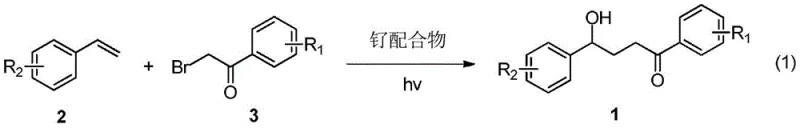
Scalable Visible Light Catalysis for Gamma-Hydroxy Ketone Derivatives Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking more sustainable and efficient pathways to construct complex molecular architectures, particularly for key intermediates used in drug discovery. A significant breakthrough in this domain is detailed in patent CN109867593B, which discloses a novel method for synthesizing gamma-hydroxy ketone derivatives through visible light photocatalysis. This technology represents a paradigm shift from traditional thermal processes, leveraging the energy of visible light to drive the cross-coupling of styrenes and alpha-bromoacetophenones. By utilizing a ruthenium complex catalyst under mild irradiation, this process achieves the simultaneous construction of carbon-carbon and carbon-oxygen bonds in a single operational step. For R&D directors and process chemists, this offers a compelling alternative to multi-step syntheses that often suffer from low overall yields and excessive waste generation. The ability to access these valuable scaffolds under such benign conditions opens new avenues for the rapid assembly of bioactive molecules and functional materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the bifunctionalization of olefins to generate gamma-hydroxy ketones has relied heavily on stoichiometric transition metal reagents or harsh oxidative conditions that pose significant safety and environmental challenges. Traditional protocols often necessitate the use of strong oxidants or reductants, which not only increase the cost of goods but also generate substantial quantities of hazardous waste that require complex disposal procedures. Furthermore, many classical methods exhibit narrow substrate scopes, failing to tolerate sensitive functional groups that are frequently encountered in advanced pharmaceutical intermediates. The requirement for elevated temperatures and high pressures in some thermal catalytic systems further exacerbates energy consumption and limits the feasibility of scaling these reactions in standard glass-lined reactors. Consequently, procurement teams often face supply chain bottlenecks when sourcing precursors that require such specialized and risky manufacturing environments.
The Novel Approach
In stark contrast, the methodology described in patent CN109867593B utilizes a photoredox catalytic cycle driven by visible light, effectively bypassing the need for aggressive chemical oxidants or reductants. This approach employs inexpensive and widely available styrene derivatives as starting materials, reacting them with alpha-bromoacetophenones in the presence of a ruthenium polypyridyl complex such as Ru(bpy)3Cl2. The reaction proceeds smoothly at temperatures ranging from 20°C to 40°C, utilizing common light sources like 26W compact fluorescent lamps (CFL) or LEDs. This mildness ensures that sensitive functional groups remain intact, thereby expanding the chemical space accessible to medicinal chemists. Moreover, the absence of external redox additives significantly improves the atom economy of the transformation, aligning perfectly with modern green chemistry principles.
Mechanistic Insights into Ru(bpy)3Cl2-Catalyzed Hydroxyalkylation
The core of this transformative process lies in the unique ability of the ruthenium photocatalyst to mediate single-electron transfer (SET) events under visible light irradiation. Upon absorption of photons, the Ru(II) complex enters an excited state capable of reducing the carbon-bromine bond of the alpha-bromoacetophenone substrate. This reduction generates a reactive carbon-centered radical species while regenerating the oxidized form of the catalyst, which is subsequently reduced back to its ground state by the sacrificial electron donor or through the catalytic cycle involving the alkene. The generated radical then adds across the double bond of the styrene derivative, forming a new benzylic radical intermediate. This intermediate undergoes further oxidation and nucleophilic attack by water or hydroxide species present in the reaction medium, ultimately yielding the gamma-hydroxy ketone product. This elegant mechanism avoids the high-energy transition states associated with thermal radical initiators, providing superior control over regioselectivity and minimizing polymerization side reactions.
From an impurity control perspective, the mild nature of the photocatalytic conditions plays a crucial role in ensuring high product purity. Unlike thermal radical reactions that can lead to uncontrolled chain propagation and oligomerization, the photon-driven process allows for precise temporal control over radical generation simply by switching the light source on or off. The use of sodium bicarbonate as a mild base further buffers the reaction environment, preventing acid-catalyzed dehydration of the alcohol product which is a common degradation pathway in acidic media. Additionally, the compatibility of the system with aqueous conditions facilitates the solubility of inorganic salts and simplifies the workup procedure. These factors collectively contribute to a cleaner crude reaction profile, reducing the burden on downstream purification units and enhancing the overall yield of the isolated target compound.
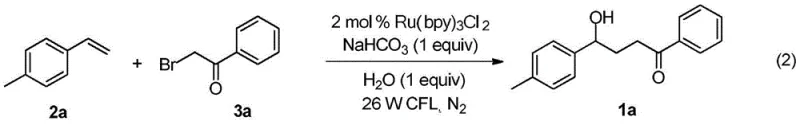
How to Synthesize Gamma-Hydroxy Ketone Derivatives Efficiently
To implement this cutting-edge synthesis in a laboratory or pilot plant setting, operators must adhere to specific parameters regarding catalyst loading, solvent composition, and irradiation intensity to maximize efficiency. The patent data indicates that a molar ratio of styrene to catalyst of 1:0.02 is optimal, ensuring sufficient turnover without excessive metal contamination. The reaction is typically conducted in acetonitrile with a small amount of water, which serves as both a co-solvent and the oxygen source for the hydroxyl group. Maintaining an inert atmosphere, such as nitrogen, is critical during the setup to prevent quenching of the excited catalyst by atmospheric oxygen, although the reaction itself does not require an external oxidant. The following guide outlines the standardized protocol derived from the exemplary embodiments to ensure reproducible results.
