Revolutionizing Cabozantinib Manufacturing: A Deep Dive into Efficient One-Pot Synthetic Strategies
Revolutionizing Cabozantinib Manufacturing: A Deep Dive into Efficient One-Pot Synthetic Strategies
The pharmaceutical landscape for oncology treatments continues to evolve, driven by the demand for more efficient and sustainable manufacturing processes for critical small molecule inhibitors. Patent CN110117254B introduces a transformative approach to the synthesis of Cabozantinib, a potent multi-target tyrosine kinase inhibitor approved for the treatment of medullary thyroid cancer and advanced renal cell carcinoma. This intellectual property outlines a streamlined three-step protocol that fundamentally reimagines the construction of the quinoline core, moving away from the lengthy and hazardous sequences characteristic of early development routes. By leveraging a strategic one-pot reduction-cyclization methodology followed by a sophisticated copper-catalyzed decarboxylative coupling, this technology offers a compelling value proposition for reliable API intermediate suppliers seeking to optimize their production portfolios. The significance of this patent lies not merely in the chemical novelty but in its direct address of industrial pain points such as waste generation, operational safety, and overall process throughput.
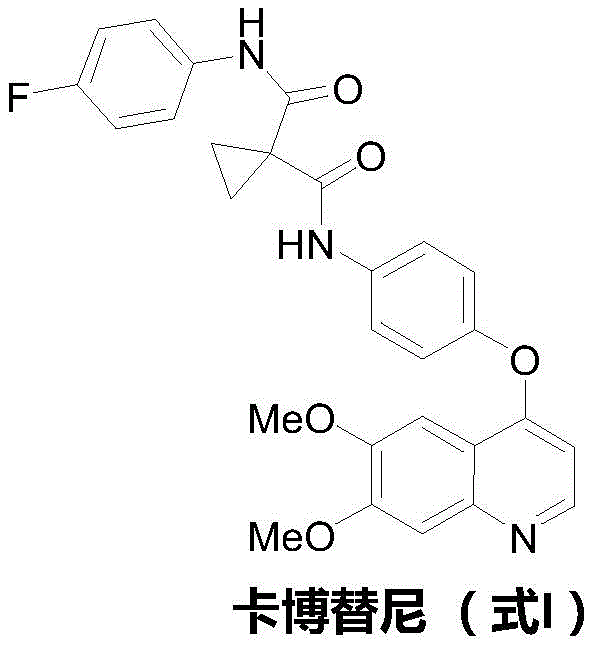
As we analyze the structural complexity of Cabozantinib, depicted in the chemical diagram above, it becomes evident that the molecule requires precise assembly of a substituted quinoline ring and a specific diamide linker. Traditional methods often struggle with the regioselectivity and harsh conditions needed to install the ether linkage at the 4-position of the quinoline. The disclosed invention circumvents these bottlenecks by utilizing 6-nitroveratraldehyde as a ubiquitous starting material. This strategic choice allows for the rapid construction of the heterocyclic scaffold through a cascade of reactions that would typically require isolation and purification of multiple intermediates. For procurement managers evaluating the long-term viability of supply chains, understanding the mechanistic underpinnings of this efficiency is crucial for forecasting cost structures and ensuring continuity of supply for this high-value pharmaceutical intermediate.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Cabozantinib has been dominated by routes that are chemically intensive and operationally cumbersome, often mirroring the initial discovery pathways reported by the originator company. As illustrated in the reaction scheme below from WO2005030140, conventional strategies typically initiate with 3,4-dimethoxyacetophenone, necessitating a discrete nitration step followed by reduction and cyclization to form the 4-hydroxy-quinoline core. This sequence is fraught with challenges, including the handling of strong acids for nitration and the requirement for high-temperature condensation reactions exceeding 165°C to couple the quinoline with the linker moiety. Furthermore, alternative routes disclosed in patents like WO2010083414 rely heavily on phosphorus oxychloride (POCl3) for chlorination, a reagent known for generating significant volumes of acidic wastewater and posing severe corrosion risks to reactor infrastructure. These legacy methods result in extended production timelines, increased solvent consumption, and a complex impurity profile that demands rigorous downstream purification, ultimately inflating the cost of goods sold (COGS) for the final active pharmaceutical ingredient.
