Advanced Catalytic Synthesis of Benzoselenophene Compounds for Commercial Scale Production
Advanced Catalytic Synthesis of Benzoselenophene Compounds for Commercial Scale Production
The pharmaceutical and advanced materials sectors are constantly seeking robust, scalable methodologies for constructing complex heterocyclic scaffolds. Patent CN110483476B introduces a transformative approach to synthesizing benzoselenophene compounds, utilizing a silver oxide-catalyzed cyclization strategy that addresses critical inefficiencies in legacy manufacturing. This technology leverages mild acidic conditions to drive the intramolecular cyclization of 2-ethynyl benzoseleno-ether derivatives, bypassing the need for harsh halogenating agents or prohibitively expensive transition metal catalysts. For R&D directors and procurement specialists, this represents a significant opportunity to streamline supply chains for high-value intermediates used in organic electronics and medicinal chemistry. The process is characterized by its operational simplicity, occurring effectively at temperatures between 10°C and 40°C, which drastically reduces energy consumption and thermal stress on sensitive functional groups. By adopting this methodology, manufacturers can achieve yields exceeding 90% while maintaining a superior impurity profile, essential for downstream applications in conductive polymers and bioactive small molecules.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the benzoselenophene core has relied heavily on electrophilic cyclization strategies that introduce significant environmental and economic burdens. Traditional protocols often employ stoichiometric amounts of molecular halogens, such as iodine or bromine, in chlorinated solvents to effect ring closure. While these methods can achieve respectable conversion rates, they invariably lead to the formation of halogenated byproducts, as illustrated in the generalized structure where X represents I, Br, or Cl.  This halogen incorporation not only limits the structural diversity of the final product but also necessitates rigorous and costly purification steps to remove residual halogens and toxic solvent waste. Furthermore, alternative methods utilizing platinum-based catalysts, while effective, impose a severe cost penalty due to the scarcity and high market price of platinum group metals. These legacy processes often require stringent anhydrous conditions or elevated temperatures, complicating the engineering controls required for safe commercial scale-up and increasing the overall carbon footprint of the manufacturing campaign.
This halogen incorporation not only limits the structural diversity of the final product but also necessitates rigorous and costly purification steps to remove residual halogens and toxic solvent waste. Furthermore, alternative methods utilizing platinum-based catalysts, while effective, impose a severe cost penalty due to the scarcity and high market price of platinum group metals. These legacy processes often require stringent anhydrous conditions or elevated temperatures, complicating the engineering controls required for safe commercial scale-up and increasing the overall carbon footprint of the manufacturing campaign.
The Novel Approach
In stark contrast to these cumbersome legacy routes, the disclosed innovation utilizes silver oxide (Ag2O) as a highly efficient, recyclable catalyst within an acidic solvent system to drive the cyclization forward. This novel pathway enables the direct conversion of 2-ethynyl benzoseleno-ether derivatives into the desired benzoselenophene scaffold without incorporating extraneous halogen atoms into the final structure. 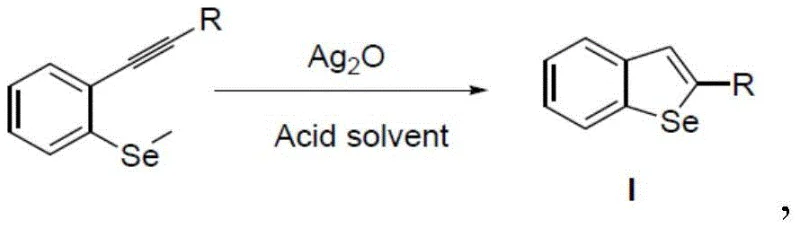 The reaction proceeds smoothly in protic acidic media, such as acetic acid or trifluoroacetic acid, which serves dual roles as both solvent and proton source to facilitate the cyclization mechanism. This approach eliminates the generation of halogenated waste streams, aligning perfectly with green chemistry principles and reducing the burden on wastewater treatment facilities. The operational window is remarkably broad, allowing the reaction to reach completion within approximately 3 hours at near-ambient temperatures, thereby offering a reliable benzoselenophene supplier with a distinct competitive advantage in terms of throughput and sustainability. The simplicity of the workup procedure, involving standard neutralization and extraction, further underscores the practical viability of this method for multi-kilogram production campaigns.
