Advanced Heck Coupling Strategy for Scalable Rosuvastatin Calcium Production
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for high-volume statin drugs, and patent CN101084197B presents a significant advancement in the manufacturing of Rosuvastatin and its pharmaceutically acceptable salts, particularly Rosuvastatin Calcium. This intellectual property details a novel chemical process that diverges from traditional methodologies by employing a palladium-catalyzed Heck reaction to construct the critical carbon-carbon bond between the pyrimidine core and the heptenoic acid side chain. Unlike earlier methods that relied heavily on Wittig olefinations involving phosphine oxides, this approach utilizes a fully substituted, sterically hindered pyrimidine derivative reacting with a vinyl-functionalized side chain precursor. The strategic implementation of this cross-coupling technology addresses long-standing challenges in impurity control and process efficiency, offering a streamlined pathway to the active pharmaceutical ingredient (API). 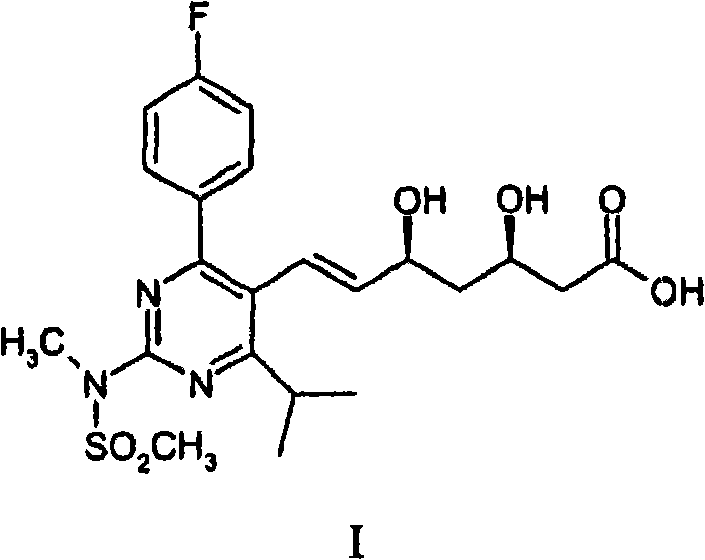
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Rosuvastatin has been dominated by processes described in patents such as WO 00/49014 and WO 04/52867, which typically involve the condensation of phosphoranylidene acetates with pyrimidine aldehydes. These conventional Wittig-type reactions generate stoichiometric amounts of triphenylphosphine oxide as a byproduct, creating significant downstream purification burdens and waste disposal issues for large-scale manufacturing facilities. Furthermore, the handling of sensitive aldehyde intermediates and the requirement for rigorous anhydrous conditions often complicate the operational workflow, leading to potential variability in yield and product quality. The removal of phosphine residues to meet stringent regulatory limits for heavy metals and organic impurities adds additional unit operations, increasing both the production timeline and the overall cost of goods sold for the final API.
