Advanced Synthesis of 3-Chloro-5-Bromophenol for Commercial Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for synthesizing complex aromatic intermediates that balance high purity with economic feasibility. Patent CN102146022A introduces a significant advancement in the preparation of 3-chloro-5-bromophenol, a critical building block for various medicinal compounds. This innovative protocol utilizes 2-chloro-4-nitroaniline as a primary feedstock, leveraging a sequence of bromination, deamination, hydrogenation, and hydrolysis reactions to achieve superior results. Unlike traditional methods that often suffer from harsh conditions or low selectivity, this approach emphasizes mild reaction parameters and simplified post-treatment procedures. For R&D directors and procurement specialists, understanding the nuances of this patent is essential for optimizing supply chains and ensuring the consistent availability of high-purity pharmaceutical intermediates. The strategic implementation of this technology allows manufacturers to bypass common bottlenecks associated with poly-substituted benzene derivatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of halogenated phenols like 3-chloro-5-bromophenol has been plagued by significant technical challenges that hinder large-scale production efficiency. Conventional routes often rely on direct halogenation of phenol, which frequently results in poor regioselectivity and the formation of difficult-to-separate isomeric byproducts. These impurities not only lower the overall yield but also necessitate expensive and time-consuming purification steps such as repeated crystallization or column chromatography. Furthermore, many traditional processes require extreme temperatures or the use of hazardous reagents that pose safety risks and increase environmental compliance costs. The inability to precisely control the position of substituents on the aromatic ring leads to inconsistent batch quality, which is unacceptable for strict pharmaceutical applications. Consequently, manufacturers face elevated production costs and extended lead times, compromising their competitiveness in the global market for fine chemical intermediates.
The Novel Approach
In stark contrast, the methodology outlined in CN102146022A offers a streamlined and highly controlled pathway that addresses these historical inefficiencies through a logical four-step sequence. By starting with 2-chloro-4-nitroaniline, the process leverages the directing effects of existing substituents to ensure precise bromination at the desired position, effectively eliminating isomer issues from the outset. 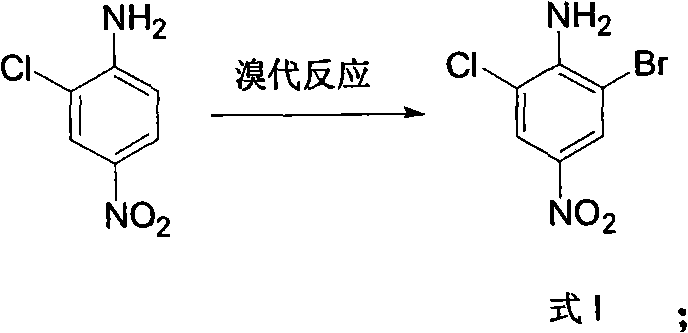 . The subsequent transformation involves a clever deamination strategy where the amino group is replaced by hydrogen via diazotization and ethanol reduction, simplifying the molecular architecture without losing the critical halogen atoms. This route operates under relatively mild conditions, typically between 10°C to 40°C for the initial steps, which reduces energy consumption and equipment stress. The result is a process that delivers high product content with simple post-treatment operations, making it an ideal candidate for cost reduction in fine chemical manufacturing and reliable pharmaceutical intermediate supplier strategies.
. The subsequent transformation involves a clever deamination strategy where the amino group is replaced by hydrogen via diazotization and ethanol reduction, simplifying the molecular architecture without losing the critical halogen atoms. This route operates under relatively mild conditions, typically between 10°C to 40°C for the initial steps, which reduces energy consumption and equipment stress. The result is a process that delivers high product content with simple post-treatment operations, making it an ideal candidate for cost reduction in fine chemical manufacturing and reliable pharmaceutical intermediate supplier strategies.
Mechanistic Insights into Diazotization and Catalytic Hydrogenation
A deep dive into the reaction mechanism reveals why this specific sequence is so effective for producing high-purity 3-chloro-5-bromophenol. The initial bromination step is governed by the electronic properties of the nitro and amino groups, where the amino group directs the incoming bromine electrophile to the ortho position relative to itself, which corresponds to the meta position relative to the nitro group. Following this, the removal of the amino group is achieved through the formation of a diazonium salt intermediate, which is then reduced by ethanol acting as a hydrogen donor. This deamination step is crucial as it resets the substitution pattern for the final functionalization.  . The subsequent hydrogenation step utilizes active nickel catalysts under moderate pressure, typically around 3atm, to reduce the nitro group back to an amine efficiently. This restoration of the amino group is a strategic move to enable the final conversion to a phenol, demonstrating a sophisticated understanding of functional group interconversion that maximizes yield while minimizing side reactions.
. The subsequent hydrogenation step utilizes active nickel catalysts under moderate pressure, typically around 3atm, to reduce the nitro group back to an amine efficiently. This restoration of the amino group is a strategic move to enable the final conversion to a phenol, demonstrating a sophisticated understanding of functional group interconversion that maximizes yield while minimizing side reactions.
Impurity control is inherently built into this synthetic design, particularly during the final hydrolysis stage where the diazonium salt is converted to the phenol. The patent highlights a surprising finding where the addition of sodium sulfate during the hydrolysis reaction markedly improves the yield compared to standard aqueous hydrolysis. This additive likely stabilizes the diazonium intermediate or modifies the ionic strength of the solution to favor the desired nucleophilic attack by water over competing decomposition pathways. By strictly controlling the temperature during diazotization, keeping it below 5°C, the formation of tar-like byproducts is suppressed, ensuring a cleaner reaction profile. Such meticulous attention to reaction conditions ensures that the final 3-chloro-5-bromophenol meets stringent purity specifications required for downstream drug synthesis. This level of mechanistic control is what distinguishes a laboratory curiosity from a commercially viable process for the commercial scale-up of complex aromatic intermediates.
