Advanced Synthesis of Cabozantinib: A Scalable Route for High-Purity Kinase Inhibitor Production
The pharmaceutical landscape for oncology treatments continues to evolve, driven by the demand for multi-target tyrosine kinase inhibitors such as Cabozantinib (XL184). A pivotal advancement in the manufacturing of this critical active pharmaceutical ingredient (API) is detailed in patent CN112979544A, which outlines a superior preparation method for Cabozantinib and its salts. This technical disclosure addresses long-standing challenges in the synthesis of complex kinase inhibitors, specifically focusing on replacing hazardous activation steps with a milder, direct condensation strategy. For R&D directors and supply chain leaders, this patent represents a significant opportunity to optimize production workflows, ensuring that the supply of this life-saving medication remains robust, cost-effective, and compliant with increasingly stringent environmental regulations. The methodology described herein shifts the paradigm from dangerous chlorination processes to a controlled amide coupling, fundamentally altering the risk profile and economic viability of large-scale manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Cabozantinib has relied on pathways that involve the activation of carboxylic acid intermediates using aggressive chlorinating agents. As illustrated in the reaction scheme below, traditional routes often employ oxalyl chloride to convert the cyclopropane carboxylic acid derivative into a highly reactive acid chloride. While chemically effective for bond formation, this approach introduces severe operational hazards, including the handling of corrosive reagents and the generation of toxic gaseous byproducts. Furthermore, the use of such harsh activators often leads to the formation of complex impurity profiles, necessitating rigorous and costly purification steps to meet pharmacopeial standards. The environmental burden associated with the disposal of chlorinated waste streams further complicates the regulatory compliance for manufacturers aiming for sustainable production practices.
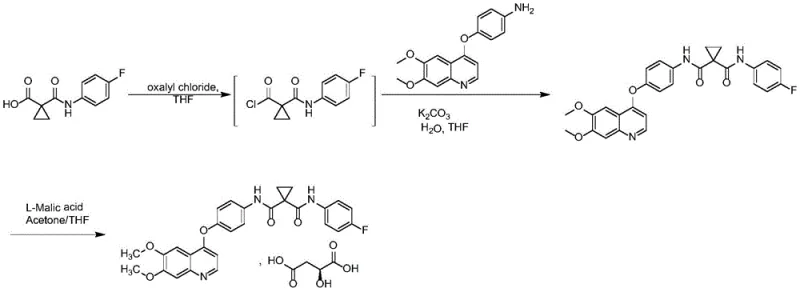
The Novel Approach
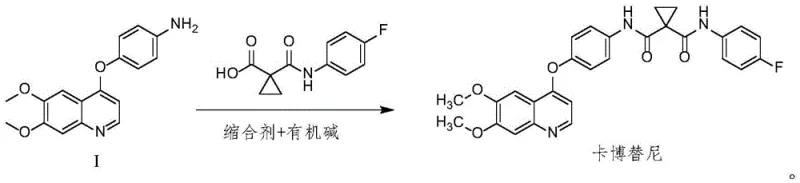
In stark contrast to the legacy methods, the innovative process disclosed in the patent utilizes a direct condensation reaction mediated by modern peptide coupling reagents. This novel approach bypasses the isolation of unstable acid chloride intermediates entirely. Instead, it employs reagents such as O-benzotriazol-N,N,N',N'-tetramethyluronium tetrafluoroborate (TBTU) or hexafluorophosphate (HBTU) in the presence of an organic base. This modification not only mitigates safety risks but also significantly streamlines the reaction workflow. The resulting process operates under mild thermal conditions, typically between 20°C and 40°C, which preserves the integrity of sensitive functional groups and minimizes thermal degradation. By eliminating the need for irritant solvents and dangerous nitration or chlorination steps, this method offers a cleaner, more efficient pathway to the target molecule, aligning perfectly with the goals of green chemistry and process intensification.
Mechanistic Insights into TBTU-Mediated Amide Coupling
The core of this synthetic breakthrough lies in the mechanism of the amide bond formation between the quinoline-aniline derivative (Compound I) and the fluorophenyl-cyclopropane carboxylic acid. In this system, the uronium-based condensing agent activates the carboxylic acid by forming an O-acylisourea intermediate, which is subsequently attacked by the nucleophilic amine group of the quinoline substrate. The presence of an organic base, such as N,N-diisopropylethylamine (DIPEA) or triethylamine, is crucial for scavenging the proton released during the nucleophilic attack, thereby driving the equilibrium towards product formation. This mechanism is highly selective and proceeds with minimal racemization or side reactions, which is critical for maintaining the stereochemical purity required for biological activity. The choice of solvent, preferably dichloromethane or dimethylformamide, ensures optimal solubility of both reactants and the coupling reagent, facilitating a homogeneous reaction environment that maximizes conversion rates.
Furthermore, the patent details a sophisticated workup procedure that is integral to the high purity of the final product. Following the coupling reaction, the mixture is subjected to a sequential washing protocol involving an alkaline aqueous solution followed by water. This step is mechanistically significant as it effectively removes unreacted carboxylic acid, urea byproducts derived from the condensing agent, and residual organic bases. The alkaline wash converts acidic impurities into water-soluble salts, which are easily partitioned into the aqueous phase. Subsequent concentration and recrystallization from solvents like ethanol or ethyl acetate at low temperatures (0-10°C) induce the precipitation of Cabozantinib in a highly crystalline form. This crystallization step is vital for rejecting trace impurities that may have survived the extraction, resulting in a final API with an HPLC purity exceeding 99.9% and single impurities below 0.02%.