The reaction proceeds smoothly in protic acidic media, such as acetic acid or trifluoroacetic acid, which serves dual roles as both solvent and proton source to facilitate the cyclization mechanism. This approach eliminates the generation of halogenated waste streams, aligning perfectly with green chemistry principles and reducing the burden on wastewater treatment facilities. The operational window is remarkably broad, allowing the reaction to reach completion within approximately 3 hours at near-ambient temperatures, thereby offering a reliable benzoselenophene supplier with a distinct competitive advantage in terms of throughput and sustainability. The simplicity of the workup procedure, involving standard neutralization and extraction, further underscores the practical viability of this method for multi-kilogram production campaigns.
Mechanistic Insights into Ag2O-Catalyzed Cyclization
The efficacy of this synthetic route lies in the unique activation mode provided by the silver oxide catalyst in conjunction with the acidic medium. Mechanistically, the silver cation acts as a soft Lewis acid, coordinating preferentially to the alkyne moiety of the 2-ethynyl benzoseleno-ether substrate. This coordination increases the electrophilicity of the triple bond, rendering it susceptible to nucleophilic attack by the adjacent selenium atom. The acidic solvent environment plays a critical role in protonating the intermediate vinyl-silver species, facilitating the elimination of the silver catalyst and restoring aromaticity to the newly formed selenophene ring. This catalytic cycle is highly turnover-efficient, requiring only 1-10 mol% of silver oxide to drive the reaction to full conversion, with optimal performance observed at loadings between 5-8 mol%. The mildness of this activation strategy ensures that sensitive functional groups on the aromatic rings, such as fluorine or methyl substituents, remain intact, preserving the chemical integrity required for subsequent derivatization in drug discovery or material science applications.
From an impurity control perspective, this mechanism offers a distinct advantage by avoiding radical pathways or harsh oxidative conditions that often lead to polymerization or decomposition of the alkyne starting material. The absence of strong oxidants like molecular iodine prevents the formation of poly-halogenated side products that are notoriously difficult to separate via standard chromatography. Instead, the reaction profile is clean, with the primary impurities being unreacted starting material which can be easily recovered and recycled. The use of acetic acid as a solvent further suppresses side reactions by maintaining a consistent proton activity throughout the reaction vessel, ensuring uniform reaction kinetics even on a large scale. This level of control is paramount for producing high-purity benzoselenophene intermediates that meet the stringent specifications required for electronic grade materials or pharmaceutical active ingredients, where trace metal or halogen contamination can compromise device performance or biological safety.
How to Synthesize 2-Phenylbenzo[b]selenophene Efficiently
To implement this robust synthetic strategy in a laboratory or pilot plant setting, operators should adhere to the optimized parameters established in the patent examples, which demonstrate exceptional reproducibility across various substrates. The process begins with the precise weighing of the 2-ethynyl benzoseleno-ether precursor and the silver oxide catalyst, followed by dissolution in glacial acetic acid under ambient atmosphere. Detailed standardized synthesis steps are provided below to ensure consistent quality and yield. This streamlined protocol minimizes operator exposure to hazardous reagents and reduces the complexity of the equipment setup, making it accessible for facilities without specialized high-pressure or cryogenic capabilities. The resulting crude product typically requires minimal purification, often yielding analytically pure material after a single column chromatography step or recrystallization, thereby maximizing overall process efficiency.
- Combine 2-ethynyl benzoseleno-ether derivatives with silver oxide catalyst (1-10 mol%) in an acidic solvent such as acetic acid.
- Stir the reaction mixture at room temperature (15-30°C) for approximately 1.5 to 3 hours until completion.
