Advanced Synthesis of Mometasone Furoate: A Technical Breakthrough for Commercial Scale-up
Advanced Synthesis of Mometasone Furoate: A Technical Breakthrough for Commercial Scale-up
The pharmaceutical industry constantly seeks robust, scalable, and environmentally sustainable pathways for synthesizing high-value corticosteroids. Patent CN113512085A, published in October 2021, introduces a transformative preparation method for Mometasone Furoate, a potent synthetic glucocorticoid widely used in dermatology for treating conditions such as eczema and psoriasis. This technical disclosure marks a significant departure from traditional methodologies by utilizing a novel starting material, Compound I, derived from 11 alpha-hydroxy-androstane-1, 4-diene-3, 17-dione. The innovation lies not merely in the chemical transformation but in the strategic redesign of the synthetic backbone to enhance yield, purity, and environmental compatibility. For R&D directors and procurement specialists, this patent represents a critical opportunity to optimize supply chains for this high-demand API intermediate.
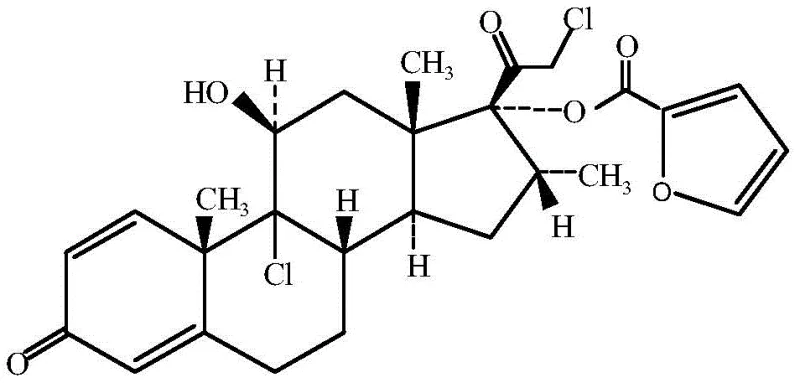
Mometasone Furoate is characterized by its complex steroidal architecture, featuring specific chlorination and hydroxylation patterns that are crucial for its anti-inflammatory potency. The structural integrity of the molecule, particularly the 9, 21-dichloro-11 beta, 17-dihydroxy configuration, demands precise stereochemical control during synthesis. The patent outlines a five-step sequence—cyanohydrin reaction, alkylation, intramolecular substitution, furoylation, and chlorohydroxy reaction—that systematically constructs these features. By shifting the focus to Compound I as the foundational building block, the process circumvents the economic and ecological bottlenecks associated with legacy routes, positioning this method as a superior choice for reliable API intermediate supplier networks aiming for long-term viability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Mometasone Furoate has relied heavily on 9 beta, 11 beta-epoxy-17 alpha, 21-dihydroxy-16 alpha-methyl-1, 4-pregnadiene-3, 20-dione, commonly known as 8DM, as the primary starting material. While chemically feasible, this conventional pathway presents severe drawbacks that impact both operational expenditure and environmental sustainability. The reliance on 8DM drives up raw material costs significantly due to its complex preparation and limited availability. Moreover, the subsequent chemical modifications often necessitate the use of pyridine, a solvent and reagent known for its toxicity and unpleasant odor, which complicates workplace safety and requires extensive ventilation systems.
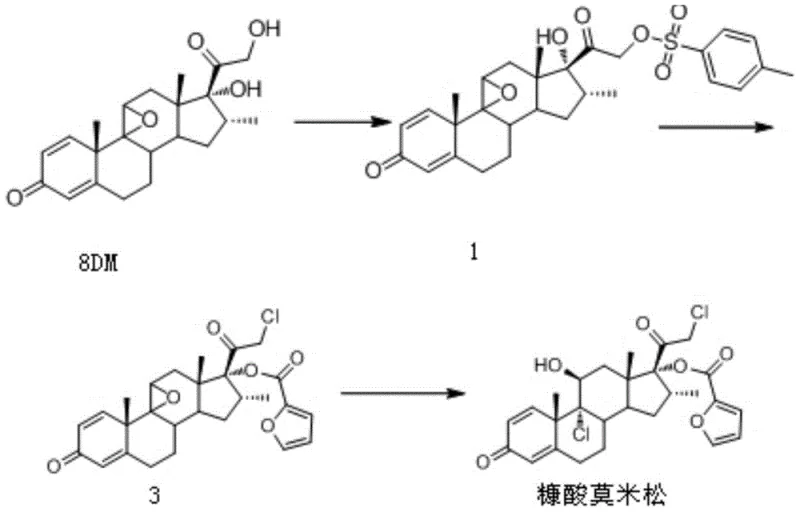
Furthermore, the traditional process generates substantial volumes of high ammonia nitrogen wastewater, posing a formidable challenge for waste treatment facilities and increasing the overall cost of compliance with environmental regulations. The multi-step nature of the old route, involving sulfonylation followed by simultaneous chlorination and esterification, often leads to lower overall yields and a more difficult purification profile. These factors collectively diminish the cost reduction in pharmaceutical manufacturing potential of the legacy method, making it less attractive for modern, green-conscious production facilities that prioritize efficiency and minimal ecological footprint.