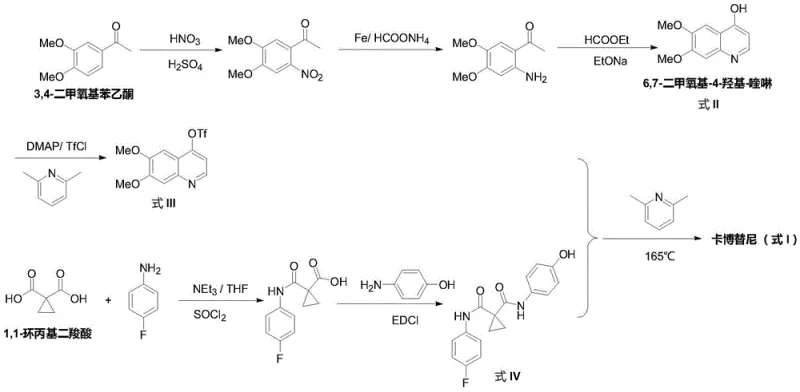
The Novel Approach
In stark contrast to the fragmented nature of prior art, the methodology described in CN110117254B consolidates the synthetic pathway into a highly efficient three-step sequence that maximizes atom economy and minimizes unit operations. The core innovation is the utilization of a one-pot transformation where 6-nitroveratraldehyde undergoes catalytic hydrogenation, cyclization with a pyruvate ester, and subsequent hydrolysis to directly yield the 6,7-dimethoxy-2-carboxyl-quinoline (Formula VII). This telescoped process eliminates the need to isolate unstable amino-intermediates and avoids the hazardous nitration step entirely. The subsequent conversion to the 4-chloro derivative (Formula VIII) using thionyl chloride is straightforward, but the true breakthrough occurs in the final step. By employing a copper catalytic system in a quinoline solvent, the process achieves a simultaneous etherification and decarboxylation reaction. This dual-functionality allows the coupling with the diamine linker (Formula IV) to proceed under significantly milder thermal conditions compared to the 165°C required in older routes, thereby preserving product integrity and reducing energy consumption. This represents a paradigm shift in cost reduction in pharmaceutical manufacturing, offering a cleaner, faster, and safer route to market.
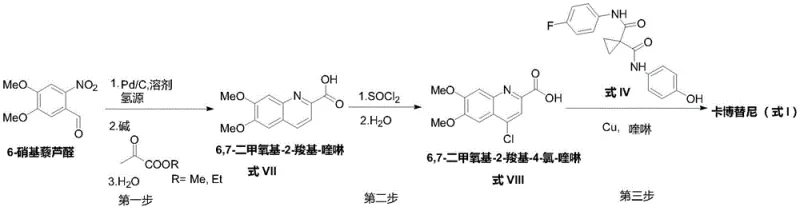
Mechanistic Insights into Copper-Catalyzed Decarboxylative Coupling
The success of the final step in this novel synthesis hinges on the intricate interplay between the electronic properties of the quinoline substrate and the copper catalyst. In traditional nucleophilic aromatic substitution, the displacement of a halide at the 4-position of a quinoline often requires extreme temperatures or strong activating groups. However, in this specific embodiment, the presence of the carboxyl group at the 2-position of the quinoline ring (Formula VIII) serves as a powerful electron-withdrawing group that activates the adjacent 4-chlorine towards nucleophilic attack. When combined with a copper catalyst, likely forming an organocopper intermediate in situ, the reaction pathway shifts towards a decarboxylative cross-coupling mechanism. This mechanism facilitates the formation of the aryl-oxygen bond while concurrently extruding carbon dioxide from the carboxyl group. The result is the direct formation of the target Cabozantinib structure without the need for a separate decarboxylation step, which is often a source of yield loss and impurity formation in other synthetic designs. For R&D directors, this mechanistic elegance translates to a simplified workup procedure and a higher purity profile for the crude product, reducing the burden on analytical quality control laboratories.
Furthermore, the choice of quinoline itself as the reaction solvent in the final step is a masterstroke of process chemistry design. Quinoline acts not only as a high-boiling solvent capable of sustaining the necessary reaction temperature but also potentially functions as a ligand for the copper catalyst, stabilizing the active species and enhancing turnover numbers. The basicity of the solvent may also assist in neutralizing the hydrochloric acid generated during the coupling, driving the equilibrium towards product formation. This self-buffering capability minimizes the need for exogenous bases that could introduce metal contaminants or complicate the salt formation of the final drug substance. By understanding these subtle mechanistic drivers, manufacturers can better optimize reaction parameters such as catalyst loading and temperature ramps to ensure consistent batch-to-batch reproducibility, a critical factor for regulatory compliance in the production of high-purity kinase inhibitors.
How to Synthesize Cabozantinib Efficiently
The implementation of this patented technology requires strict adherence to the specific reaction conditions outlined to maximize yield and minimize side reactions. The process begins with the careful control of hydrogen pressure and temperature during the initial reduction phase to ensure complete conversion of the nitro group without over-reduction of the aldehyde functionality. Following the one-pot cyclization, the isolation of the quinoline carboxylic acid (Formula VII) is achieved through pH adjustment, a technique that leverages the solubility differences of the zwitterionic species to obtain a high-purity solid suitable for the next step. The chlorination step utilizes thionyl chloride, a standard reagent, but requires careful management of off-gases and moisture control to prevent hydrolysis of the acid chloride intermediate. Finally, the copper-catalyzed coupling demands an inert atmosphere and precise stoichiometry to facilitate the decarboxylation event efficiently. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized synthesis guide below.
- Perform a one-pot reduction and cyclization of 6-nitroveratraldehyde using Pd/C and pyruvate ester to form 6,7-dimethoxy-2-carboxyl-quinoline (Formula VII).
