Advanced Synthesis of 3-Chloro-5-Bromophenol for High-Purity Pharmaceutical Applications
The pharmaceutical industry constantly demands intermediates with exceptional purity and structural integrity to ensure the safety and efficacy of final drug products. A significant breakthrough in the synthesis of critical phenolic compounds is detailed in patent CN102146022B, which outlines a robust four-step method for preparing 3-chloro-5-bromophenol. This specific intermediate is vital for constructing complex molecular scaffolds used in various therapeutic areas. The disclosed methodology leverages 2-chloro-4-nitroaniline as a foundational starting material, utilizing a sequence of bromination, diazotization, hydrogenation, and hydrolysis. By strictly adhering to the reaction parameters defined in this intellectual property, manufacturers can achieve high product content while maintaining mild reaction conditions. This report analyzes the technical merits of this route, providing R&D and procurement leaders with a clear understanding of its viability for commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional routes to substituted phenols often suffer from significant regioselectivity issues, where controlling the position of halogen substituents on the aromatic ring proves challenging and costly. Direct bromination of phenol typically results in a mixture of ortho and para isomers, necessitating expensive and yield-reducing separation processes that complicate the supply chain. Furthermore, many legacy methods rely on harsh reaction conditions, such as extreme temperatures or the use of hazardous Lewis acids, which pose safety risks and increase the burden on waste treatment facilities. The inability to precisely direct the substitution pattern without multiple protection and deprotection steps leads to elongated synthetic timelines and inflated production costs. These inefficiencies create bottlenecks for procurement managers seeking reliable sources of high-purity building blocks, as the variability in crude quality can disrupt downstream synthesis.
The Novel Approach
The method described in the patent data overcomes these historical challenges by employing a strategic sequence that utilizes the directing effects of existing functional groups to ensure precise substitution. By starting with 2-chloro-4-nitroaniline, the process leverages the strong deactivating nature of the nitro group and the directing influence of the amino group to achieve specific bromination at the desired position without isomer contamination. The initial step involves a controlled bromination reaction in acetic acid at room temperature, which eliminates the need for energy-intensive heating or cooling systems. This approach not only simplifies the operational protocol but also significantly reduces the risk of side reactions that typically degrade product quality. The subsequent transformation steps are designed to be operationally simple, with easy post-treatment procedures that facilitate high recovery rates of the target molecule.
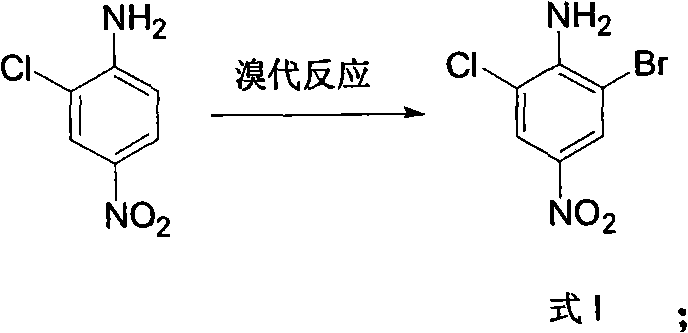
Mechanistic Insights into Diazotization and Hydrogenation
A deep understanding of the reaction mechanism is crucial for R&D directors evaluating the robustness of this synthetic pathway. The core of this methodology lies in the precise manipulation of the aniline functionality through diazotization, followed by distinct substitution strategies. In the second step, the amino group is converted into a diazonium salt using sodium nitrite and sulfuric acid, which is subsequently reduced by ethanol to effect a hydrogen substitution. This deamination step is critical for resetting the substitution pattern on the ring, allowing for the subsequent reduction of the nitro group without interference. The electronic environment of the aromatic ring is carefully managed throughout the sequence to prevent unwanted nucleophilic attacks or oxidative degradation, ensuring that the chlorine and bromine substituents remain intact. This level of control is essential for maintaining the impurity profile within strict specifications required for pharmaceutical applications.
The third and fourth steps highlight the versatility of catalytic reduction and hydrolytic substitution in this process. The reduction of the nitro group to an amine is achieved using active nickel under moderate hydrogen pressure, a method known for its chemoselectivity and tolerance of halogenated substrates. Following this, the newly formed amine undergoes a second diazotization, but this time the diazonium salt is subjected to hydrolysis in the presence of sodium sulfate. The inclusion of sodium sulfate is a key mechanistic innovation, as it stabilizes the reaction medium and promotes the efficient replacement of the diazo group with a hydroxyl group. This specific modification in the hydrolysis step prevents the formation of tar-like byproducts and ensures a clean conversion to the final phenolic product, thereby enhancing the overall purity of the 3-chloro-5-bromophenol.