The Novel Approach
In stark contrast, the novel approach detailed in CN113512085A leverages Compound I, a readily accessible and cost-effective precursor, to drive the synthesis forward. This strategic shift eliminates the dependency on expensive 8DM and removes the need for pyridine entirely, thereby addressing the core pain points of the conventional method. The new route employs a logical progression of reactions that build molecular complexity with high fidelity. By initiating the sequence with a cyanohydrin reaction, the process efficiently installs the necessary carbon framework for the side chain, setting the stage for subsequent functionalization.
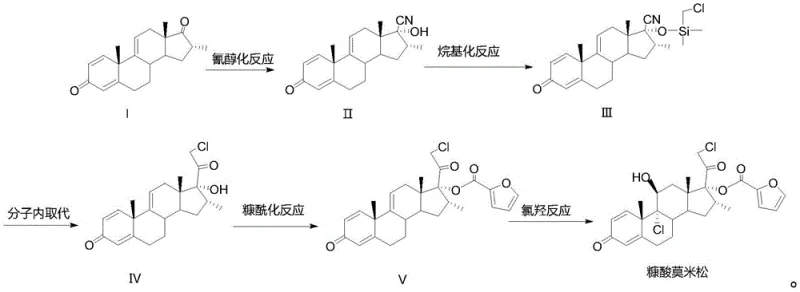
The elegance of this novel approach is further evidenced by its modularity; each step, from alkylation to the final chlorohydroxy reaction, is optimized for high conversion and selectivity. The use of standard industrial solvents such as dichloromethane, tetrahydrofuran, and methanol ensures that the process is easily adaptable to existing manufacturing infrastructure without requiring specialized equipment. This accessibility facilitates the commercial scale-up of complex pharmaceutical intermediates, allowing producers to meet global demand with greater agility. The result is a streamlined workflow that not only reduces the environmental burden but also enhances the economic feasibility of producing high-purity Mometasone Furoate.
Mechanistic Insights into the Five-Step Synthetic Sequence
The core of this patented technology lies in its meticulously designed reaction mechanism, which ensures precise control over stereochemistry and functional group placement. The sequence begins with a cyanohydrin reaction on Compound I, where acetone cyanohydrin acts as the cyanide source in the presence of a mild carbonate catalyst. This step is critical for introducing the two-carbon side chain precursor at the C17 position with high regioselectivity. Following this, an alkylation reaction is performed using chloromethyl dimethylchlorosilane under nitrogen protection. This step serves a dual purpose: it protects the hydroxyl group formed in the previous step and activates the molecule for the subsequent ring-closing event, demonstrating a sophisticated use of protecting group strategy to guide the synthesis.
The third step involves a crucial intramolecular substitution reaction facilitated by a strong base, such as lithium diisopropylamide (LDA), at cryogenic temperatures ranging from -60 to -50 °C. This low-temperature condition is vital for controlling the reactivity of the enolate intermediate, preventing side reactions, and ensuring the formation of the desired cyclic structure in Compound IV. The subsequent furoylation reaction introduces the 2-furoate ester group at the C17 position using furoyl chloride and a tertiary amine catalyst. Finally, the chlorohydroxy reaction installs the critical chlorine atoms at the C9 and C21 positions and the hydroxyl group at C11 using a chlorinating agent like dichlorohydantoin. This final transformation is the key to unlocking the biological activity of the molecule, and the patent specifies conditions that minimize degradation while maximizing yield.
