Advanced Synthesis of Mometasone Furoate: Technical Breakthroughs and Commercial Scalability for Global Supply Chains
Advanced Synthesis of Mometasone Furoate: Technical Breakthroughs and Commercial Scalability for Global Supply Chains
The pharmaceutical industry continuously seeks robust, scalable, and environmentally benign pathways for the production of critical corticosteroids like mometasone furoate. A pivotal advancement in this domain is detailed in patent CN109206468B, which outlines a novel preparation method that fundamentally reimagines the chlorination and functionalization steps of the steroid backbone. Unlike conventional routes that rely on hazardous sulfonyl chlorides and suffer from low yields, this innovative process leverages sulfur dioxide (SO2) in conjunction with organic amines to facilitate a highly efficient chlorination reaction. This technical insight report analyzes the mechanistic superiority of this approach, its implications for impurity control, and the tangible commercial advantages it offers to procurement and supply chain stakeholders seeking a reliable pharmaceutical intermediate supplier.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of mometasone furoate has been plagued by significant chemical and operational challenges that hinder cost-effective manufacturing. Traditional protocols, such as those disclosed in earlier literature, typically employ methylene dichloride as a solvent alongside sulfonyl chlorides like p-toluenesulfonyl chloride (TsCl) or methanesulfonyl chloride (MsCl) for the critical 21-position chlorination. These reagents not only introduce the risk of genotoxic impurities due to the formation of sulfonate esters but also often result in suboptimal reaction yields and difficult purification profiles. Furthermore, the reliance on biological fermentation methods, as seen in some alternative patents, imposes stringent technical requirements and frequently delivers inconsistent yields, creating bottlenecks for consistent commercial supply. The accumulation of side products and the necessity for rigorous purification to meet safety standards significantly inflate production costs and extend lead times.
The Novel Approach
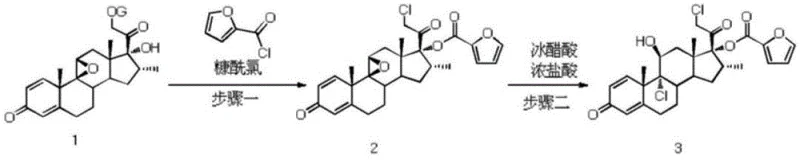
The methodology presented in patent CN109206468B offers a transformative solution by replacing hazardous sulfonylating agents with a sulfur dioxide-mediated chlorination system. This new route initiates with the reaction of the steroid precursor (Compound 1) with a chlorinating agent such as N-chlorosuccinimide (NCS) or acetyl chloride in the presence of SO2 and an organic amine base. This specific combination creates a reactive environment that promotes selective chlorination at the 21-position with exceptional efficiency, achieving molar yields exceeding 96% in optimized examples. Following chlorination, the intermediate undergoes furoylation with furoyl chloride and a subsequent acid-catalyzed ring-opening step to yield the final active pharmaceutical ingredient. This streamlined three-step sequence eliminates the formation of genotoxic sulfonate byproducts, simplifies the workup procedure to basic aqueous quenching and filtration, and operates under mild temperature ranges, thereby enhancing overall process safety and throughput.
Mechanistic Insights into SO2-Catalyzed Chlorination
The core innovation of this synthesis lies in the unique role of sulfur dioxide when complexed with organic amines during the chlorination phase. In the absence of SO2, comparative experiments within the patent data demonstrate that the reaction between the steroid substrate and chlorinating agents like acetyl chloride or NCS proceeds negligibly or not at all. The introduction of SO2, typically adsorbed into an organic amine solvent such as pyridine or lutidine, generates a highly nucleophilic species that activates the 21-hydroxyl group of the steroid for substitution. This activation allows for the rapid displacement of the hydroxyl group by chlorine without the need for forming stable sulfonate intermediates that characterize older, riskier methods. The reaction kinetics are further optimized by maintaining temperatures between -20°C and 40°C, with preferred embodiments operating near 0°C to 10°C to minimize thermal degradation while maximizing conversion rates.
From an impurity control perspective, this mechanism is superior because it bypasses the formation of sulfonate esters, which are known potential genotoxicants. By utilizing reagents like N-chlorosuccinimide (NCS) or dichlorohydantoin in the SO2/amine system, the process ensures that the chlorine atom is introduced directly and cleanly. The patent data highlights that HPLC purity for the chlorinated intermediate (Compound 2) can reach upwards of 98.9%, indicating a remarkably clean reaction profile. This high level of selectivity reduces the burden on downstream purification steps, ensuring that the final mometasone furoate meets stringent pharmacopeial standards with minimal effort. The avoidance of heavy metal catalysts or exotic reagents further simplifies the impurity landscape, making the validation of the drug substance more straightforward for regulatory filings.