How to Synthesize 3-Chloro-5-Bromophenol Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and temperature control to maximize the benefits outlined in the patent documentation. The process is designed to be scalable, moving from laboratory benchtop to industrial reactors with minimal modification to the core chemical principles. Operators must ensure that the bromination is conducted at room temperature to prevent poly-bromination, and the diazotization steps must be kept cool to maintain the stability of the diazonium intermediates. The detailed standardized synthesis steps see the guide below for specific weight ratios and timing.
- Bromination of 2-chloro-4-nitroaniline using liquid bromine in acetic acid at room temperature to form 2-chloro-4-nitro-6-bromoaniline.
- Diazotization followed by hydrogen substitution using sulfuric acid, ethanol, and sodium nitrite to remove the amino group.
- Catalytic hydrogenation of the nitro group using active nickel under pressure to form 3-chloro-5-bromoaniline.
- Final diazotization and hydrolysis in the presence of sodium sulfate to substitute the amino group with a hydroxyl group.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the technical advantages of this patent translate directly into tangible commercial benefits that enhance the reliability of the supply base. The use of 2-chloro-4-nitroaniline as a starting material is a strategic choice, as this chemical is widely available in the global market, reducing the risk of raw material shortages that can plague more exotic synthetic routes. The mild reaction conditions, particularly the room temperature bromination and moderate pressure hydrogenation, lower the energy consumption of the manufacturing process, contributing to substantial cost savings in utility expenses. Furthermore, the simplified post-treatment operations, which involve straightforward filtration and extraction, reduce the labor hours and solvent volumes required for purification. These factors combine to create a manufacturing process that is not only economically efficient but also resilient against market fluctuations.
- Cost Reduction in Manufacturing: The elimination of complex protection and deprotection sequences significantly streamlines the production workflow, removing the need for expensive reagents and additional unit operations. By avoiding the use of precious metal catalysts in favor of active nickel, the process reduces the capital tied up in catalyst recovery and regeneration systems. The high yield achieved through the sodium sulfate-assisted hydrolysis minimizes raw material waste, ensuring that a greater proportion of the input mass is converted into saleable product. This efficiency drives down the cost per kilogram, allowing for more competitive pricing strategies in the global market for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as acetic acid, ethanol, and sulfuric acid ensures that the supply chain is not vulnerable to the bottlenecks associated with specialized reagents. The robustness of the reaction conditions means that production can be maintained consistently across different seasons and geographic locations without significant variance in output quality. This stability is critical for long-term supply agreements, as it guarantees the continuity of material flow to downstream drug manufacturers. The ability to scale this process from small batches to multi-ton production runs without re-optimizing the chemistry provides a secure foundation for meeting increasing market demand.
- Scalability and Environmental Compliance: The process generates fewer hazardous byproducts compared to traditional halogenation methods, simplifying the waste treatment process and reducing the environmental footprint of the facility. The use of ethanol as a solvent and reducing agent is preferable from a safety and regulatory standpoint, as it is less toxic and easier to handle than many organic alternatives. The straightforward workup procedures reduce the volume of organic waste streams, aligning with modern green chemistry principles and regulatory requirements. This environmental compatibility facilitates smoother permitting processes and enhances the sustainability profile of the final product, which is increasingly important for corporate social responsibility goals.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route. These answers are derived directly from the experimental data and beneficial effects described in the patent literature, providing clarity on the operational parameters. Understanding these details is essential for technical teams evaluating the feasibility of adopting this method for their specific production needs.
Q: What is the key advantage of using sodium sulfate in the final hydrolysis step?
A: The addition of sodium sulfate during the hydrolysis of the diazonium salt significantly increases the reaction yield compared to processes that omit this salt, ensuring better material efficiency.
Q: Are the reaction conditions suitable for large-scale manufacturing?
A: Yes, the process utilizes mild conditions such as room temperature for the initial bromination and moderate pressure for hydrogenation, which facilitates safer and more scalable commercial production.
Q: What is the starting material for this synthesis route?
A: The synthesis begins with 2-chloro-4-nitroaniline, which is a cheap and readily available raw material, contributing to overall cost reduction in the supply chain.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Chloro-5-Bromophenol Supplier
NINGBO INNO PHARMCHEM stands at the forefront of chemical manufacturing, leveraging deep technical expertise to bring complex synthetic routes like this one to commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent to plant is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3-chloro-5-bromophenol meets the exacting standards required by the pharmaceutical industry. Our commitment to quality and consistency makes us a trusted partner for companies seeking to secure their supply chain for critical intermediates.
We invite you to collaborate with us to optimize your sourcing strategy and achieve significant efficiencies in your production costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and proven performance. Partnering with us ensures access to a reliable supply of high-quality intermediates backed by world-class manufacturing capabilities.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