How to Synthesize Cabozantinib Efficiently
The implementation of this synthesis route requires precise control over stoichiometry and temperature to ensure reproducibility on a commercial scale. The process begins with the dissolution of the key intermediates in a suitable organic solvent, followed by the controlled addition of the coupling reagent and base. Maintaining the reaction temperature within the specified range is essential to prevent exothermic runaways while ensuring complete conversion. Once the reaction is deemed complete via HPLC monitoring, the workup phase involves careful phase separation and washing to remove soluble impurities. The final isolation of the free base is achieved through crystallization, which can be further processed into the pharmaceutically acceptable L-malate salt. For a detailed, step-by-step guide on the specific quantities, timing, and equipment requirements for this synthesis, please refer to the standardized protocol below.
- React 4-((6,7-dimethoxyquinolin-4-yl)oxy)aniline with 1-(4-fluorophenylcarbamoyl)cyclopropanecarboxylic acid using a condensing agent like TBTU and an organic base in dichloromethane.
- Wash the reaction mixture with alkaline aqueous solution and water, concentrate to dryness, and recrystallize the residue from ethanol or ethyl acetate to obtain high-purity Cabozantinib.
- Dissolve the purified Cabozantinib in a solvent like 2-butanone, add L-malic acid, and cool to precipitate Cabozantinib malate.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthesis route offers profound advantages for procurement managers and supply chain heads tasked with securing reliable sources of high-value oncology intermediates. The elimination of hazardous reagents like oxalyl chloride translates directly into reduced operational costs associated with safety infrastructure, specialized waste treatment, and regulatory compliance. By simplifying the synthetic sequence and improving the overall yield, manufacturers can achieve significant cost reductions in pharmaceutical manufacturing without compromising on quality. The robustness of the crystallization process ensures consistent batch-to-batch quality, reducing the risk of production delays caused by failed purification attempts or off-spec material. This reliability is paramount for maintaining uninterrupted supply chains for critical cancer therapies.
- Cost Reduction in Manufacturing: The transition away from expensive and hazardous chlorinating agents significantly lowers the raw material costs and the overhead associated with handling dangerous chemicals. The simplified workup procedure, which relies on standard aqueous washes rather than complex chromatographic purifications, drastically reduces solvent consumption and processing time. These efficiencies compound to offer a substantially lower cost of goods sold (COGS), making the final API more competitive in the global market while preserving healthy margins for suppliers.
- Enhanced Supply Chain Reliability: The raw materials required for this process, including the quinoline aniline derivative and the cyclopropane acid, are readily available from established chemical suppliers, reducing the risk of raw material shortages. The mild reaction conditions and tolerance to slight variations in parameters make the process highly robust, ensuring high success rates even when scaling up from pilot plants to multi-ton production vessels. This stability allows for more accurate forecasting and shorter lead times for high-purity intermediates, enabling pharmaceutical companies to better manage their inventory levels.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing common organic solvents and standard unit operations like filtration and crystallization that are easily replicated in large-scale reactors. The avoidance of heavy metals and toxic chlorinated byproducts aligns with modern environmental, social, and governance (ESG) goals, facilitating easier permitting and community acceptance for manufacturing sites. This environmental compatibility ensures long-term operational continuity and reduces the liability associated with hazardous waste disposal.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this manufacturing route, we have compiled answers to common questions regarding the process specifics and quality outcomes. These insights are derived directly from the experimental data and comparative examples provided in the patent literature, offering a transparent view of the technology's capabilities. Understanding these nuances is essential for making informed decisions about technology transfer and vendor qualification.
Q: Why is the oxalyl chloride route avoided in modern Cabozantinib synthesis?
A: The traditional route utilizing oxalyl chloride generates hazardous byproducts and requires strict safety controls. The novel condensation method eliminates these risks, offering a safer and more environmentally friendly process suitable for large-scale production.
Q: What represents the key advantage of the new crystallization method?
A: Unlike previous methods that resulted in viscous oils or poor solid states, the new protocol employs specific alkaline washing and recrystallization steps. This ensures the product precipitates as a free-flowing solid with high purity (>99.9%) and excellent filtration properties.
Q: Is this synthesis route suitable for industrial scale-up?
A: Yes, the process utilizes readily available raw materials and mild reaction conditions (20-40°C). The robust workup procedure involving simple washing and crystallization makes it highly adaptable for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cabozantinib Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a dependable partner for the production of complex kinase inhibitors like Cabozantinib. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify every batch against the highest industry standards. Our facility is equipped to handle the specific solvent systems and crystallization requirements outlined in this advanced synthesis route, guaranteeing a seamless transition from development to commercial supply.
We invite you to engage with our technical procurement team to discuss how we can support your specific project requirements. Whether you need a Customized Cost-Saving Analysis for your current supply chain or require specific COA data and route feasibility assessments for Cabozantinib intermediates, our experts are ready to provide the detailed technical support you need. Let us collaborate to secure a stable, high-quality supply of this vital oncology ingredient, driving your drug development programs forward with confidence.
