limits for heavy metals. This de-metalation step adds significant operational burden, increases solvent consumption, and introduces potential points of failure in the supply chain. For procurement managers, the requirement for specialized scavenging resins or complex extraction procedures translates directly into higher processing costs and longer lead times, creating a barrier to efficient cost reduction in pharmaceutical intermediate manufacturing.
A critical drawback of these intermediate-generation processes was the reliance on copper catalysts during the crucial ring-formation stage. The presence of transition metals necessitates rigorous post-reaction purification protocols to meet stringent pharmacopeial limits for heavy metals. This de-metalation step adds significant operational burden, increases solvent consumption, and introduces potential points of failure in the supply chain. For procurement managers, the requirement for specialized scavenging resins or complex extraction procedures translates directly into higher processing costs and longer lead times, creating a barrier to efficient cost reduction in pharmaceutical intermediate manufacturing.
The Novel Approach
In stark contrast to these legacy methodologies, the invention disclosed in CN101245064A presents a paradigm shift by utilizing 2,3-dimethoxybenzyl alcohol and piperonyl ethylamine as the foundational building blocks. This strategic selection of starting materials allows for a highly convergent six-step synthesis comprising alkylation, chloromethylation, cyanation, alcoholysis, condensation, and cyclization. 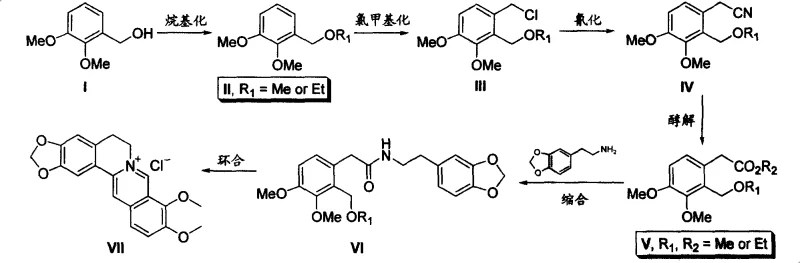 The novelty of this approach lies not just in the sequence, but in the specific optimization of each transformation to maximize throughput. For instance, the initial alkylation protects the hydroxyl group efficiently, while the subsequent chloromethylation and cyanation steps utilize phase transfer catalysis to enhance reaction kinetics without requiring extreme conditions. By avoiding the use of copper catalysts entirely and replacing them with phosphorus pentachloride for the final cyclization, the process eliminates the need for heavy metal removal. This results in a cleaner crude product profile, significantly simplifying downstream purification and enabling a more direct path to high-purity pharmaceutical intermediates suitable for immediate formulation or further processing.
The novelty of this approach lies not just in the sequence, but in the specific optimization of each transformation to maximize throughput. For instance, the initial alkylation protects the hydroxyl group efficiently, while the subsequent chloromethylation and cyanation steps utilize phase transfer catalysis to enhance reaction kinetics without requiring extreme conditions. By avoiding the use of copper catalysts entirely and replacing them with phosphorus pentachloride for the final cyclization, the process eliminates the need for heavy metal removal. This results in a cleaner crude product profile, significantly simplifying downstream purification and enabling a more direct path to high-purity pharmaceutical intermediates suitable for immediate formulation or further processing.
Mechanistic Insights into Phosphorus-Mediated Cyclization and Condensation
The core of this synthetic innovation rests on the precise control of the amide condensation and the subsequent Bischler-Napieralski-type cyclization. In the fifth step, the reaction between the ester intermediate and piperonyl ethylamine is conducted under thermal conditions ranging from 100°C to 200°C. This direct aminolysis avoids the formation of unstable acid chloride intermediates that often degrade or polymerize, thereby preserving the integrity of the sensitive methylenedioxy and methoxy functional groups. The mechanistic pathway ensures that the amide bond forms with high fidelity, setting the stage for the final ring closure. The use of a slight molar excess of the amine (1.0:1.0-1.2 ratio) drives the equilibrium towards product formation without generating excessive unreacted amine that would complicate purification, demonstrating a sophisticated understanding of reaction thermodynamics essential for commercial scale-up of complex pharmaceutical intermediates.
The final cyclization step employs phosphorus pentachloride (PCl5) as the dehydrating agent, a critical deviation from oxidative or metal-catalyzed methods. The mechanism involves the activation of the amide carbonyl by PCl5 to form an imidoyl chloride intermediate, which then undergoes intramolecular electrophilic aromatic substitution to close the isoquinoline ring. This reaction is typically conducted at moderate temperatures (30-80°C) over a period of several days to ensure complete conversion. The choice of PCl5 is strategically advantageous because the byproducts are phosphorus oxychloride and HCl, which are easily removed during the aqueous workup and neutralization phases. This contrasts sharply with metal-catalyzed routes where metal residues can coordinate with the product, leading to persistent impurities. The result is a robust process capable of delivering berberine hydrochloride with exceptional purity, meeting the rigorous quality control standards required by regulatory bodies for antibiotic substances.