How to Synthesize Mometasone Furoate Efficiently
Implementing this synthesis requires strict adherence to the reaction parameters outlined in the patent to ensure reproducibility and safety. The process demands careful temperature control, particularly during the lithiation and chlorination steps, to maintain product quality. Operators must utilize high-purity solvents and reagents to prevent the introduction of trace metal impurities that could complicate downstream purification. The detailed standardized synthesis steps provided below offer a roadmap for translating this laboratory-scale innovation into a robust manufacturing protocol.
- Perform a cyanohydrin reaction on Compound I using acetone cyanohydrin and a carbonate catalyst to obtain Compound II.
- Conduct an alkylation reaction on Compound II with chloromethyl dimethylchlorosilane under nitrogen protection to yield Compound III.
- Execute an intramolecular substitution reaction on Compound III using a strong base like LDA to form the key intermediate Compound IV.
- Perform a furoylation reaction on Compound IV with furoyl chloride to introduce the furan ester group, obtaining Compound V.
- Complete the synthesis via a chlorohydroxy reaction on Compound V using a chlorinating agent and acid catalyst to finalize Mometasone Furoate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers tangible strategic benefits that extend beyond simple chemical yield. The primary advantage lies in the substantial cost savings driven by the replacement of the expensive 8DM starting material with Compound I. Since Compound I is derived from more abundant and cheaper precursors, the raw material cost baseline is significantly lowered. Additionally, the elimination of pyridine from the process removes the need for costly hazardous waste disposal and specialized handling protocols, further driving down operational expenditures. This aligns perfectly with the goal of cost reduction in pharmaceutical manufacturing, allowing companies to improve their margins without compromising on quality.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the simplification of the supply chain for raw materials. By avoiding the use of high-cost substrates like 8DM and toxic reagents like pyridine, manufacturers can achieve a leaner cost structure. The process utilizes common industrial solvents and reagents that are readily available in the global market, reducing the risk of supply shortages and price volatility. Furthermore, the high purity of the crude product reduces the burden on purification steps, saving time and resources in the final stages of production.
- Enhanced Supply Chain Reliability: Reliability is paramount in the pharmaceutical sector, and this method enhances it by diversifying the source of starting materials. Compound I is easier to synthesize and store compared to the sensitive epoxide structures found in 8DM, leading to better inventory stability. The robustness of the reaction conditions means that production schedules are less likely to be disrupted by minor variations in input quality. This stability ensures a consistent flow of high-purity API intermediates, supporting the continuous operation of downstream formulation lines.
- Scalability and Environmental Compliance: From an environmental perspective, this route is a clear winner, generating significantly less hazardous waste. The absence of high ammonia nitrogen wastewater simplifies effluent treatment, making it easier for facilities to comply with increasingly strict environmental regulations. The process is inherently scalable, as the reaction steps do not require exotic catalysts or extreme pressures that are difficult to manage on a large scale. This scalability ensures that the method can grow with market demand, providing a future-proof solution for commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These answers are derived directly from the technical specifications and beneficial effects described in the patent documentation, providing clarity for stakeholders evaluating this technology for adoption.
Q: What are the primary advantages of the new synthesis route over the conventional 8DM method?
A: The new route utilizes Compound I, which is significantly cheaper and easier to prepare than the traditional 8DM starting material. Furthermore, it eliminates the use of pyridine, thereby reducing toxic wastewater generation and improving environmental compliance.
Q: What purity levels can be achieved with this patented method?
A: The method described in patent CN113512085A achieves a final product purity exceeding 99.20%, with any single impurity controlled to less than 0.10%, meeting stringent pharmaceutical standards.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process employs standard industrial solvents like dichloromethane and tetrahydrofuran and avoids exotic catalysts, making it highly scalable for commercial manufacturing while maintaining safety and cost-efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Mometasone Furoate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthesis route described in CN113512085A and are fully equipped to leverage this technology for our global partners. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our commitment to quality is unwavering, supported by stringent purity specifications and rigorous QC labs that verify every batch against the highest industry standards. We understand that the transition to a new synthetic route requires confidence, and our track record in delivering complex steroid intermediates serves as a testament to our capability.
We invite you to collaborate with us to explore how this advanced manufacturing process can optimize your supply chain and reduce costs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, taking the first step towards a more efficient and sustainable partnership for your Mometasone Furoate needs.