The Novel Approach
In contrast, the methodology disclosed in CN101084197B leverages a transition metal-catalyzed cross-coupling strategy that fundamentally alters the disconnection approach for the molecule. By reacting a halogenated or sulfonate-substituted pyrimidine (Formula II) with a vinyl-containing side chain (Formula III), the process eliminates the generation of phosphine oxide waste entirely. This shift allows for the use of more environmentally benign solvent systems, including aqueous mixtures, which simplifies work-up procedures and enhances the safety profile of the manufacturing plant. The ability to tolerate various protecting groups on the side chain during the coupling step provides chemists with greater flexibility in designing the synthetic sequence, ultimately leading to a more robust and adaptable process suitable for commercial scale-up. 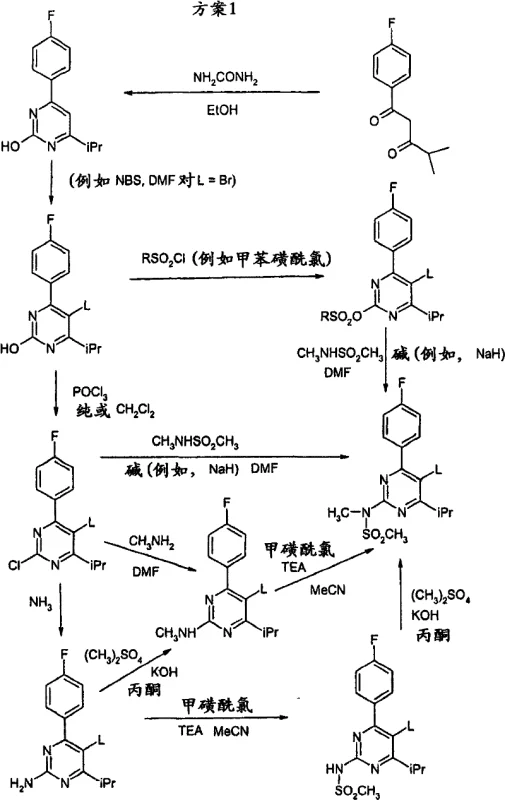
Mechanistic Insights into Palladium-Catalyzed Heck Coupling
The core innovation of this patent lies in the successful application of the Heck reaction to a highly sterically hindered substrate, a feat that was previously considered challenging due to the electronic and steric properties of the pyrimidine ring. The process utilizes a catalytically effective amount of palladium, specifically highlighting complexes with trialkylphosphine ligands such as bis(tri-tert-butylphosphine)palladium(0). These bulky ligands facilitate the oxidative addition of the palladium catalyst into the carbon-halogen bond of the pyrimidine, even in the presence of the adjacent isopropyl group and the 4-fluorophenyl substituent. The reaction proceeds through a standard catalytic cycle involving oxidative addition, migratory insertion of the vinyl alkene, beta-hydride elimination, and catalyst regeneration, but the specific choice of ligand and base is critical to preventing catalyst deactivation and ensuring high turnover numbers.
Furthermore, the patent details the versatility of the side chain precursors, which can exist in various protected forms such as acetonides or silyl ethers to maintain stereochemical integrity during the coupling. The reaction conditions are remarkably mild, typically operating between 30°C and 110°C, with a preference for the 50°C to 60°C range, which minimizes thermal degradation of sensitive functional groups. The use of bases such as N-methyldicyclohexylamine or cesium carbonate, often in conjunction with phase transfer catalysts like tetrabutylammonium salts, ensures efficient neutralization of the acid byproducts generated during the cycle. This mechanistic understanding allows for precise control over the E/Z selectivity of the resulting double bond, ensuring the formation of the pharmacologically active (E)-isomer with high fidelity. 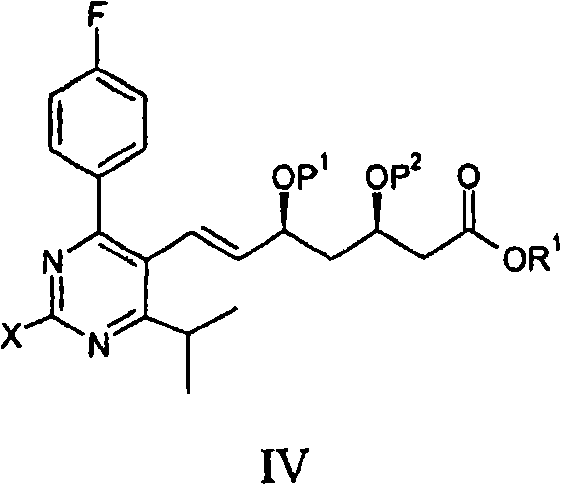
How to Synthesize Rosuvastatin Intermediate Efficiently
The synthesis of the key Rosuvastatin intermediate via this novel Heck coupling route involves a convergent strategy that joins two complex fragments late in the synthetic sequence. The process begins with the preparation of the pyrimidine halide, followed by the coupling with the chiral vinyl side chain under optimized palladium catalysis. Detailed standardized synthetic steps for this specific transformation, including precise reagent ratios, temperature profiles, and work-up protocols, are outlined in the technical guide below to ensure reproducibility and compliance with GMP standards.