How to Synthesize Berberine Hydrochloride Efficiently
The synthesis of berberine hydrochloride via this patented route offers a clear roadmap for laboratory and pilot-scale production, emphasizing safety and yield optimization at every stage. The process begins with the protection of 2,3-dimethoxybenzyl alcohol, followed by functionalization to introduce the nitrile group, which serves as the precursor for the ester linkage. Detailed procedural parameters, including specific solvent choices like dichloromethane and toluene, and precise temperature controls for exothermic steps such as chloromethylation, are critical for success. The following guide summarizes the standardized operational protocol derived from the patent examples, ensuring reproducibility and safety for technical teams aiming to implement this technology.
- Perform alkylation of 2,3-dimethoxybenzyl alcohol with dimethyl sulfate and base at 80°C to form the protected ether intermediate.
- Execute chloromethylation using paraformaldehyde and HCl, followed by cyanation with sodium cyanide under phase transfer conditions.
- Complete the sequence via alcoholysis to ester, thermal condensation with piperonyl ethylamine, and final ring closure using phosphorus pentachloride.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders responsible for sourcing and logistics, the transition to this novel synthesis route offers profound strategic benefits that extend beyond simple chemical yield. The elimination of transition metal catalysts fundamentally alters the cost structure of production by removing the need for expensive metal scavengers and the associated validation testing for heavy metal residuals. This simplification of the purification train reduces solvent usage and waste disposal costs, contributing to substantial cost savings in the overall manufacturing budget. Furthermore, the reliance on commodity chemicals like 2,3-dimethoxybenzyl alcohol and piperonyl ethylamine ensures that the supply chain is not vulnerable to the volatility of niche reagent markets. This stability is crucial for maintaining continuous production schedules and meeting the Just-In-Time delivery expectations of downstream pharmaceutical manufacturers who rely on consistent availability of high-purity API intermediates.
- Cost Reduction in Manufacturing: The economic advantage of this process is primarily driven by the drastic simplification of the downstream processing workflow. By circumventing the copper-catalyzed cyclization step found in older methods, manufacturers avoid the capital expenditure and operational expense associated with heavy metal removal technologies. The high yields reported in the patent examples, such as 98% for the alkylation step and 95% for the cyanation, indicate a highly efficient atom economy that minimizes raw material waste. Additionally, the use of standard solvents like toluene and dichloromethane, which are easily recovered and recycled in industrial settings, further lowers the variable costs per kilogram of finished product, making this a highly competitive option for cost reduction in pharmaceutical intermediate manufacturing.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of widely available starting materials that are produced by multiple global suppliers. Unlike routes dependent on specialized or proprietary reagents, the precursors for this synthesis are bulk commodities, reducing the risk of supply disruptions due to single-source dependency. The robustness of the reaction conditions, which tolerate moderate temperature ranges and do not require cryogenic cooling or ultra-high pressure, also means that the process can be executed in a wider variety of manufacturing facilities. This flexibility allows for diversified production strategies, ensuring that lead times for high-purity pharmaceutical intermediates remain stable even during periods of global logistical stress or regional raw material shortages.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this synthesis aligns well with modern green chemistry principles by reducing the generation of hazardous heavy metal waste. The absence of copper simplifies the effluent treatment process, lowering the burden on wastewater treatment facilities and ensuring easier compliance with increasingly strict environmental regulations. The process is inherently scalable, as demonstrated by the straightforward workup procedures involving simple phase separations and crystallizations. This scalability ensures that the transition from pilot batches to multi-ton commercial production can be achieved with minimal re-engineering, providing a clear path for rapid capacity expansion to meet growing market demand for broad-spectrum antibiotics without compromising on environmental stewardship.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical implications of adopting this technology for large-scale production.
Q: What are the primary advantages of the new berberine synthesis route over the Pictet-Gams method?
A: The new route described in CN101245064A significantly shortens the synthetic pathway compared to the traditional Pictet-Gams method. It avoids the lengthy dehydration and oxidation steps, resulting in higher overall yields and reduced operational complexity, making it more suitable for industrial scale-up.
Q: How does this process address heavy metal contamination concerns found in older methods?
A: Unlike previous industrial methods that utilized copper catalysts requiring complex post-reaction removal steps, this novel synthesis relies on phosphorus pentachloride for cyclization. This eliminates the need for expensive and operationally difficult heavy metal scavenging processes, ensuring a cleaner final product profile.
Q: Are the raw materials for this synthesis readily available for commercial production?
A: Yes, the process utilizes 2,3-dimethoxybenzyl alcohol and piperonyl ethylamine as starting materials. These are commercially accessible bulk chemicals, which enhances supply chain reliability and reduces the risk of raw material bottlenecks compared to routes requiring specialized or rare precursors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Berberine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex antibiotics like berberine hydrochloride requires more than just a patent; it demands deep process engineering expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this six-step synthesis are fully realized in a GMP-compliant environment. We leverage our rigorous QC labs and stringent purity specifications to guarantee that every batch meets the exacting standards required for pharmaceutical applications, providing our partners with a secure and reliable source of critical intermediates.
We invite procurement leaders and R&D directors to collaborate with us to evaluate the feasibility of integrating this optimized route into your supply chain. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us directly to request specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and profitability in your antibiotic manufacturing operations.