- Neutralize with sodium hydroxide, extract with dichloromethane, and purify via column chromatography to obtain high-purity benzoselenophene.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this silver oxide-catalyzed process translates directly into tangible economic and logistical benefits that enhance the resilience of the supply network. By shifting away from precious metal catalysts like platinum and eliminating the need for bulk halogen reagents, the raw material cost structure is significantly optimized, allowing for more competitive pricing models in the global market. The reliance on commodity chemicals such as silver oxide and acetic acid ensures a stable supply chain that is less vulnerable to the geopolitical fluctuations often associated with rare earth or platinum group metals. Furthermore, the simplified waste profile reduces the regulatory burden and disposal costs associated with hazardous halogenated waste, contributing to substantial cost savings in environmental compliance and facility maintenance. This process stability also means fewer batch failures and more predictable delivery schedules, which is critical for maintaining continuous manufacturing operations in downstream industries.
- Cost Reduction in Manufacturing: The substitution of expensive platinum catalysts with economical silver oxide results in a drastic reduction in catalyst expenditure per kilogram of product. Additionally, the elimination of halogenating agents removes the need for specialized corrosion-resistant equipment and extensive scrubbing systems, lowering capital expenditure requirements. The high atom economy of this cyclization reaction ensures that the majority of the starting material mass is incorporated into the final product, minimizing raw material waste and maximizing yield efficiency. These factors combine to create a leaner, more cost-effective manufacturing process that delivers significant value to the bottom line without compromising on product quality or performance specifications.
- Enhanced Supply Chain Reliability: Sourcing silver oxide and acetic acid is far more straightforward and reliable than securing specialized halogenating reagents or platinum complexes, which often have long lead times and limited supplier bases. The robustness of the reaction conditions allows for production in a wider range of manufacturing facilities, diversifying the potential supply base and reducing the risk of single-source dependency. The mild reaction temperatures reduce the risk of thermal runaway incidents, enhancing plant safety and ensuring uninterrupted production schedules. This reliability is crucial for long-term supply agreements, providing partners with the confidence that their demand for high-purity benzoselenophene derivatives will be met consistently and on time.
- Scalability and Environmental Compliance: The one-pot nature of this synthesis, combined with its operation at near-ambient temperatures, makes it inherently scalable from gram to ton quantities without the need for complex process re-engineering. The absence of toxic halogenated byproducts simplifies the effluent treatment process, aligning with increasingly strict global environmental regulations and sustainability goals. The use of acetic acid, a biodegradable and relatively benign solvent, further enhances the green credentials of the process, appealing to environmentally conscious stakeholders. This scalability ensures that the technology can grow with market demand, supporting the commercial scale-up of complex benzoselenophene derivatives for emerging applications in organic electronics and next-generation therapeutics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this patented synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a clear understanding of the process capabilities. Understanding these nuances helps stakeholders make informed decisions about integrating this technology into their existing production portfolios or sourcing strategies for advanced heterocyclic building blocks.
Q: What are the advantages of using Silver Oxide over Platinum catalysts?
A: Silver oxide is significantly more cost-effective than platinum chloride while maintaining high catalytic activity. It eliminates the need for expensive heavy metals, reducing overall production costs and simplifying waste management protocols.
Q: Does this process generate halogenated byproducts?
A: No, unlike conventional electrophilic cyclization methods using iodine or bromine, this Ag2O-catalyzed route produces non-halogenated benzoselenophene derivatives, resulting in a cleaner impurity profile and higher product purity.
Q: What is the typical yield and reaction temperature?
A: The process operates under mild conditions, typically between 15°C and 30°C, achieving yields consistently above 90%. This mild thermal profile enhances safety and energy efficiency during commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Phenylbenzo[b]selenophene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in driving innovation within the pharmaceutical and electronic materials sectors. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. Our expertise in heterocyclic chemistry allows us to adapt and optimize processes like the Ag2O-catalyzed cyclization to meet the specific needs of our clients, guaranteeing a consistent supply of premium-grade benzoselenophene compounds.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your supply chain efficiency and product performance. Let us be your partner in turning complex chemical challenges into commercial successes.
