Industrial Scale Adenine Production: Overcoming Supply Constraints with Novel Chemical Synthesis
The pharmaceutical industry constantly seeks robust, scalable, and cost-efficient pathways for critical nucleobase intermediates. Patent CN103709164A introduces a transformative chemical synthesis method for Adenine (6-aminopurine), a vital building block for antiviral drugs like Adefovir Dipivoxil and Tenofovir Dipivoxil. Unlike traditional biotechnological approaches, this innovation leverages a purely chemical route starting from malononitrile and thiourea. This shift represents a strategic advancement for supply chain resilience, decoupling production from the volatility of biological fermentation sources. The methodology outlines three distinct schemes, with the primary route demonstrating exceptional operational simplicity by allowing intermediate products to proceed to subsequent steps without rigorous drying or refining. This telescoping capability not only accelerates throughput but also minimizes material loss, positioning this technology as a superior alternative for commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, industrial adenine production has been heavily reliant on biological precursors, creating inherent bottlenecks in the global supply chain. As illustrated in prior art, methods utilizing hypoxanthine require cleavage from inosine, which is exclusively obtained through biological fermentation. Similarly, routes starting from adenosine face identical constraints, tying chemical output to agricultural cycles and fermentation yields. Furthermore, alternative chemical syntheses using diethyl malonate suffer from poor atom economy and excessive reaction steps, involving nitration, chlorination, and multiple ammoniation stages. These legacy processes often necessitate the use of hazardous reagents like phosphorus oxychloride, generating substantial volumes of acidic wastewater containing phosphorus and chlorine. Such environmental liabilities increase disposal costs and complicate regulatory compliance, making these older routes increasingly untenable for modern cost reduction in pharmaceutical intermediates manufacturing.
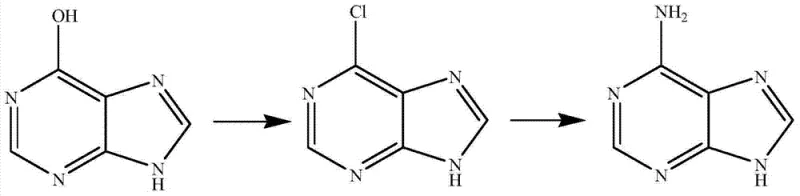
The Novel Approach
The patented methodology fundamentally reengineers the synthetic logic by constructing the pyrimidine ring directly from inexpensive petrochemical feedstocks. By reacting malononitrile with thiourea under sodium alkoxide catalysis, the process generates 4,6-diamino-2-mercaptopyrimidine in a single efficient step. Crucially, this reaction directly installs two amino groups, bypassing the need for subsequent conversion of hydroxyl groups found in diethyl malonate routes. The subsequent transformation involves oxidation, desulfurization, nitrosation, and reduction, culminating in a final cyclization with formamide or orthoformates. This streamlined approach significantly reduces the total number of unit operations. The ability to utilize wet filter cakes directly in downstream reactions eliminates energy-intensive drying phases, thereby enhancing overall process efficiency and establishing a new benchmark for a reliable pharmaceutical intermediate supplier.
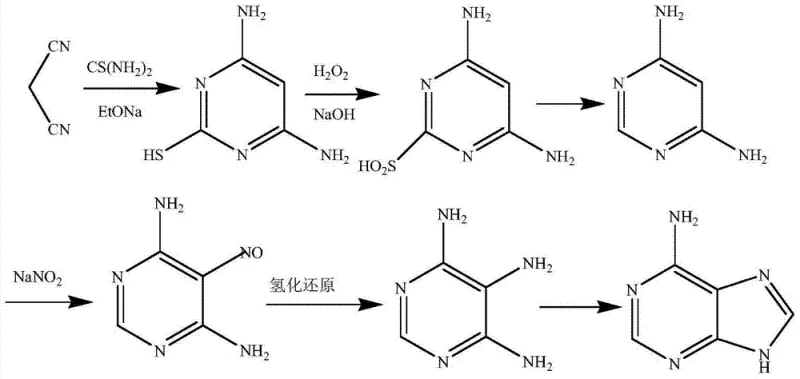
Mechanistic Insights into Malononitrile-Based Cyclization and Desulfurization
The core chemical innovation lies in the initial condensation and the subsequent management of the sulfur moiety. In the first step, the active methylene group of malononitrile attacks the electrophilic carbon of thiourea, facilitated by the strong base sodium ethoxide. This cyclization is highly exothermic and proceeds rapidly at reflux temperatures to form the mercaptopyrimidine core. Unlike routes that generate hydroxyl substituents requiring harsh chlorination agents for activation, this mechanism yields amino groups directly. The sulfur atom, initially introduced as a thiocarbonyl, acts as a leaving group precursor. Through controlled oxidation with hydrogen peroxide in an alkaline medium, the thiol group is converted into a sulfite derivative. This intermediate is unstable in acidic conditions, undergoing hydrolytic desulfurization to yield the 4,6-diaminopyrimidine skeleton. This specific sequence avoids the introduction of halogenated impurities that are notoriously difficult to purge in later stages.
Impurity control is further optimized during the nitrosation and reduction phases. The 4,6-diaminopyrimidine is selectively nitrosated at the 5-position using sodium nitrite under acidic conditions. The resulting nitroso compound is then reduced to the triaminopyrimidine using either sodium dithionite or catalytic hydrogenation. The choice of reducing agent offers flexibility; catalytic hydrogenation using Pd/C or Raney Nickel provides a cleaner profile suitable for high-purity applications. Finally, the triaminopyrimidine undergoes cyclocondensation with a one-carbon donor such as formamide or triethyl orthoformate in the presence of concentrated sulfuric acid. This final ring closure forms the imidazole portion of the purine system. The reaction conditions are tuned to minimize polymerization or degradation, ensuring that the final high-purity pharmaceutical intermediates meet stringent quality standards required for antiviral drug synthesis.