- Activate the quinoline intermediate by treating Formula VII with thionyl chloride (SOCl2) to generate the 4-chloro derivative (Formula VIII).
- Execute a copper-catalyzed coupling reaction between Formula VIII and the diamine linker (Formula IV) in quinoline solvent to achieve simultaneous etherification and decarboxylation.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders responsible for the economic and logistical aspects of drug manufacturing, the transition to the synthetic route described in CN110117254B offers substantial strategic benefits that extend beyond simple chemical yield. The consolidation of multiple reaction steps into telescoped one-pot operations drastically reduces the number of processing vessels required, effectively shrinking the physical footprint of the manufacturing campaign. This intensification of the process leads to a significant reduction in solvent usage and energy consumption, as heating and cooling cycles are minimized, and solvent recovery loads are lowered. From a supply chain reliability perspective, the reliance on 6-nitroveratraldehyde, a commodity chemical with a stable global supply, mitigates the risk of bottlenecks associated with custom-synthesized building blocks. This ensures that production schedules can be maintained with greater predictability, safeguarding against delays that could impact downstream formulation and patient access.
- Cost Reduction in Manufacturing: The elimination of hazardous reagents such as phosphorus oxychloride and the avoidance of high-temperature nitration steps translate directly into lower operational expenditures. By removing the need for specialized corrosion-resistant equipment and extensive wastewater treatment facilities required for acidic effluents, the capital investment and ongoing maintenance costs for the production facility are significantly optimized. Additionally, the one-pot nature of the initial steps reduces labor hours and utility costs associated with intermediate isolations, drying, and quality testing, creating a leaner and more cost-effective manufacturing model that enhances overall profit margins for the final API.
- Enhanced Supply Chain Reliability: The robustness of the new synthetic route contributes to a more resilient supply chain by simplifying the logistics of raw material procurement. Since the starting materials are widely available and the process tolerances are improved through the use of catalytic systems rather than stoichiometric hazardous reagents, the risk of batch failures due to raw material variability is diminished. This stability allows for longer production campaigns and larger batch sizes, enabling manufacturers to build strategic inventory buffers more easily. Consequently, partners can rely on a steady flow of commercial scale-up of complex quinolines without the frequent interruptions often seen in more fragile, multi-step synthetic pathways.
- Scalability and Environmental Compliance: As regulatory scrutiny on environmental impact intensifies globally, the greener profile of this synthesis provides a distinct competitive advantage. The reduction in waste generation, particularly the avoidance of phosphate-rich waste streams from POCl3 usage, simplifies the permitting process for manufacturing sites and reduces the liability associated with hazardous waste disposal. The process is inherently scalable, as the exothermic profiles of the one-pot reactions are manageable with standard industrial cooling systems, allowing for a seamless transition from pilot plant to multi-ton commercial production. This alignment with green chemistry principles not only satisfies corporate sustainability goals but also future-proofs the manufacturing asset against tightening environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Cabozantinib synthesis. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the market positioning of the resulting product. The answers below clarify the specific advantages of the copper-catalyzed system and the safety profile of the new route compared to historical methods.
Q: What are the primary safety advantages of the new Cabozantinib synthesis route compared to traditional methods?
A: The novel route eliminates the need for hazardous nitration reactions and phosphorus oxychloride (POCl3) chlorination found in prior art. By utilizing a one-pot hydrogenation/cyclization sequence and thionyl chloride under controlled conditions, the process significantly reduces the generation of toxic waste streams and lowers the risk profile associated with high-temperature exothermic reactions.
Q: How does the copper catalytic system improve the final coupling step?
A: The introduction of a copper catalytic system allows for a decarboxylative coupling mechanism that proceeds under milder conditions than the traditional high-temperature nucleophilic substitution. The electron-withdrawing carboxyl group on the quinoline ring activates the 4-position chlorine, facilitating the reaction with the phenolic linker while simultaneously removing the carboxyl group, thereby streamlining the final purification.
Q: Is the starting material 6-nitroveratraldehyde readily available for large-scale production?
A: Yes, 6-nitroveratraldehyde is a commercially available commodity chemical, which contrasts with some custom-synthesized intermediates required in older pathways. This availability ensures a robust supply chain foundation, reducing lead times and mitigating the risk of raw material shortages for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cabozantinib Supplier
At NINGBO INNO PHARMCHEM, we recognize that the adoption of advanced synthetic methodologies like the one described in CN110117254B is critical for maintaining competitiveness in the global oncology market. Our team of process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical efficiencies of this patent are fully realized in a GMP-compliant environment. We are committed to delivering stringent purity specifications for Cabozantinib and its intermediates, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to handle complex heterocyclic chemistry and catalytic couplings positions us as a strategic partner capable of navigating the intricacies of this optimized route to deliver high-quality material consistently.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis can be integrated into your supply chain. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this greener, more efficient process for your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our technical expertise can drive value and security for your Cabozantinib supply needs.