How to Synthesize Mometasone Furoate Efficiently
Implementing this synthesis requires precise control over reagent stoichiometry and reaction conditions to replicate the high yields reported in the patent. The process begins with the dissolution of the starting steroid in a suitable solvent like dichloromethane or acetone, followed by cooling and the sequential addition of the organic amine, SO2 source, and chlorinating agent. Detailed standard operating procedures regarding specific molar ratios, addition rates, and quenching protocols are essential for maintaining batch-to-batch consistency. For a comprehensive guide on the exact experimental parameters, including specific solvent volumes and stirring times, please refer to the standardized synthesis steps outlined below.
- Perform chlorination of the steroid precursor (Compound 1) using sulfur dioxide (SO2) and a chlorinating agent like N-chlorosuccinimide (NCS) in the presence of an organic amine.
- Conduct a furoylation reaction on the resulting chlorinated intermediate (Compound 2) using furoyl chloride and an acid-binding agent.
- Execute a ring-opening reaction on the furoylated intermediate (Compound 3) using concentrated hydrochloric acid in acetic acid to yield the final mometasone furoate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this SO2-catalyzed process represents a strategic opportunity to optimize both cost structures and supply reliability. The shift away from expensive and hazardous sulfonyl chlorides to commodity chemicals like sulfur dioxide and NCS drastically reduces raw material costs. Additionally, the simplified workup procedure, which often involves mere dilution with water and filtration rather than complex chromatographic separations, significantly lowers processing time and waste disposal expenses. This efficiency translates directly into a more competitive pricing model for the final API, allowing manufacturers to offer cost reduction in API manufacturing without compromising on quality or compliance.
- Cost Reduction in Manufacturing: The elimination of costly sulfonylating reagents and the reduction in solvent usage due to higher concentration tolerances lead to substantial cost savings. The high yield of the chlorination step, often surpassing 96%, minimizes the loss of valuable steroid starting materials, which are typically the most expensive component of the bill of materials. Furthermore, the avoidance of genotoxic impurities removes the need for specialized, expensive purification technologies or extensive testing regimes, streamlining the overall production budget.
- Enhanced Supply Chain Reliability: The reagents utilized in this process, including sulfur dioxide, organic amines, and furoyl chloride, are widely available commodities with stable global supply chains. This reduces the risk of production stoppages caused by the scarcity of niche reagents. The robustness of the reaction conditions, which tolerate a broad temperature range and do not require cryogenic cooling or high-pressure equipment, ensures that manufacturing can proceed consistently across different facilities and seasons, securing a steady flow of high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, avoiding high-risk reactions such as hydrogenation or the use of pyrophoric reagents. The mild reaction conditions and the use of common solvents facilitate easy transfer from pilot plant to commercial scale. Moreover, the reduction in hazardous waste generation, particularly the absence of sulfonate byproducts, lightens the environmental protection treatment pressure, aligning with increasingly strict global environmental regulations and sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this method compares to existing technologies. Understanding these nuances is critical for technical teams evaluating the feasibility of adopting this process for their own manufacturing lines.
Q: How does the SO2-catalyzed method improve upon traditional chlorination routes?
A: Traditional methods often utilize sulfonyl chlorides like TsCl or MsCl, which can introduce genotoxic impurities and require harsh conditions. The patented SO2-catalyzed approach operates under mild conditions (-20°C to 40°C), eliminates genotoxic risks associated with sulfonate esters, and achieves significantly higher yields (up to 98% in the chlorination step).
Q: What are the key reagents required for this synthesis?
A: The process relies on readily available industrial reagents including sulfur dioxide (SO2), organic amines (such as pyridine or lutidine), N-chlorosuccinimide (NCS) or acetyl chloride for chlorination, furoyl chloride for esterification, and concentrated hydrochloric acid for the final ring opening.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is designed for industrial scalability. It avoids high-risk reactions, utilizes common solvents like dichloromethane or acetone, and features simple workup procedures (e.g., water dilution and filtration), making it highly adaptable for metric-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Mometasone Furoate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to maintain competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the SO2-catalyzed process are fully realized in a GMP-compliant environment. We are committed to delivering mometasone furoate and its intermediates with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify every batch against the highest industry standards.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic impact of switching to this greener, more efficient method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project timelines and volume needs.
