Advanced Visible Light Catalysis for Scalable Gamma-Hydroxy Ketone Production
The pharmaceutical and fine chemical industries are constantly seeking greener, more efficient pathways to construct complex molecular scaffolds, particularly those containing oxygenated functionalities essential for drug discovery. Patent CN109867593B introduces a groundbreaking methodology for the synthesis of gamma-hydroxy ketone derivatives, a versatile class of intermediates widely used in the construction of bioactive molecules. This innovation leverages the power of visible light photocatalysis, utilizing a ruthenium complex to mediate the cross-coupling of readily available styrenes with alpha-bromoacetophenones. Unlike traditional thermal methods that often demand harsh conditions and stoichiometric oxidants, this approach operates under mild, ambient conditions, marking a significant leap forward in sustainable chemical manufacturing. By harnessing light energy to generate radical species, the process achieves high atom economy and eliminates the need for hazardous redox additives, positioning it as an ideal candidate for modern, eco-conscious production lines.
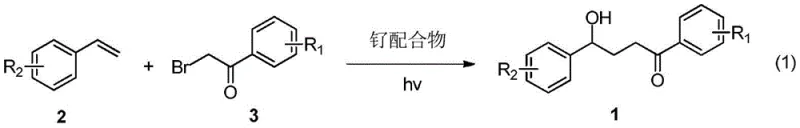
For R&D directors evaluating new synthetic routes, the distinction between conventional methodologies and this novel photocatalytic approach is stark and commercially significant. Traditional syntheses of gamma-hydroxy ketones often rely on multi-step sequences involving stoichiometric organometallic reagents or harsh acidic/basic conditions that can degrade sensitive functional groups. These legacy methods frequently suffer from poor atom economy, generating substantial amounts of salt waste and requiring rigorous purification protocols to remove metal residues. Furthermore, the necessity for strong oxidants or reductants in classical pathways introduces safety hazards and complicates the regulatory approval process for pharmaceutical intermediates due to impurity profiles. In contrast, the novel approach detailed in the patent utilizes a catalytic amount of a ruthenium polypyridyl complex, such as Ru(bpy)3Cl2, activated by simple household-grade CFL lighting. This shift not only simplifies the operational setup but also drastically reduces the environmental footprint by avoiding toxic reagents, thereby streamlining the path from bench-scale discovery to commercial production.
The mechanistic elegance of this transformation lies in the photoredox cycle initiated by the ruthenium catalyst under visible light irradiation. Upon absorption of photons, the ruthenium complex enters an excited state capable of single-electron transfer (SET), facilitating the homolytic cleavage of the carbon-bromine bond in the alpha-bromoacetophenone precursor. This generates a nucleophilic carbon-centered radical which subsequently adds across the double bond of the styrene substrate with high regioselectivity. The resulting benzylic radical is then trapped, likely through interaction with water or hydroxide sources present in the reaction medium, to form the final gamma-hydroxy ketone product while regenerating the ground-state catalyst. This radical pathway is remarkably tolerant of various electronic environments, allowing for the successful coupling of styrenes bearing electron-donating groups like methyl and methoxy, as well as electron-withdrawing substituents such as halogens and trifluoromethyl groups. The ability to control impurity formation through mild radical trapping mechanisms ensures a cleaner crude reaction profile, reducing the burden on downstream purification processes.
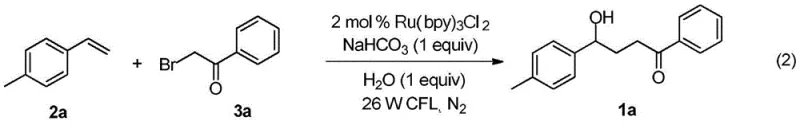
To facilitate the practical adoption of this technology, the patent provides a robust and reproducible protocol for synthesizing these valuable intermediates. The procedure involves combining the styrene derivative and the alpha-bromoacetophenone in a polar aprotic solvent like acetonitrile, with a mild inorganic base such as sodium bicarbonate to neutralize acid byproducts. The reaction is conducted under an inert nitrogen atmosphere to prevent unwanted side reactions with oxygen, although the mild nature of the radicals suggests potential robustness. Irradiation with a standard 26W white CFL lamp at room temperature drives the reaction to completion typically within 24 to 48 hours. Following the reaction, standard workup procedures involving solvent removal and silica gel chromatography yield the pure gamma-hydroxy ketone derivatives in good yields, as exemplified by the 70% isolated yield reported for the para-methyl substituted product.