- Weigh styrene derivative (0.3 mmol), Ru(bpy)3Cl2·6H2O catalyst (0.006 mmol), alpha-bromoacetophenone (0.6 mmol), and sodium bicarbonate (0.3 mmol) into a reaction vessel under inert atmosphere.
- Add acetonitrile (3 mL) and water (0.3 mmol) to the mixture and stir under nitrogen protection.
- Irradiate the reaction mixture with a 26W white CFL lamp at room temperature for 24 hours, then purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this visible light catalytic technology offers profound strategic benefits that extend beyond mere technical novelty. The reliance on commodity chemicals like styrene and alpha-bromoacetophenone as feedstocks ensures a stable and cost-effective supply chain, mitigating the risks associated with sourcing exotic or highly regulated precursors. The elimination of expensive stoichiometric oxidants and the reduction in energy consumption due to ambient temperature operation translate directly into lower manufacturing costs. Furthermore, the simplicity of the equipment requirements—essentially a reaction vessel equipped with standard lighting—means that existing infrastructure can often be retrofitted for this chemistry without massive capital expenditure. This flexibility allows for rapid scale-up and responsive production scheduling to meet fluctuating market demands.
- Cost Reduction in Manufacturing: The economic viability of this process is significantly enhanced by the removal of costly redox reagents and the minimization of waste treatment expenses. By avoiding the use of heavy metal oxidants or strong reducing agents, the downstream purification process is streamlined, reducing the consumption of silica gel and solvents required for chromatography. The high atom economy inherent in this addition reaction means that a larger proportion of the raw material mass is incorporated into the final product, drastically lowering the effective cost per kilogram of the active pharmaceutical ingredient intermediate. Additionally, the use of earth-abundant light sources instead of high-energy heating mantles or cryogenic cooling systems contributes to substantial operational expenditure savings over the lifecycle of the product.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent batch-to-batch quality, which is paramount for maintaining uninterrupted supply to downstream customers. Since the raw materials are bulk commodities with multiple global suppliers, the risk of supply disruption due to vendor-specific issues is minimized. The mild reaction environment also reduces the wear and tear on reactor vessels and associated piping, leading to lower maintenance downtime and higher overall equipment effectiveness. This reliability allows supply chain planners to forecast production timelines with greater accuracy, ensuring that critical intermediates are available exactly when needed for subsequent synthetic steps.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been a challenge, but the use of simple CFL or LED arrays makes this specific protocol highly adaptable to flow chemistry or large batch reactors. The absence of hazardous byproducts and the use of non-toxic solvents like acetonitrile (which is easily recovered) align with increasingly stringent environmental regulations globally. This compliance reduces the regulatory burden on the manufacturing site and minimizes the risk of fines or shutdowns due to environmental violations. The ability to run the reaction at near-ambient pressure further simplifies the engineering controls required for safe operation, facilitating a smoother path from gram-scale optimization to ton-scale commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible light catalyzed synthesis. These answers are derived directly from the experimental data and technical specifications provided in the patent literature to assist decision-makers in evaluating the feasibility of this route for their specific applications. Understanding these nuances is essential for integrating this technology into existing manufacturing portfolios.
Q: What are the advantages of using visible light catalysis over traditional transition metal methods?
A: Visible light catalysis utilizes abundant natural resources and operates under significantly milder conditions compared to traditional methods which often require harsh temperatures or pressures. This approach eliminates the need for external oxidants or reductants, resulting in higher atom economy and a cleaner reaction profile with fewer byproducts.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the process is designed for scalability due to its simple operation and use of readily available raw materials like styrene. The reaction proceeds at ambient temperatures (20-40°C) and uses standard lighting sources, making the transition from laboratory scale to commercial production straightforward and cost-effective.
Q: What is the substrate scope for this gamma-hydroxy ketone synthesis?
A: The method demonstrates excellent functional group tolerance, accommodating various substituents on both the styrene and alpha-bromoacetophenone components. Groups such as methyl, methoxy, halogens (fluoro, chloro, bromo), and trifluoromethyl are well-tolerated, allowing for the synthesis of a diverse library of derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gamma-Hydroxy Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of visible light photocatalysis in modern organic synthesis and have integrated these capabilities into our CDMO service offerings. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of gamma-hydroxy ketone derivatives meets the highest international standards. Our commitment to quality and consistency makes us the preferred partner for multinational corporations seeking reliable sources of complex fine chemical intermediates.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this greener methodology. We encourage potential partners to contact us directly to obtain specific COA data for our catalog compounds and to request comprehensive route feasibility assessments for your proprietary targets. Let us collaborate to drive efficiency and sustainability in your supply chain.