- Prepare the pyrimidine core (Formula II) containing a leaving group such as bromine or chlorine at the 5-position and the N-methyl-N-methanesulfonylamino group at the 2-position.
- React the Formula II compound with a vinyl-containing side chain precursor (Formula III) in the presence of a palladium catalyst like bis(tri-tert-butylphosphine)palladium(0) and a base.
- Perform subsequent deprotection steps to remove hydroxyl and carboxyl protecting groups, followed by salt formation to yield Rosuvastatin Calcium.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this Heck coupling technology represents a strategic opportunity to optimize the sourcing of Rosuvastatin intermediates. By eliminating the reliance on phosphine-based reagents, manufacturers can significantly reduce the complexity of their raw material inventory and mitigate the risks associated with the supply volatility of specialized organophosphorus compounds. The simplified purification train resulting from the absence of phosphine oxide byproducts translates directly into higher throughput and reduced solvent consumption, which are key drivers for lowering the overall manufacturing costs without compromising product quality. Additionally, the use of aqueous solvent systems in certain embodiments aligns with modern green chemistry initiatives, potentially reducing waste disposal fees and enhancing the environmental sustainability profile of the supply chain.
- Cost Reduction in Manufacturing: The elimination of stoichiometric phosphine reagents removes a major cost center associated with both reagent purchase and waste treatment. The streamlined purification process reduces the number of chromatographic or crystallization steps required, leading to substantial savings in labor, energy, and solvent usage. Furthermore, the higher atom economy of the coupling reaction compared to Wittig alternatives ensures that a greater proportion of raw materials are incorporated into the final product, maximizing resource efficiency and driving down the cost per kilogram of the active intermediate.
- Enhanced Supply Chain Reliability: The starting materials for this process, such as halogenated pyrimidines and vinyl esters, are derived from commodity chemicals with established global supply networks, reducing the risk of single-source bottlenecks. The robustness of the palladium catalyst system allows for consistent performance across different batches, minimizing the likelihood of production delays caused by failed reactions or out-of-specification intermediates. This reliability ensures a steady flow of materials to downstream formulation sites, supporting uninterrupted market availability of the finished dosage forms.
- Scalability and Environmental Compliance: The process conditions described in the patent are inherently scalable, having been designed to function effectively in standard reactor configurations without requiring exotic equipment or extreme pressure conditions. The ability to operate in aqueous or mixed aqueous-organic solvent systems simplifies solvent recovery and recycling operations, facilitating compliance with increasingly stringent environmental regulations regarding volatile organic compound emissions. This scalability ensures that production volumes can be ramped up rapidly to meet market demand surges while maintaining a low environmental footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the feasibility and advantages of the technology for potential licensees and manufacturing partners.
Q: What is the key innovation in patent CN101084197B for Rosuvastatin synthesis?
A: The patent introduces a novel Heck reaction coupling a fully substituted, sterically hindered pyrimidine with a vinyl side chain, overcoming traditional limitations associated with Wittig reactions and phosphine oxide byproducts.
Q: Which palladium catalyst is preferred for this Heck coupling process?
A: The patent specifically highlights bis(tri-tert-butylphosphine)palladium(0) as a particularly suitable catalyst, often used in combination with bulky tertiary amines or inorganic bases in solvents like water or DMF.
Q: How does this process improve supply chain reliability for API manufacturers?
A: By utilizing more stable intermediates and avoiding complex phosphine-based reagents, the process simplifies purification and reduces the risk of supply disruptions associated with specialized reagents, enhancing overall manufacturing continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rosuvastatin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our technical team is well-versed in the nuances of palladium-catalyzed cross-coupling reactions and maintains stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of Rosuvastatin intermediate meets the highest international standards. We understand the critical nature of API supply chains and are committed to delivering high-purity pharmaceutical intermediates that enable our partners to bring life-saving medications to patients faster and more cost-effectively.
We invite you to engage with our technical procurement team to discuss how this advanced Heck coupling technology can be integrated into your existing supply network. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your operation. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and competitiveness in the global pharmaceutical market.