How to Synthesize 3-Chloro-5-Bromophenol Efficiently
Implementing this synthesis route requires careful adherence to the specified stoichiometric ratios and thermal profiles to replicate the high yields reported in the patent embodiments. The process begins with the dissolution of 2-chloro-4-nitroaniline in glacial acetic acid, followed by the controlled dropwise addition of liquid bromine to manage exothermicity. Operators must maintain the reaction temperature within the optimal range of 20°C to 30°C to prevent poly-bromination while ensuring complete conversion of the starting material. Subsequent steps involve precise pH adjustments and temperature ramps, particularly during the diazotization phases where stability is temperature-dependent. The detailed standardized synthesis steps see the guide below for specific operational parameters and safety precautions necessary for scaling this chemistry.
- Perform regioselective bromination of 2-chloro-4-nitroaniline using liquid bromine in acetic acid at controlled temperatures.
- Execute diazotization followed by ethanol reduction to replace the amino group with hydrogen, forming the nitro-bromo intermediate.
- Conduct catalytic hydrogenation to reduce the nitro group to an amine, followed by diazotization and hydrolysis with sodium sulfate to yield the final phenol.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented method offers substantial benefits that translate directly into improved margins and operational reliability for chemical buyers. The reliance on 2-chloro-4-nitroaniline as a starting material is a strategic advantage, as this commodity chemical is widely produced and easily sourced from multiple suppliers, mitigating the risk of raw material shortages. The simplicity of the reaction steps means that existing multipurpose reactors can be utilized without requiring specialized high-pressure or cryogenic equipment, lowering the barrier to entry for production. This accessibility facilitates reducing lead time for high-purity phenols, allowing suppliers to respond more敏捷 ly to fluctuating market demands. Furthermore, the mild reaction conditions reduce energy consumption and wear on manufacturing assets, contributing to long-term sustainability goals and lower overhead costs.
- Cost Reduction in Manufacturing: The elimination of complex purification stages significantly lowers the operational expenditure associated with solvent usage and waste disposal. By achieving high selectivity in the bromination step, the need for expensive chromatographic separation is removed, relying instead on simple filtration and crystallization techniques. The use of ethanol as a reducing agent in the deamination step is also economically favorable compared to costly metal hydrides or catalytic hydrogenation for that specific transformation. These cumulative efficiencies result in substantial cost savings that can be passed down the supply chain or retained as improved margin. Additionally, the high yield per batch means less raw material is wasted, further optimizing the cost structure of the final API intermediate.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures consistent batch-to-batch quality, which is critical for maintaining trust with downstream pharmaceutical partners. Because the process does not rely on exotic catalysts or unstable reagents, the risk of production delays due to supply chain disruptions for specialized chemicals is minimized. The ability to operate at near-ambient pressures and temperatures enhances plant safety, reducing the likelihood of unplanned shutdowns due to safety incidents. This stability allows for better production planning and inventory management, ensuring a steady flow of materials to customers. Ultimately, this reliability strengthens the partnership between the manufacturer and the client, fostering long-term contracts and strategic collaboration.
- Scalability and Environmental Compliance: Scaling this process from kilogram to tonnage levels is straightforward due to the absence of extreme thermal or pressure hazards. The waste streams generated are primarily aqueous and organic solvents that can be treated using standard effluent treatment protocols, simplifying regulatory compliance. The high atom economy of the bromination and the efficient recovery of solvents contribute to a greener manufacturing footprint. This alignment with environmental standards is increasingly important for multinational corporations seeking sustainable suppliers. The process design inherently supports the principles of green chemistry, making it an attractive option for companies aiming to reduce their carbon footprint while maintaining production efficiency.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 3-chloro-5-bromophenol using this specific patent methodology. These answers are derived directly from the technical specifications and experimental data provided in the source documentation to ensure accuracy. Understanding these details helps stakeholders make informed decisions about integrating this intermediate into their broader supply networks.
Q: What is the key advantage of using sodium sulfate in the hydrolysis step?
A: The addition of sodium sulfate during the hydrolysis of the diazonium salt significantly improves the reaction yield compared to processes without this additive, ensuring higher efficiency.
Q: Is the starting material 2-chloro-4-nitroaniline readily available?
A: Yes, 2-chloro-4-nitroaniline is a commercially abundant and cost-effective raw material, which contributes to the overall economic viability of this synthesis route.
Q: What are the typical reaction conditions for the bromination step?
A: The bromination is preferably conducted at mild temperatures between 20°C to 30°C using glacial acetic acid as the solvent, ensuring safety and selectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Chloro-5-Bromophenol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development pipelines. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 3-chloro-5-bromophenol meets the highest industry standards. We understand the complexities of aromatic substitution chemistry and have the technical capability to optimize this patented route for maximum efficiency and yield. Partnering with us means gaining access to a team dedicated to technical excellence and supply chain stability.
We invite you to contact our technical procurement team to discuss how we can support your specific requirements with a Customized Cost-Saving Analysis. Whether you need specific COA data for regulatory filings or route feasibility assessments for new projects, we are ready to provide the detailed information you need. Let us help you secure a stable supply of this vital intermediate while optimizing your overall production costs. Reach out today to initiate a conversation about your upcoming projects and discover the NINGBO INNO PHARMCHEM difference.