How to Synthesize Adenine Efficiently
Executing this synthesis requires precise control over stoichiometry and temperature, particularly during the oxidation and nitrosation steps which are sensitive to exotherms. The patent details a robust protocol where the initial cyclization is performed in ethanol, followed by a direct slurry transfer to the oxidation vessel. This minimizes exposure to air and reduces handling losses. For the reduction step, while chemical reduction with dithionite is viable, catalytic hydrogenation is preferred for maximizing yield and minimizing inorganic salt waste. The final cyclization demands careful temperature management between 130°C and 170°C to drive the ring closure without charring the organic material. Detailed standardized operating procedures regarding reagent grades, addition rates, and safety protocols are essential for safe technology transfer.
- Cyclize malononitrile and thiourea with sodium ethoxide to form 4,6-diamino-2-mercaptopyrimidine.
- Oxidize the mercapto group using hydrogen peroxide in alkaline solution, followed by acid hydrolysis to remove sulfur.
- Perform nitrosation with sodium nitrite, reduce the nitroso group to an amino group, and finally cyclize with formamide to yield adenine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement strategists and supply chain directors, the transition to this malononitrile-based route offers profound logistical and financial benefits. The primary advantage is the decoupling from biological supply chains. Fermentation-derived starting materials like inosine or adenosine are subject to seasonal variations, contamination risks, and capacity constraints inherent to bioprocessing. By switching to petrochemical-derived malononitrile, manufacturers secure a stable, year-round supply of raw materials that is immune to biological variability. This stability translates directly into reducing lead time for high-purity pharmaceutical intermediates, as sourcing becomes predictable and less prone to sudden market shocks. Furthermore, the simplification of the workflow reduces the working capital tied up in inventory, as the faster cycle times allow for more responsive production scheduling.
- Cost Reduction in Manufacturing: The economic model of this process is driven by the low cost of starting materials and the elimination of intermediate isolation steps. Malononitrile and thiourea are commodity chemicals available at significantly lower price points than fermentation extracts. Moreover, the process design allows for "telescoping," where wet intermediates are used directly in the next reaction. This eliminates the need for expensive drying equipment, reduces energy consumption associated with solvent removal, and minimizes yield losses typically incurred during filtration and drying operations. The avoidance of phosphorus oxychloride also removes the cost burden associated with neutralizing and disposing of hazardous phosphorous waste streams.
- Enhanced Supply Chain Reliability: Reliability is bolstered by the simplicity and robustness of the chemical steps. The reactions operate under relatively mild conditions compared to high-pressure hydrogenolysis or extreme temperature pyrolysis found in other routes. The use of common solvents like ethanol and methanol ensures that solvent recovery and recycling can be easily integrated into existing infrastructure. This operational flexibility means that production can be scaled up or down rapidly in response to market demand without requiring specialized, hard-to-source equipment. Consequently, partners can rely on consistent delivery schedules, mitigating the risk of production stoppages that plague fermentation-dependent supply chains.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is markedly superior. Traditional chlorination methods generate large volumes of acidic wastewater laden with chloride and phosphate salts, requiring extensive treatment facilities. The new method largely circumvents these issues by utilizing oxidative desulfurization and direct amination. The waste profile is simpler, consisting mainly of inorganic salts and aqueous streams that are easier to treat. This reduced environmental footprint facilitates smoother regulatory approvals and lowers the long-term liability associated with waste management. Scalability is further enhanced because the reaction kinetics are well-understood and do not rely on the complex mass transfer limitations often seen in large-scale fermentation tanks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. Understanding these nuances is critical for R&D teams evaluating technology transfer and procurement officers assessing vendor capabilities. The answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: How does the malononitrile route improve supply stability compared to fermentation methods?
A: Unlike traditional methods relying on hypoxanthine or adenosine derived from biological fermentation, this synthetic route uses petrochemical-based malononitrile. This eliminates dependency on agricultural or biological supply chains, ensuring consistent availability and price stability for large-scale pharmaceutical manufacturing.
Q: What are the purity specifications achievable with this synthesis method?
A: The patent data indicates that the final product can achieve a purity of 99.8% (HPLC) after recrystallization. The process avoids complex purification between intermediate steps, yet maintains high quality through controlled reaction conditions and final crystallization.
Q: Does this process generate significant hazardous waste?
A: This method significantly reduces environmental impact compared to routes using phosphorus oxychloride for chlorination. By directly forming amino groups during cyclization and avoiding harsh chlorination steps, the generation of high-concentration acidic wastewater containing phosphorus and chlorine is eliminated.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Adenine Supplier
The technical potential of the malononitrile route for adenine synthesis is immense, offering a clear path to sustainable and economical production. At NINGBO INNO PHARMCHEM, we possess the technical expertise to translate this patent data into commercial reality. As a seasoned CDMO partner, we have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with state-of-the-art reactors capable of handling the specific thermal and pressure requirements of the cyclization and hydrogenation steps described in the patent. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of adenine meets the exacting standards required for antiviral API synthesis, guaranteeing consistency and quality for your downstream processes.
We invite you to collaborate with us to optimize this synthesis for your specific volume requirements. Our team is prepared to provide a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this route for your specific application. We encourage you to contact our technical procurement team to request specific COA data from our pilot runs and to discuss detailed route feasibility assessments. Let us help you secure a competitive advantage through superior supply chain engineering and advanced chemical manufacturing.