For procurement managers and supply chain heads, the commercial implications of adopting this visible light catalyzed route are profound, offering tangible benefits in cost structure and operational reliability. The elimination of expensive and hazardous stoichiometric oxidants or reductants directly translates to a reduction in raw material costs and waste disposal fees, which are often hidden but significant expenses in fine chemical manufacturing. Moreover, the use of commodity chemicals like styrene and alpha-bromoacetophenone, which are produced on a massive global scale, ensures a stable and continuous supply chain, mitigating the risks associated with sourcing exotic or proprietary reagents. The mild reaction conditions, specifically the ability to run the process at ambient temperatures (20-40°C) without high-pressure equipment, lower the capital expenditure required for reactor infrastructure and reduce energy consumption for heating or cooling, further enhancing the overall cost-efficiency of the manufacturing process.
- Cost Reduction in Manufacturing: The economic advantage of this process is driven primarily by the substitution of costly reagents with abundant light energy and the use of low-loading catalyst systems. By operating with only 2 mol% of the ruthenium catalyst and avoiding the purchase of specialized oxidizing agents, the direct material costs are significantly minimized. Additionally, the simplified workup procedure, which avoids complex quenching steps required for reactive metals, reduces labor hours and solvent usage, contributing to a leaner and more profitable production model for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Reliability in the supply of critical intermediates is paramount for maintaining uninterrupted drug production schedules. Since the key starting materials, styrenes and alpha-bromoacetophenones, are bulk chemicals with established global supply networks, the risk of supply disruption is inherently low. The robustness of the reaction conditions also means that production is less susceptible to fluctuations in utility availability, such as steam for heating, as the process relies on electrical lighting which is easily backed up, ensuring consistent output and dependable lead times for downstream customers.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been a challenge, but the use of simple CFL lamps and the absence of exothermic hazards associated with strong oxidants make this process highly scalable. The environmental compliance aspect is particularly strong, as the process generates minimal hazardous waste and avoids the release of volatile organic compounds associated with aggressive solvents or reagents. This alignment with green chemistry principles facilitates easier regulatory approval and reduces the environmental taxes and penalties often levied on traditional chemical manufacturing facilities.
Understanding the technical nuances of this synthesis is critical for partners looking to integrate this technology into their existing portfolios. The following questions address common inquiries regarding the operational parameters and scope of this photocatalytic method, providing clarity on its applicability to diverse chemical challenges. These insights are derived directly from the experimental data and optimization studies presented in the patent documentation, ensuring that the information provided is accurate and actionable for technical decision-makers evaluating this route for potential licensing or contract manufacturing opportunities.
- Weigh styrene derivative (0.3 mmol), Ru(bpy)3Cl2·6H2O catalyst (0.006 mmol), alpha-bromoacetophenone (0.6 mmol), and sodium bicarbonate (0.3 mmol) into a reaction vessel under inert atmosphere.
- Add acetonitrile (3 mL) and water (0.3 mmol) to the mixture and stir under nitrogen protection.
- Irradiate the reaction mixture with a 26W white CFL lamp at room temperature for 24 hours, then purify via silica gel column chromatography.
Frequently Asked Questions (FAQ)
Q: What are the optimal reaction conditions for this photocatalytic coupling?
A: The patent specifies that the optimal conditions involve using Ru(bpy)3Cl2·6H2O as the catalyst at a 2 mol% loading, with sodium bicarbonate as the base in acetonitrile solvent. The reaction proceeds best at room temperature (20-40°C) under 26W white CFL illumination for 24 to 48 hours.
Q: Can this method tolerate diverse functional groups on the styrene substrate?
A: Yes, the methodology demonstrates excellent substrate tolerance. The patent highlights that substituents such as methyl, methoxy, halogens (fluorine, chlorine, bromo), trifluoromethyl, cyano, and ester groups on the styrene ring are well-tolerated, allowing for the synthesis of a wide variety of functionalized gamma-hydroxy ketone derivatives.
Q: Is this process suitable for large-scale manufacturing without hazardous oxidants?
A: Absolutely. A key advantage of this visible light catalyzed process is its high atom economy and safety profile. It does not require the addition of external oxidants or reductants, utilizing light energy instead to drive the radical generation, which significantly simplifies waste treatment and enhances safety for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gamma-Hydroxy Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of visible light catalysis in modernizing the synthesis of complex pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN109867593B can be seamlessly translated into robust industrial processes. Our state-of-the-art facilities are equipped with specialized photochemical reactors and stringent purity specifications, supported by rigorous QC labs that guarantee the highest quality standards for every batch of gamma-hydroxy ketone derivatives we produce. We are committed to delivering not just chemicals, but reliable solutions that enhance your supply chain efficiency.
We invite you to explore how this advanced synthetic route can optimize your production costs and improve your product quality. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise in photocatalytic manufacturing can become a strategic asset for your organization.
